10 The functional differences between the various tissues of the skeletal system relate principally to the different nature and proportion of the ground substance and fibrous elements of the extracellular matrix. The cells of all the skeletal tissues, like the cells of the less specialised supporting/connective tissues, have close structural and functional relationships and a common origin from primitive mesenchymal cells (see Ch. 4). Proteoglycans (see Ch. 4), disposed in proteoglycan aggregates of 100 or more molecules, make up the ground substance and account for the solid, yet flexible, consistency of cartilage. Sulphated glycosaminoglycans (GAGs, chondroitin sulphate and keratan sulphate) predominate in the proteoglycan aggregates, with molecules of the non-sulphated GAG hyaluronic acid forming the central backbone of the complex. The different types of cartilage vary in the amount and nature of fibres in the ground substance: hyaline cartilage contains few fibres, fibrocartilage contains abundant collagen fibres and elastic cartilage contains elastin fibres. Cartilage formation commences with the differentiation of stellate-shaped primitive mesenchymal cells (see Fig. 4.2) to form rounded cartilage precursor cells called chondroblasts. Subsequent mitotic divisions give rise to aggregations of closely packed chondroblasts which grow and begin synthesis of ground substance and fibrous extracellular material. Secretion of extracellular material traps each chondroblast within the cartilaginous matrix, thereby separating the chondroblasts from one another. Each chondroblast then undergoes one or two further mitotic divisions to form a small cluster of mature cells separated by a small amount of extracellular material. The role of cartilage in bone formation is discussed in Figs 10.18 to 10.21. FIG. 10.1 Hyaline cartilage FIG. 10.2 Fibrocartilage FIG. 10.3 Elastic cartilage FIG. 10.4 Chondrocyte • Osteoblasts which synthesise osteoid and mediate its mineralization. These are found lined up along bone surfaces. • Osteocytes which represent largely inactive osteoblasts trapped within formed bone. These may assist in the nutrition of bone. • Osteoclasts are phagocytic cells which are capable of eroding bone. These are important, along with osteoblasts, in the constant turnover and refashioning of bone. Bone forms the strong and rigid endoskeleton to which skeletal muscles are attached to permit movement. It also acts as a calcium reservoir and is important in calcium homeostasis. Bone is heavy and its architecture is optimally arranged to provide maximum strength for the least weight. Most bones have a dense, rigid outer shell of compact bone, the cortex, and a central medullary or cancellous zone of thin, interconnecting narrow bone trabeculae. The number, thickness and orientation of these bone trabeculae is dependent upon the stresses to which the particular bone is exposed. For example, there are many thick intersecting trabeculae in the constantly weight-bearing vertebrae, but very few in the centre of the ribs, which are not subjected to constant stress. The space in the medullary bone between trabeculae is occupied by haematopoietic bone marrow (see Fig. 3.3). FIG. 10.5 Active osteoblasts and osteoid FIG. 10.6 Osteoclasts FIG. 10.7 Woven and lamellar bone, polarising microscopy FIG. 10.8 Long bone FIG. 10.9 Fracture callus FIG. 10.10 Cortical (compact) bone
Skeletal tissues
Introduction
Cartilage
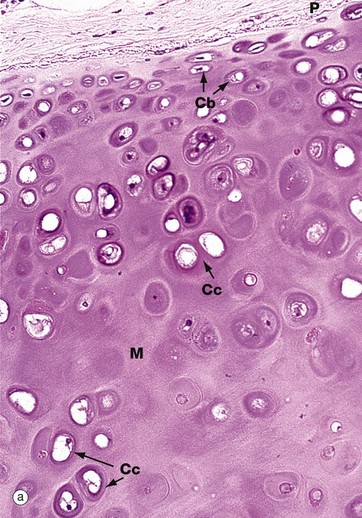
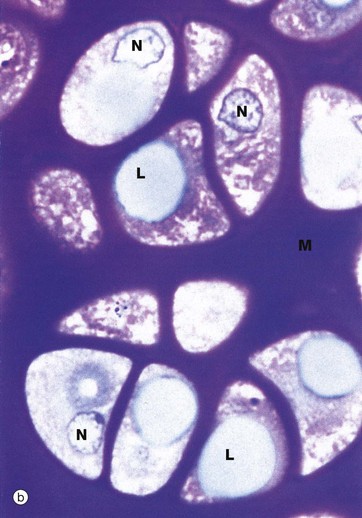
(a) H&E (MP) (b) Thin epoxy resin section, toluidine blue (HP)
Hyaline cartilage is the most common type of cartilage. It is found in the nasal septum, larynx, tracheal rings, most articular surfaces and the sternal ends of the ribs. It also forms the precursor of bone in the developing skeleton. Mature hyaline cartilage is characterised by small aggregates of chondrocytes embedded in an amorphous matrix of ground substance, reinforced by collagen fibres.
Micrograph (a) shows a hyaline cartilage mass with its outer perichondrium P. The chondrocytes of the formed cartilage Cc are arranged in clusters, usually of 2 to 4 cells, each cluster being separated from its neighbours by amorphous cartilage matrix M. The perichondrium is composed of parallel collagen fibres containing a few spindle-shaped nuclei of inactive fibrocytes but, on its inner surface, these cells are transforming into small chondroblasts Cb which are in the process of enlarging, dividing and synthesising new cartilage matrix.
The matrix of hyaline cartilage appears fairly amorphous, since the ground substance and collagen have similar refractive properties. With the exception of articular cartilage, the collagen of hyaline cartilage, designated as type II collagen (see Ch. 4), is not cross-banded and is arranged in an interlacing network of fine fibrils. This collagen cannot be demonstrated by light microscopy.
The thin epoxy resin section of hyaline cartilage in micrograph (b) shows the cellular details of mature chondrocytes. Note that the chondrocytes fully occupy the spaces in the matrix M, each space containing a single chondrocyte. Mature chondrocytes are characterised by small nuclei N with dispersed chromatin and basophilic granular cytoplasm, reflecting a well-developed rough endoplasmic reticulum. Lipid droplets L, often larger than the nuclei, are a prominent feature of larger chondrocytes. The cytoplasm is also rich in glycogen. These characteristics reflect the active role of chondrocytes in synthesis of both the ground substance and fibrous elements of the cartilage matrix. In fully formed cartilage, the constituents of the extracellular matrix are continuously turned over, the integrity of the matrix being thus absolutely dependent on the viability of the chondrocytes.
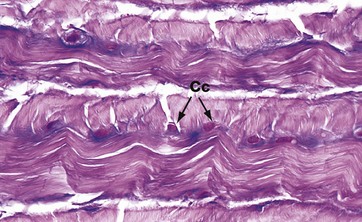
H&E/Alcian blue (HP)
Fibrocartilage has features intermediate between cartilage and dense fibrous supporting tissue. It is found in intervertebral discs, some articular cartilages, the pubic symphysis, in association with dense collagenous tissue in joint capsules, in ligaments and in the connections of some tendons to bone. It consists of alternating layers of hyaline cartilage matrix with thick layers of dense collagen fibres, orientated in the direction of the functional stresses.
This micrograph is taken from the same specimen of intervertebral disc illustrated in Fig. 10.30. Pink-stained collagen characteristically permeates the blue-stained cartilage ground substance. Chondrocytes Cc are arranged in rows between the dense collagen layers within lacunae in the glycoprotein matrix.
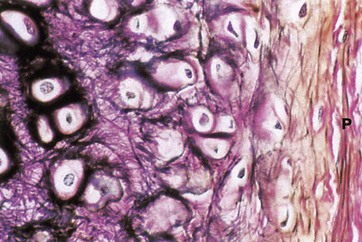
Elastic van Gieson (HP)
Elastic cartilage occurs in the external ear and external auditory canal, the epiglottis, parts of the laryngeal cartilages and in the walls of the Eustachian tubes.
The histological structure of elastic cartilage is similar to that of hyaline cartilage. Its elasticity is derived from the presence of numerous bundles of branching elastin fibres in the cartilage matrix. This network of elastin fibres (stained black in this preparation) is particularly dense in the immediate vicinity of the chondrocytes. Collagen (stained red) is also a major constituent of the cartilage matrix and makes up the bulk of the perichondrium P, intermingled with a few elastic fibres. Development and growth of elastic cartilage occurs by both interstitial and appositional growth, in the same manner as for hyaline cartilage.
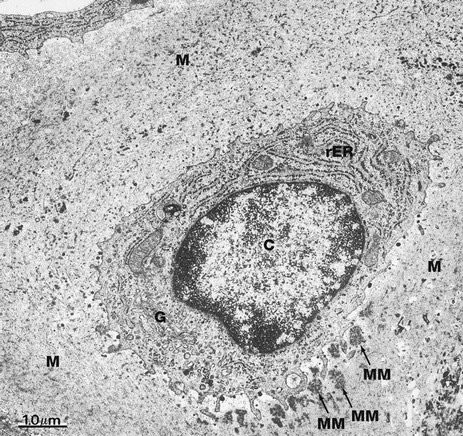
EM ×16 000
This electron micrograph illustrates a single chondrocyte C lying within its lacuna and surrounded by abundant cartilaginous matrix material M. As is typical of cells active in protein synthesis (in this case matrix turnover), chondrocytes have very prominent rough endoplasmic reticulum rER which is distended with secretory material. A well-developed Golgi apparatus G is present. Some glycogen granules are scattered in the cytoplasm. Note that the chondrocyte completely fills its lacuna within the matrix. Small cytoplasmic extensions mediate the constant interaction between chondrocytes and the surrounding extracellular matrix material.
At this magnification, the fibrous elements of the extracellular matrix can just be discerned. The deposits of electron-dense material which can be seen lying adjacent to the deep aspect of the cell represent some recently secreted extracellular matrix material MM.
Bone
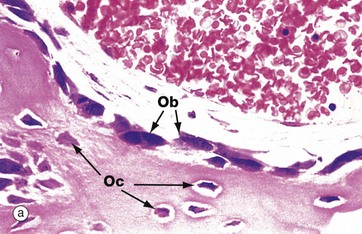
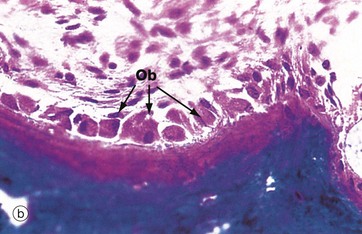
(a) H&E (HP) (b) Undecalcified resin section, Goldner trichrome stain (HP)
These micrographs illustrate osteoblasts actively depositing new osteoid on a bone surface. When active, the osteoblasts Ob are large, broad, spindle-shaped or cuboidal cells with abundant basophilic cytoplasm containing much rough endoplasmic reticulum and a large Golgi apparatus. These features reflect a high rate of protein (type I collagen) and proteoglycan synthesis.
In micrograph (a), the tissue has been decalcified before sectioning and staining, so the distinction between mineralised bone and the newly formed unmineralised osteoid cannot be seen. In micrograph (b), which has not been decalcified, the mineralised bone (blue) can easily be distinguished from the new osteoid (red) which is being produced by the row of cuboidal osteoblasts. There is always a short delay between osteoid production and its mineralisation.
When inactive, osteoblasts are narrow, attenuated, spindle-shaped cells lying on the bone surface. In (a) the burst of new bone formation is nearly over, and the osteoblasts are becoming spindle-shaped again and will soon become virtually undetectable, only the long, narrow nucleus being visible histologically. A few cells are being incorporated in the newly formed bone as osteocytes Oc.
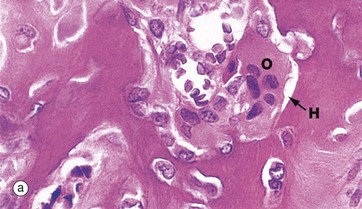
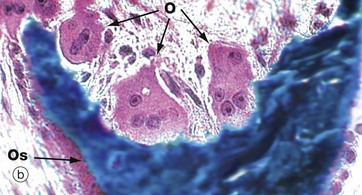
(a) H&E (HP) (b) Undecalcified resin section, Goldner trichrome (HP)
Resorption of bone is performed by large multinucleate cells called osteoclasts O, which are often seen lying in depressions resorbed from the bone surface called Howship lacunae H. The aspect of the osteoclast in apposition to bone is characterised by fine microvilli which form a ruffled border that is readily visible with the electron microscope. The ruffled border secretes several organic acids which dissolve the mineral component, while lysosomal proteolytic enzymes are employed to destroy the organic osteoid matrix.
Osteoclastic resorption contributes to bone remodelling in response to growth or due to changing mechanical stresses upon the skeleton. Osteoclasts also participate in the long-term maintenance of blood calcium homeostasis by their response to parathyroid hormone and calcitonin (see Ch. 17). Parathyroid hormone stimulates osteoclastic resorption and so increases the release of calcium ions from bone, whereas calcitonin inhibits osteoclastic activity.
Micrographs (a) and (b) are taken from bone showing excessive osteoclastic activity due to the effects of Paget disease of bone, a disorder characterised by continuous disorganised bone resorption and associated new bone formation (see textbox). Micrograph (b) shows uncoordinated new osteoid formation by a row of cuboidal osteoblasts Ob.

H&E (MP)
Bone exists in two main forms, woven bone W and lamellar bone L. Woven bone is an immature form with randomly arranged collagen fibres in the osteoid. Lamellar bone is composed of regular parallel bands of collagen arranged in sheets.
Woven bone is produced when osteoblasts synthesise osteoid rapidly, as in fetal bone development. It can also occur in adults when there is pathological rapid new bone formation e.g. at the site of a healing fracture and in Paget disease (see textbox).
The rapidly formed woven bone is eventually remodelled to form lamellar bone, which is physically stronger and more resilient. Virtually all bone in a healthy adult is lamellar.
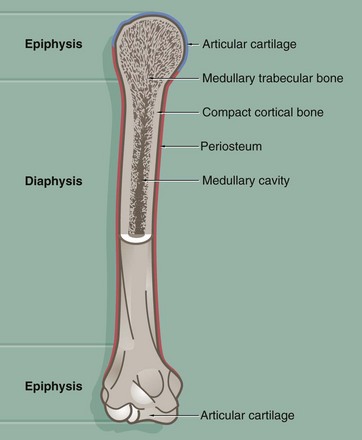
This diagram illustrates the general structure of long bones in the mature skeleton and the gross morphological appearance of the two types of lamellar bone found in the mature skeleton, i.e. compact (cortical) bone and cancellous (medullary) bone.
Compact bone forms the dense walls of the shaft or diaphysis, while cancellous bone occupies part of the large central medullary cavity. Cancellous bone consists of a network of fine, irregular plates called trabeculae separated by intercommunicating spaces.
The articular (joint) surfaces of the expanded ends, or epiphyses, of long bones are protected by a layer of specialised hyaline cartilage called articular cartilage. The external surface of the bone is invested in a dense fibrous layer called the periosteum, into which are inserted muscles, tendons and ligaments. The inner surfaces of the bone, including the trabeculae of cancellous bone, are invested by a delicate layer called the endosteum. The endosteum and periosteum contain cells of the osteogenic series which are responsible for growth, continuous remodelling and repair of bone fractures (see textbox).
Prior to the attainment of skeletal maturity, the long bones grow in length by the process of endochondral ossification which occurs at a growth or epiphysial plate situated at each end of the bone at the junction of the diaphysis (shaft) and epiphysis.
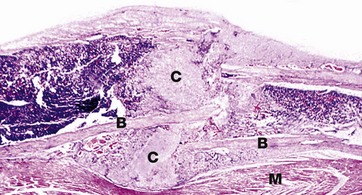
H&E (LP)
This image shows fracture callus C around the site of a rib fracture. This mass of healing tissue acts to stabilise the broken ends of the bone B so that repair can occur. There is new bone formation within the callus and, over time, this will become organised into mature lamellar bone. Striated muscle M is present at the lower border of the bone.
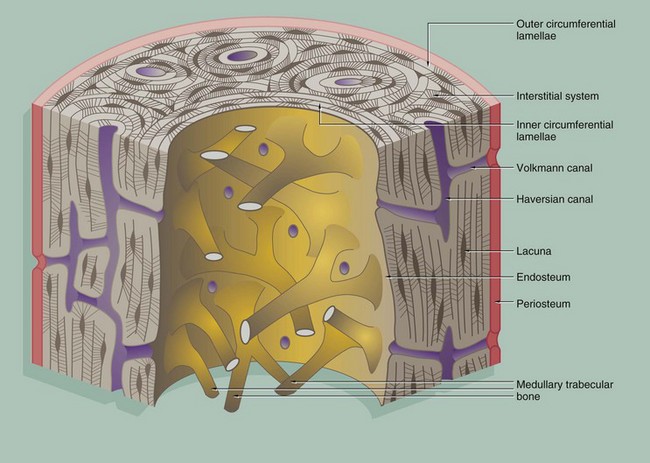
Compact bone is made up of parallel bony columns which, in long bones, are disposed parallel to the long axis, i.e. in the line of stress exerted on the bone. Each column is made up of concentric bony layers or lamellae arranged around a central channel containing blood vessels, lymphatics and nerves. These neurovascular channels are known as canals of Havers or Haversian canals and, with their concentric lamellae, form Haversian systems. The neurovascular bundles interconnect with one another and with the endosteum and periosteum via Volkmann canals which pierce the columns at right angles (or obliquely) to the Haversian canals.
Each Haversian system (osteon) develops by osteoclastic tunnelling of a mass of compact bone to form a broad channel into which blood vessels and nerves grow, after which it becomes lined internally by active osteoblasts which lay down concentric lamellae of bone.
With the deposition of successive lamellae, the diameter of the Haversian canal decreases and osteoblasts are trapped as osteocytes in spaces called lacunae in the matrix. The osteocytes are thus arranged in concentric rings within the lamellae. Between adjacent lacunae and the central canal, there are numerous minute interconnecting canals called canaliculi which contain fine cytoplasmic extensions of the entrapped osteocytes.
As a result of the continuous resorption and redeposition of bone, complete newly formed Haversian systems are disposed between partly resorbed systems formed earlier. The remnants of lamellae no longer surrounding Haversian canals form irregular interstitial systems between intact Haversian systems.
At the outermost aspect of compact bone, Haversian systems give way to concentric lamellae of dense cortical bone, laid down partly by the osteoblasts of the periosteum (outer circumferential lamellae). Similar circumferential lamellae line the inside of the cortical bone (inner circumferential lamellae) where it abuts the marrow cavity.
The inner surface of cortical bone (endosteum) is composed of the innermost layer of the inner circumferential lamellae, with a layer of inactive flat osteoblasts on its surface. When activated, these cells enlarge to become active cuboidal osteoblasts and synthesise new lamellar osteoid which, on mineralisation, forms another layer of inner circumferential lamella. This occurs regularly as part of the constant dynamic refashioning of bone and is particularly prominent during bone growth. It is also seen in response to increased or altered stress on the cortical bone, for example in the leg bones during periods of increased physical training for running and other sports.
An interconnecting network of trabecular or cancellous bone occupies the central marrow cavity of the bone, and the ends of these bony trabeculae are attached to the inner circumferential lamellae of the cortical bone (see Fig. 10.13). The inactive osteoblasts of the endosteum also extend onto the surface of the trabecular bone and similarly deposit new osteoid when required for strengthening or remodelling.
Small blood vessels and nerves enter the cortical bone from the marrow space through defects in the endosteum and inner circumferential lamellae. These connect with the Volkmann canals, which in turn connect with Haversian canals.![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Basicmedical Key
Fastest Basicmedical Insight Engine
