Fig. 17.1
Relational (Venn) diagram showing distribution of study patients and embryos
In this study group, the mean (±SD) number of oocytes which underwent fertilization by ICSI was 17.7 ± 7.8 (range = 6–35), and this yielded an average of 15.1 ± 6.7 2pn zygotes per patient (range = 6–32). Most embryos (86 %) were biopsied on day 3, while the remainder (14 %) were biopsied on day 5. Although the number of blastocyst biopsies was relatively small (n = 44), it was possible to record embryo ploidy as a function of biopsy timing. Using this approach, we found the incidence of missed calls (“no signal”) on chromosomes to be significantly higher among embryos biopsied at day 3, resulting in reduced reporting efficiency for this group compared to the blastocyst biopsy group (92 % vs. 100 %; p = 0.05).
Assessment of all embryos produced from oocytes contributed by an anonymous donor identified euploidy in 133 of 284 (46.8 %) embryos with full chromosomal reporting (i.e., zero “no calls”). Complete data on all 23 chromosome pairs was reported for 93.1 % of embryos sampled (284 of 305). Considering all embryo chromosomes, mean error rate was 18 %. A chromosome-specific analysis found error present in all chromosomes; chromosome 22 was most often affected, and chromosome 15 was the least likely to have an abnormality (see Fig. 17.2). The relatively high Phi correlation coefficients (see Fig. 17.3) among embryo chromosome pairs with aneuploidy (r = 0.60, range 0.42–0.77; p < 0.01 by chi-square test) indicate that chromosomes tend to have multiple and simultaneous errors (complex aneuploidy).
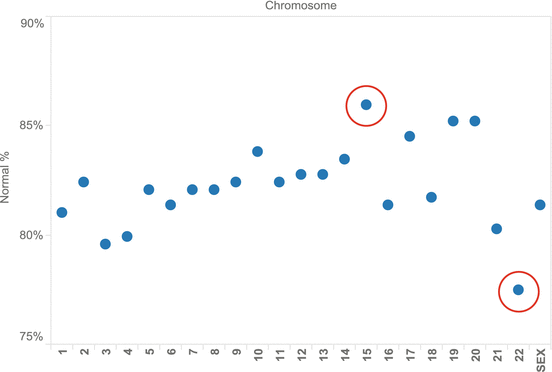
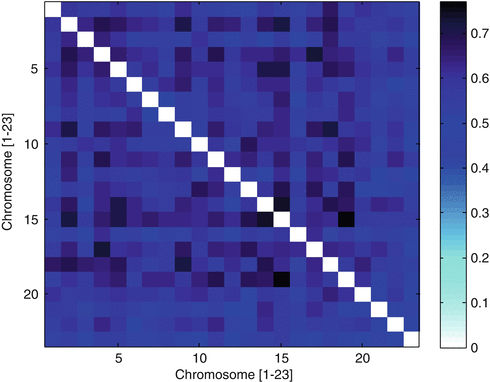

Fig. 17.2
Distribution of aneuploidy as a function of specific chromosomal error measured in embryos (n = 305) produced from anonymous donor oocyte IVF cycles

Fig. 17.3
Pairwise correlations of autosomal aneuploidy by mean square contingency (Phi) coefficient, observed in 305 embryos derived from anonymous donor oocyte IVF treatments
When analysis was confined only to those embryos with no missed calls for any chromosome, errors attributable to a maternal source (i.e., from the oocyte donor) were noted in 133 of 284 embryos (46.8 %). Conversely, an embryo genetic abnormality of paternal origin was present in 104 of 284 embryos (36.6 %). Among all aneuploid embryos (n = 151), chromosomal errors from both genetic parents (i.e., oocyte donor and partner’s husband) were present in 57.0 % (see Fig. 17.4). While oocyte donor age ranged from 20 to 29 years, some genetically abnormal embryos were produced from donors of each age, and there was no correlation between oocyte donor age and embryo aneuploidy. Likewise, these data did not confirm a correlation between embryo aneuploidy and male partner age or any semen parameter.
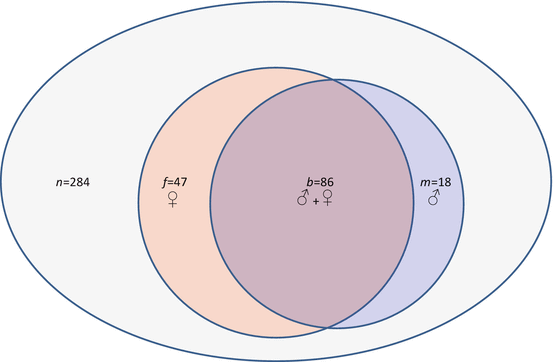

Fig. 17.4
Distribution of aneuploidy origin by gamete source for embryos produced from anonymous donor oocyte IVF
Discussion
The role of PGS on the menu of clinical IVF services has evolved substantially in recent years. Although it is tempting to classify PGS applications as simply an accessory to “mainstream” IVF, genetic testing of embryos has been (and will continue to be) a crucial development in the progress of our field. Certainly the successful passage of the world’s first IVF regulatory legislation (Human Fertilisation & Embryology Act, 1990) was strongly influenced and enabled by the arrival of PGD in the United Kingdom [20]; further applications of this technique have continued to push the ethical boundaries for IVF into unfamiliar terrain [21].
In humans, most aneuploidies are triploidies, yet only those involving chromosomes 21, 18, and 13 are compatible with survival to term [22]. Duplication of other autosomes is poorly tolerated and is rarely seen in live births. Viable monosomies are only known to exist for chromosome X, while additional copies of sex chromosomes are developmentally permissive. PGS is a powerful clinical tool to assist in embryo selection to minimize transfer of such embryos, thus improving clinical outcomes.
The arrival of oocyte donation preceded PGS and was originally offered as a treatment for premature ovarian failure or oophorectomy [23]. Egg donation is now commonly in use for many settings besides diminished ovarian reserve, including its use to circumvent transmission of severe genetic disorder(s) in the birth mother to her offspring [24]. While the corrosive effect of age on female infertility can be successfully assuaged for couples using donated oocytes from a younger (presumably more fertile) woman [25], the degree of chromosomal error in embryos derived from such treatment has yielded some unexpected preliminary results [11].
For example, one IVF group recently conducted a 12-year retrospective study on genetic test data collected from anonymous oocyte donor applicants and found that genetic abnormalities caused a significant number of candidates to be excluded from their oocyte donor program [26]. We agree with this approach and, like many institutions, require any potential anonymous oocyte donor to first undergo a careful genetic testing regime before entering the roster of eligible oocyte donors. Indeed, all of the anonymous donors who supplied oocytes for the current study already had been screened for hundreds of genetic disorders in advance of their accession into our egg donor group. However, despite this reassuring clearance (and in the absence of any obvious reproductive pathology in the oocyte donors) the rate of chromosomal error among embryos produced from their eggs was surprisingly high (e.g., 55 % aneuploidy rate).
Previous research attempted to characterize the role of “defective” gametes resulting in the generation of abnormal embryos using an egg-sharing model (where one IVF patient agrees to share her eggs with another IVF patient) [27]. Unfortunately, this can yield an undesirable outcome for the recipient since what she ultimately gets are simply eggs from another infertile patient. Such a study is unsatisfying experimentally because the variable of oocyte pathology cannot be controlled if all the oocytes for study are generated by other patients with manifold infertility diagnoses.
This problem was also addressed when the aneuploidy rate for eight chromosomes in embryos derived from young (<35 years) oocyte donors using fluorescence in situ hybridization analysis was studied. Using this study approach, all oocytes were provided by healthy women who did not have any infertility diagnosis. The authors reported considerable variation between donor cycles with nearly one-third having <30 % genetically normal embryos [11]. Starting from these data where less than half of the embryo’s chromosomes had been evaluated, our work was built on this foundation to screen all 23 pairs of embryo chromosomes in an anonymous donor oocyte IVF setting. Importantly, since the behavior of each parental allelic group is a function of the underlying chromosome copy number of the embryo, and because these modifications may be satisfactorily estimated from additional allelic content contributed by (or omitted from) either the oocyte donor or the recipient’s husband (sperm source), we were able to supply additional information on the parental origin of the genetic problems identified in the embryos derived therefrom.
Earlier research has shown a significantly higher observed pregnancy loss rate among IVF patients with age ≥40 compared to women younger than age 40 [28], establishing that the distribution of genetic error in embryos as a function of maternal age is not stationary. This physiologic process of natural ovarian senescence has been sidestepped for many years by using oocytes provided by younger donors [29]. With further refinement of donor oocyte protocols, acceptance of this treatment in routine IVF practice has increased greatly over the last decade, and when donor oocytes are used, the likelihood of an excellent IVF outcome seems independent of recipient age [30]. In the United States, the incidence of twins is markedly higher among anonymous oocyte donor IVF cycles compared to IVF using native (autologous) oocytes (37 % vs. 29 %, respectively), which provides direct evidence that most clinics are not to following a current ASRM recommendation which encourages single embryo transfers when oocyte donor age is young [31]. Indeed, there now appears to be international consensus that elective single embryo transfers are appropriate for oocyte donor–recipient cycles where the donor has good prognosis and when good quality embryos are available [32].
Of note, comprehensive chromosomal screening has not been applied to embryos of donor oocyte origin to quantify the level of genetic abnormality which persists in such embryos until now. If ever the domain of anonymous donor oocyte IVF were regarded as a realm where the role of genetic error in embryos could be dismissed as unimportant, the current study highlights an important supporting role for PGS in this population of IVF patients, too. Moreover, these data provide some fresh observations on human embryo genetics. Here, we focused on the specific topic of parental origin with respect to chromosomal errors which may be harbored by IVF embryos. Our observation that a high rate of embryonic genetic anomaly could be traced back to the oocyte donor was not anticipated. Thus, it appears that the traditional view that most chromosomal errors are of maternal origin caused by malsegregation in the first meiotic division [33] remains valid, even when the age of the oocyte source is very low.
Our report has some limitations which should be acknowledged. Our data come from a retrospective analysis as an initial step to analyze readily accessible existing data. We aimed to produce a hypothesis about aneuploidy rate in embryos derived from anonymous donor oocytes which could then be tested prospectively [34]. Retrospective work has the potential for incomplete documentation, unrecoverable or unrecorded data, and variance in the quality of information recorded. The reliability of data entry is considered as high for this sample, and the proportion of incomplete records was marginal. Also, because our sample was limited and represented the chance event of an IVF patient using anonymous donor oocytes also incorporating preimplantation testing of embryos produced from this treatment, it is uncertain if these findings can generalize to all anonymous donor-egg IVF cases (it should be noted that a secondary chart review for our study population did not reveal any obvious characteristic which may have influenced the patient’s decision to include PGS in her IVF treatment). Perhaps the high economic cost of IVF in general (and donor oocyte treatment in particular) introduced some selection bias, since only the most affluent IVF patients could have afforded this treatment [35]. It would be interesting to query the remaining donor oocyte IVF patients in this series who declined PGS (n = 27), to understand better why they decided not to request genetic testing of their embryos; this represents an area of future research here. Finally, our analysis of male factor data was confined to the age of the recipient’s husband and only two semen parameters (sperm concentration and motility). We did not include sperm DNA fragmentation data in this study, although this has not yet been correlated with embryo ploidy [36].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


