Severe Sepsis and Septic Shock
KEY CONCEPTS
![]() The spectrum of microorganisms associated with sepsis has changed from predominantly gram-negative bacteria in the late 1970s and 1980s to gram-positive bacteria as the major pathogens since the late 1980s.
The spectrum of microorganisms associated with sepsis has changed from predominantly gram-negative bacteria in the late 1970s and 1980s to gram-positive bacteria as the major pathogens since the late 1980s.
![]() Candidemia is a major cause of morbidity and mortality. Candida albicans remains the most common pathogen (45.6%); however, non–albicans Candida species collectively is more frequently isolated (54.4%).
Candidemia is a major cause of morbidity and mortality. Candida albicans remains the most common pathogen (45.6%); however, non–albicans Candida species collectively is more frequently isolated (54.4%).
![]() Sepsis presents a complex pathophysiology, characterized by the activation of multiple overlapping and interacting cascades leading to systemic inflammation, a procoagulant state, and decreased fibrinolysis.
Sepsis presents a complex pathophysiology, characterized by the activation of multiple overlapping and interacting cascades leading to systemic inflammation, a procoagulant state, and decreased fibrinolysis.
![]() Mortality rates with sepsis are higher for older patients with preexisting disease, intensive care unit care, and multiple organ failure.
Mortality rates with sepsis are higher for older patients with preexisting disease, intensive care unit care, and multiple organ failure.
![]() Prompt initiation of broad-spectrum, parenteral antibiotic therapy is required due to the high incidence of complications and mortality with sepsis.
Prompt initiation of broad-spectrum, parenteral antibiotic therapy is required due to the high incidence of complications and mortality with sepsis.
![]() A significant volume of fluid leaks from the vasculature occurs with sepsis, and initial fluid resuscitation with large volumes of fluid is required. There is no difference in clinical outcomes between colloid and crystalloid fluid resuscitation.
A significant volume of fluid leaks from the vasculature occurs with sepsis, and initial fluid resuscitation with large volumes of fluid is required. There is no difference in clinical outcomes between colloid and crystalloid fluid resuscitation.
![]() Norepinephrine is generally preferred over dopamine as the vasopressor to correct hypotension in septic shock.
Norepinephrine is generally preferred over dopamine as the vasopressor to correct hypotension in septic shock.
![]() Early goal-directed therapy during the first 6 hours, consisting of hemodynamic monitoring with a central venous catheter, volume resuscitation, inotropic therapy, and red blood cell transfusions, demonstrated a significant clinical outcome benefit with a 16% absolute reduction in 28-day mortality.
Early goal-directed therapy during the first 6 hours, consisting of hemodynamic monitoring with a central venous catheter, volume resuscitation, inotropic therapy, and red blood cell transfusions, demonstrated a significant clinical outcome benefit with a 16% absolute reduction in 28-day mortality.
![]() A blood glucose level less than 150 mg/dL (8.3 mmol/L) is recommended for the majority of critically ill patients to reduce morbidity and mortality without the detrimental effects associated with hypoglycemia.
A blood glucose level less than 150 mg/dL (8.3 mmol/L) is recommended for the majority of critically ill patients to reduce morbidity and mortality without the detrimental effects associated with hypoglycemia.
![]() IV hydrocortisone is recommended for adult patients with septic shock whose blood pressure is unresponsive to fluids and vasopressors.
IV hydrocortisone is recommended for adult patients with septic shock whose blood pressure is unresponsive to fluids and vasopressors.
Sepsis and severe sepsis continue to pose major healthcare burden. The incidence of sepsis in the United States increased from 82.7 cases per 100,000 population in 1979 to 240.4 cases per 100,000 population in 2000, for an annualized increase of 8.7 percent.1 Severe sepsis increased from 200 cases per 100,000 in 2003 to 300 cases per 100,000 in 2007, a 50% increase.2 Despite aggressive medical care and advances, overall in-hospital deaths increased from 75 per 100,000 in 2003 to 87 per 100,000 in 2007, a 16% increase.2 With increasing total hospital costs to $24.3 billion, there is a vital need for clinicians to comprehend the pathophysiology and to appreciate the management options for acutely ill patients with severe sepsis or septic shock.2
DEFINITIONS
In 1992, a joint committee of the American College of Chest Physicians and the Society of Critical Care Medicine standardized the terminology related to sepsis for several reasons: (a) widespread confusion with the use of these terms, (b) the need to provide a flexible classification scheme for patient identification, (c) identification of an earlier therapeutic intervention, and (d) standardization of research protocols.3
The criteria for the new terms provide specific physiologic variables that can be used to categorize a patient as having bacteremia, systemic inflammatory response syndrome (SIRS), sepsis, severe sepsis, septic shock, or multiple-organ dysfunction syndrome (MODS), suggesting an important continuum of progressive physiologic decline (Table 97–1). The classification of sepsis was modified to include severe sepsis, septic shock, and refractory septic shock (Fig. 97–1).4 Severe sepsis refers to patients with an acute organ dysfunction, such as acute renal failure or respiratory failure. Septic shock refers to sepsis patients with arterial hypotension that is refractory to adequate fluid resuscitation, thus requiring vasopressor administration. It is important to note that progression from sepsis to MODS can occur in the absence of an intervening period of septic shock. Finally, refractory septic shock exists if dopamine IV infusion greater than 15 mcg/kg/min or norepinephrine greater than 0.25 mcg/kg/min is required to maintain a mean blood pressure greater than 60 mm Hg.
TABLE 97-1 Definitions Related to Sepsis
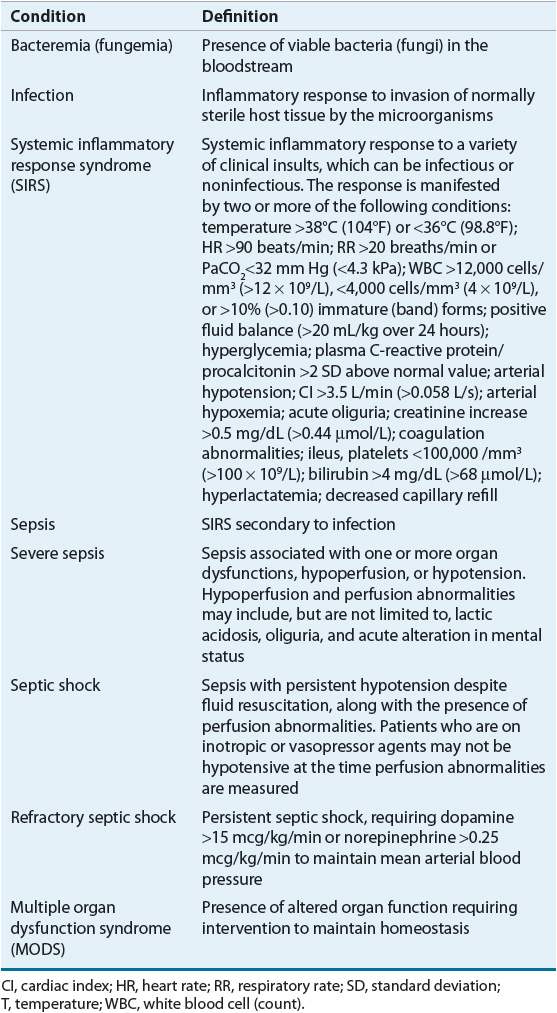
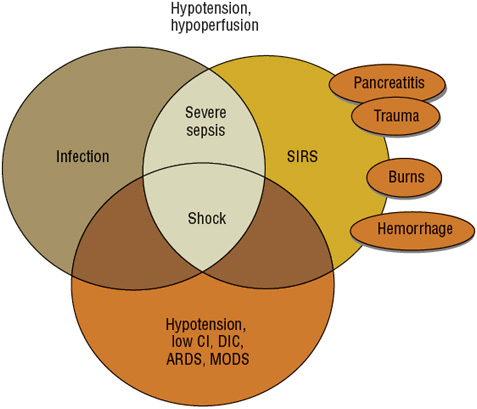
FIGURE 97-1 Relationship of infection, systemic inflammatory response syndrome (SIRS), sepsis, severe sepsis, and septic shock. (ARDS, acute respiratory distress syndrome; CI, cardiac index; DIC, disseminated intravascular coagulation; MODS, multiple-organ dysfunction syndrome.)
INFECTION SITES AND PATHOGENS
Predisposing factors of septic shock include age, male gender, nonwhite ethnic origin in North Americans, comorbid diseases, malignancy, immunodeficiency or immunocompromised state, chronic organ failure, alcohol dependence, genetic factors, and seasonal variation.1,5–8
The leading primary sites of microbiologically documented infections that lead to sepsis are the respiratory tract (39% to 50%), intraabdominal space (8% to 16%), and urinary tract (5% to 37%).2,9 Although almost any microorganism can be associated with sepsis and septic shock, the most common etiologic pathogens are gram-positive bacteria (52% of patients), followed by gram-negative bacteria (37.6%), polymicrobial infections (4.7%), anaerobes (1.0%), and fungi (4.6%).1,10
Gram-Positive Bacterial Sepsis
![]() Since 1987, gram-positive organisms have been the predominant pathogens in sepsis and septic shock, accounting for 52.1% of all cases.1 They are commonly caused by Staphylococcus aureus, Streptococcus pneumoniae, coagulase-negative staphylococci, and Enterococcus species.9–12
Since 1987, gram-positive organisms have been the predominant pathogens in sepsis and septic shock, accounting for 52.1% of all cases.1 They are commonly caused by Staphylococcus aureus, Streptococcus pneumoniae, coagulase-negative staphylococci, and Enterococcus species.9–12
S. aureus bacteremia is associated with an overall mortality rate ranging between 10% and 30%.13 Factors related to a higher mortality include older age, shock, preexisting renal failure, and the presence of a rapidly fatal underlying disease. Staphylococcus epidermidis is most often related to infected intravascular devices, such artificial heart valves and stents, and the use of IV and intraarterial catheters. Enterococci are isolated most commonly in blood cultures following a prolonged hospitalization and treatment with broad-spectrum cephalosporins.
Gram-Negative Bacterial Sepsis
While the overall percentage of gram-negative sepsis has decreased, the number of cases remains substantial.1,10 Escherichia coli (8% to 30%), Klebsiella species (8% to 23%), and Pseudomonas aeruginosa (7% to 18%) are the most commonly isolated gram-negative microorganisms in sepsis.9–12,14 Other common gram-negative pathogens include Serratia species, Enterobacter species, and Proteus species. P. aeruginosa and Acinetobacter species are more likely to be associated with prior antibiotic exposure.14
A greater proportion of patients with gram-negative bacteremia develop clinical sepsis, and also more likely to produce septic shock in comparison to gram-positive organisms, 50% versus 25%, respectively.9,10,12 Specifically, P. aeruginosa sepsis has been associated in a higher mortality rate.10,14 The major factor associated with the outcome of gram-negative sepsis appears to be the severity of any underlying conditions. Patients with rapidly fatal conditions, such as acute leukemia, aplastic anemia, and burn injury to >70% of the body’s surface, have a significantly worse prognosis than those patients with nonfatal underlying conditions such as diabetes mellitus and chronic renal insufficiency.1,2
Anaerobic and Miscellaneous Bacterial Sepsis
Anaerobic bacteria such as Bacteroides fragilis and Clostridium species are usually considered low-risk organisms for the development of sepsis. If present, anaerobes are often found together with other pathogenic bacteria that are commonly found in sepsis. Polymicrobial infections accounted for 5% to 39% of sepsis.1,10–12,14 Mortality rates associated with polymicrobial infections are similar to sepsis caused by a single organism. Although some clinicians believe the particular combination of organisms present in polymicrobial sepsis can provide clues to the source of infection, no clear source for the infection can be identified in up to 25% of cases.
Fungal Sepsis
![]() Candidemia is among the most common fungal etiologic agents of bloodstream infections. Although Candida albicans was the most commonly isolated fungus from blood cultures (45.6%), collectively, non-albicans Candida species were more frequently isolated (54.4%).10–12,15,16 Non-albicans Candida species include C. glabrata (26%), C. parapsilosis (15.7%), C. tropicalis (8.1%), and C. krusei (2.5%). Other fungi identified as causes of sepsis are Cryptococcus, Coccidioides, Fusarium, and Aspergillus.11 Risk factors for fungal infection include abdominal surgery, poorly controlled diabetes mellitus, prolonged granulocytopenia, broad-spectrum antibiotic treatment, corticosteroid treatment, prolonged hospitalization, central venous catheter, total parenteral nutrition, hematologic malignancy, and chronic indwelling bladder (Foley) catheter. Recent exposure to azoles is an important risk factor for infection with fluconazole-resistant Candida spp.17 Furthermore, a prospective nationwide surveillance study of candidemia suggested a close correlation between antibacterial drug exposure and bloodstream infection with C. glabrata and fluconazole-resistant Candida isolates.17
Candidemia is among the most common fungal etiologic agents of bloodstream infections. Although Candida albicans was the most commonly isolated fungus from blood cultures (45.6%), collectively, non-albicans Candida species were more frequently isolated (54.4%).10–12,15,16 Non-albicans Candida species include C. glabrata (26%), C. parapsilosis (15.7%), C. tropicalis (8.1%), and C. krusei (2.5%). Other fungi identified as causes of sepsis are Cryptococcus, Coccidioides, Fusarium, and Aspergillus.11 Risk factors for fungal infection include abdominal surgery, poorly controlled diabetes mellitus, prolonged granulocytopenia, broad-spectrum antibiotic treatment, corticosteroid treatment, prolonged hospitalization, central venous catheter, total parenteral nutrition, hematologic malignancy, and chronic indwelling bladder (Foley) catheter. Recent exposure to azoles is an important risk factor for infection with fluconazole-resistant Candida spp.17 Furthermore, a prospective nationwide surveillance study of candidemia suggested a close correlation between antibacterial drug exposure and bloodstream infection with C. glabrata and fluconazole-resistant Candida isolates.17
In a prospective analysis of the Antifungal Therapy Alliance database, the overall crude 12-week mortality rate for sepsis due to candidemia was 35.2%.15 A higher in-hospital mortality was reported (61%) among healthcare-associated candidemia.16 The highest mortality rate of 52.9% was observed in patients with C. krusei candidemia; C. parapsilosis candidemia was associated with the lowest 12-week mortality rate (23.7%). Hematologic diseases, neutropenia, and a higher number of positive blood cultures were associated with poor outcome irrespective of the patient’s gender, age, or days of antifungal drug treatment.
PATHOPHYSIOLOGY
Sepsis is the result of complex interactions among the invading pathogen, the host immune system, and the inflammatory responses. The inflammatory response leads to damage to host tissue, and the antiinflammatory response causes leukocytes to activate. Once the balance to control the local inflammatory process to eradicate the invading pathogens is lost, systemic inflammatory response occurs, converting the infection to sepsis, severe sepsis, or septic shock.
Cellular Components for Initiating the Inflammatory Process
The pathophysiologic focus of gram-negative sepsis has been on the lipopolysaccharide component of the gram-negative bacterial cell wall. Commonly referred to as endotoxin, this substance is unique to the outer membrane of the gram-negative cell wall and is generally released with bacterial lysis. Lipid A, the innermost region of the lipopolysaccharide, is highly immunoreactive and is considered responsible for most of the toxic effects. Although lipid A can affect tissues directly, its predominant effect is to activate macrophages and trigger inflammatory cascades critical in the progression to sepsis and septic shock.18 Endotoxin forms a complex with an endogenous protein called a lipopolysaccharide-binding protein, which then engages the CD14 receptor on the surface of a macrophage. Subsequently, cytokine mediators are activated and released by the macrophages.
In gram-positive sepsis, the exotoxin peptidoglycan on the cell wall surface appears to exhibit proinflammatory activity. Although it competes with lipid A for similar binding sites on CD14, the potency of peptidoglycan is less than that of endotoxin.18 However, an important feature of gram-positive bacteria such as S. aureus and S. pyogenes is the production of potent exotoxins, some of which have been associated with septic shock.
Pro- and Antiinflammatory Mediators
Sepsis involves activation of inflammatory pathways, and a complex interaction between proinflammatory and antiinflammatory mediators plays a major role in the pathogenesis of sepsis. The key proinflammatory mediators are tumor necrosis factor-α (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6), which are released by activated macrophages.18–20 Other mediators that may be important for the pathogenesis of sepsis are interleukin-8 (IL-8), platelet-activating factor (PAF), leukotrienes, and thromboxane A2.
The TNF-α levels in plasma can be increased in patients with a variety of diseases and in many healthy people. However, there is a correlation of plasma TNF-α levels with the severity of sepsis. It is highly elevated early in the inflammatory response in most patients with sepsis.19–21 The TNF-α release leads to activation of other cytokines (IL-1 and IL-6) associated with cellular damage. In addition, TNF-α stimulates the release of cyclooxygenase-derived arachidonic acid metabolites (thromboxane A2 and prostaglandins) that contribute to vascular endothelial damage. Higher levels of IL-6 and IL-8 have been reported in patients with septic shock than those with SIRS.8
The significant antiinflammatory mediators include interleukin-1 receptor antagonist (IL-1RA), interleukin-4 (IL-4), and interleukin-10 (IL-10).18,19 These antiinflammatory cytokines inhibit the production of the proinflammatory cytokines and down regulate some inflammatory cells. Levels of IL-10 and IL-1RA are higher in septic shock than in sepsis, and higher levels are found among nonsurviving patients than in survivors.19,20
The activation and secretion of pro- and antiinflammatory mediators in septic shock occur as a simultaneous immune response as early as the first 24 hours of diagnosis.14 As Fig. 97–2 illustrates, the balance between pro- and antiinflammatory mechanisms determines the degree of inflammation, ranging from local antibacterial activity to systemic tissue toxicity, organ failure, or death.19–21
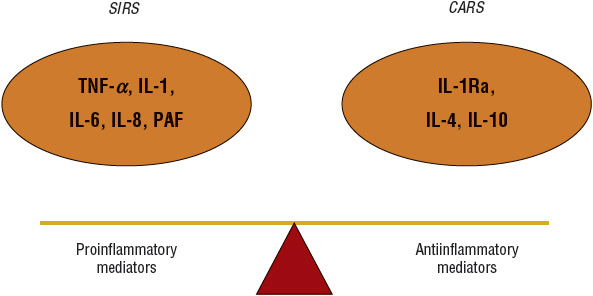
FIGURE 97-2 The balance between pro- and antiinflammatory mediators. (CARS, compensatory antiinflammatory response syndrome; IL, interleukin; IL-1RA, interleukin-1 receptor antagonist; SIRS, systemic inflammatory response syndrome; TNF, tumor necrosis factor.)
Cascade of Sepsis
![]() The cascade leading to development of sepsis is complex and multifactorial, involving various mediators and cell lines. Endothelial cells produce a variety of cytokines that mediate a primary mechanism of injury in sepsis. When injured, endothelial cells allow circulating cells such as granulocytes and plasma constituents to enter inflamed tissues, which can result in organ damage.
The cascade leading to development of sepsis is complex and multifactorial, involving various mediators and cell lines. Endothelial cells produce a variety of cytokines that mediate a primary mechanism of injury in sepsis. When injured, endothelial cells allow circulating cells such as granulocytes and plasma constituents to enter inflamed tissues, which can result in organ damage.
The microcirculation is affected by sepsis-induced inflammation. The arterioles become less responsive to either vasoconstrictors or vasodilators. The capillaries are less perfused even at the early phases of septic shock, and there is neutrophil infiltration and protein leakage into the venules.22
The inflammatory process in sepsis is also directly linked to the coagulation system. Proinflammatory mechanisms that promote sepsis are also procoagulant and antifibrinolytic, whereas fibrinolytic mechanisms can be antiinflammatory.23 A key endogenous substance involved in inflammation of sepsis is activated protein C, which enhances fibrinolysis and inhibits inflammation. Levels of protein C are reduced in patients with sepsis.23
COMPLICATIONS
Septic shock is the most ominous complication associated with sepsis, and it may lead to several complications including disseminated intravascular coagulation (DIC), acute respiratory distress syndrome (ARDS), and multiple organ failure. The organs that failed most frequently are lungs (18%) and kidneys (15%).1 The less frequent are cardiovascular failure (7%), hematologic failure (6%), metabolic failure (4%), and neurologic failure (2%).1 Mortality occurs in approximately half of the patients with septic shock.
Disseminated Intravascular Coagulation
DIC is the inappropriate activation of the clotting cascade that causes formation of microthrombi, resulting in consumption of coagulation factors, organ dysfunction, and bleeding. Sepsis remains the most common cause of DIC, and the incidence of DIC increases as the severity of sepsis increases. In sepsis alone, the incidence was 16% in comparison to 38% in septic shock.24,25 DIC occurs in up to 50% of patients with gram-negative sepsis, but it is also common in patients with gram-positive sepsis.
DIC begins with the activation and production of the proinflammatory cytokines, such as TNF, IL-1, and IL-6, which appear to be the principal mediators, along with endotoxin. The combination of excessive fibrin formation, inhibited fibrin removal from a depressed fibrinolytic system, and endothelial injury result in microvascular thrombosis and DIC.
Complications of DIC vary and depend on the target organ affected and the severity of the coagulopathy. DIC can produce acute renal failure, hemorrhagic necrosis of the GI mucosa, liver failure, acute pancreatitis, ARDS, and pulmonary failure. Furthermore, as the procoagulant state appears to be the key in the pathogenesis of MODS, coagulation dysfunction and MODS often coexist in sepsis.
Acute Respiratory Distress Syndrome
Pulmonary dysfunction, the most common organ dysfunction in sepsis, usually precedes other organs, and it can even initiate the development of SIRS with resultant MODS. Activated neutrophils and platelets adhere to the pulmonary capillary endothelium, initiating multiple inflammatory cascades with a release of a variety of toxic substances. There is diffuse pulmonary endothelial cell injury, increased capillary permeability, and alveolar epithelial cell injury. Consequently, interstitial pulmonary edema occurs that gradually progresses to alveolar flooding and collapse. The end result is loss of functional alveolar volume, impaired pulmonary compliance, and profound hypoxemia.
Coagulation is locally upregulated in the injured lung, whereas fibrinolytic activity is depressed. These abnormalities occur concurrently and favor alveolar fibrin deposition. Anticoagulant interventions that block the extrinsic coagulation pathway can protect against the development of pulmonary fibrin deposition as well as lung dysfunction and acute inflammation.25 Overall, fibrin deposition in the injured lung and abnormalities of coagulation and fibrinolysis are integral to the pathogenesis of ARDS.
Hemodynamic Effects
The hallmark of the hemodynamic effect of sepsis is the hyperdynamic state characterized by high cardiac output and an abnormally low systemic vascular resistance (SVR).22,26 TNF-α and endotoxin directly depress cardiovascular function. Endotoxin depresses left ventricular (LV) function independent of changes in LV volume or vascular resistance. Myocardial dysfunction is common in severe sepsis and septic shock, affecting 64% of patients, and involves LV in more than half of the patients.26
Persistent hypotension raises concern for the balance of oxygen delivery (DO2) to the tissues and oxygen consumption (VO2) by the tissues. Sepsis results in a distributive shock characterized by inappropriately increased blood flow to particular tissues at the expense of other tissues, which is independent of specific tissue oxygen needs. This perfusion defect is accentuated by an increased precapillary atrioventricular shunt. If perfusion decreases, oxygen extraction increases, and the arteriovenous oxygen gradient widens. Cellular DO2 is decreased, but VO2 remains unaffected. When increased oxygen demand occurs without increased blood flow, the increased VO2 is compensated by increased oxygen extraction. If perfusion decreases sufficiently in the face of high metabolic demands, then the reserve DO2 can be exceeded, and tissue ischemia results. Significant tissue ischemia leads to organ dysfunction and failure. Therefore, systemic DO2 relative to VO2 should be optimized by increasing oxygen delivery or decreasing oxygen consumption in a hypermetabolic patient.
Acute Renal Failure
Early acute kidney injury occurs in 42% to 64% of adult patients with sepsis and septic shock.27,28 Without normal urine output, fluid overload in extravascular space including the lungs develops, leading to impairment of pulmonary gas exchange and severe hypoxemia. Consequently, compromised oxygen delivery exacerbates peripheral ischemia and organ damage. Adequate renal perfusion and a trial of loop diuretics should be initiated promptly in oliguric or anuric patients with MODS along with dialysis to facilitate volume and electrolytes.
CLINICAL PRESENTATION
Table 97–2 lists some of the common clinical features of sepsis, although several of these findings are not limited to infectious processes. The initial clinical presentation can be referred to as signs and symptoms of early sepsis, defined as the first 6 hours. They are typically fever, chills, and change in mental status. Hypothermia can occur with a systemic infection, and this is often associated with a poor prognosis.29 In patients with sepsis caused by gram-negative bacilli, hyperventilation can occur even before fever and chills, and it can lead to respiratory alkalosis as the earliest metabolic change.
TABLE 97-2 Signs and Symptoms Associated
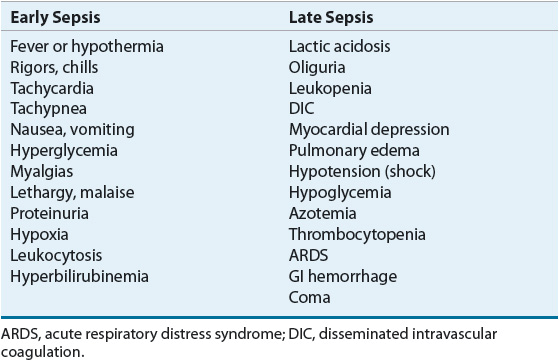
Progression of uncontrolled sepsis leads to clinical evidence of organ system dysfunction as represented by the signs and symptoms attributed to late sepsis. With the exception of rapidly progressing cases as in meningococcemia, P. aeruginosa, or Aeromonas infection, the onset of shock is slow and usually follows a period of several hours of hemodynamic instability. Oliguria often follows hypotension. Increased glycolysis with impaired clearance of the resulting lactate by the liver and kidneys and tissue hypoxia because of hypoperfusion result in elevated lactate levels, contributing to metabolic acidosis. Altered glucose metabolism, including impaired gluconeogenesis and excessive insulin release, is evidenced by either hyperglycemia or hypoglycemia.
PROGNOSIS
![]() As the patient progresses from SIRS to sepsis to severe sepsis to septic shock, mortality increases in a stepwise fashion. Mortality rates are higher for patients with advanced age, preexisting disease, including chronic obstructive pulmonary disease, neoplasm, and human immunodeficiency virus disease, intensive care unit (ICU) care, more failed organs, positive blood cultures, and Pseudomonas species infection.10,13 In one analysis of cases, mortality increased with age from 10% in children to 38.4% in those 85 years or older.1 ICU admission was required in 51.1% of patients with severe sepsis; of those patients, mortality was reported in 34.1%.1 Mortality from severe sepsis and MODS is most closely related to the number of dysfunctioning organs. As the number of failing organs increased from two to five, mortality increased from 54% to 100% (Fig. 97–3).30 Duration of organ dysfunction can also affect the overall mortality rate.
As the patient progresses from SIRS to sepsis to severe sepsis to septic shock, mortality increases in a stepwise fashion. Mortality rates are higher for patients with advanced age, preexisting disease, including chronic obstructive pulmonary disease, neoplasm, and human immunodeficiency virus disease, intensive care unit (ICU) care, more failed organs, positive blood cultures, and Pseudomonas species infection.10,13 In one analysis of cases, mortality increased with age from 10% in children to 38.4% in those 85 years or older.1 ICU admission was required in 51.1% of patients with severe sepsis; of those patients, mortality was reported in 34.1%.1 Mortality from severe sepsis and MODS is most closely related to the number of dysfunctioning organs. As the number of failing organs increased from two to five, mortality increased from 54% to 100% (Fig. 97–3).30 Duration of organ dysfunction can also affect the overall mortality rate.
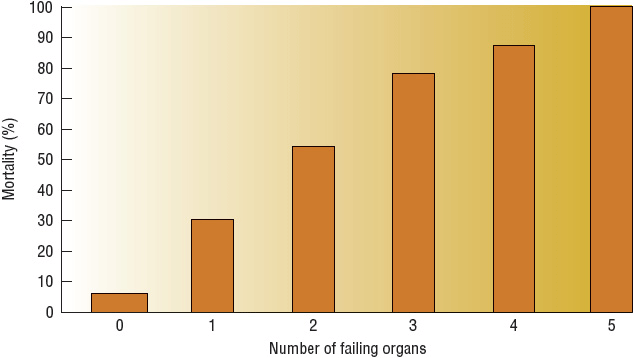
FIGURE 97-3 Mortality related to the number of failing organs.
An elevated lactate concentration of >4 mmol/L in the presence of SIRS significantly increases ICU admission rates, and persistent elevations in lactate for more than 24 hours are associated with a mortality rate as high as 89%.31 Inversely, patients with higher lactate clearance after 6 hours of emergency department intervention have improved outcome compared with those with lower lactate clearance. There was an ~11% decrease in the likelihood of mortality for each 10% increase in lactate clearance.31
Diagnosis and Identification of Pathogen
The presence of clinical features suggesting sepsis should prompt further evaluation of the patient. In addition to obtaining a careful history of any underlying conditions and recent travel, injury, animal exposure, infection, or use of antibiotics, a complete physical examination should be performed to determine the source of the infection.
A collection of specimens should be sent for culture prior to initiating any antimicrobial therapy. In critically ill septic patients, two or three sets of blood cultures should be collected without temporal separation between the sets.32,33 With suspected catheter-related infection, a pair of blood cultures should be drawn through every lumen of each vascular access device.33 In severe community-acquired pneumonia, blood cultures and respiratory secretions must be obtained. Urinary antigen detection of Legionella sero group 1 is recommended during outbreaks. To document a soft tissue infection, a Gram stain and bacterial culture of any obvious wound exudates should be performed. A needle aspiration of a closed infection such as cellulitis or abscess may be needed for stain and bacterial culture. In abdominal infections, fluid collections identified by imaging studies should be aspirated for Gram stains and aerobic and anaerobic cultures. Recent development of accurate and rapid identification tests has demonstrated positive impact on prescribing appropriate therapy in bloodstream infections such as methicillin-resistant Staphylococcus aureus (MRSA) and Candida spp.34,35
A lumbar puncture is indicated in the case of mental alteration, severe headache, or a seizure, assuming that there are no focal cranial lesions identified by computed tomography scan. Further tests may be indicated to assess any systemic organ dysfunction caused by severe sepsis. The laboratory tests should include hemoglobin, white blood cell (WBC) count with differential, platelet count, complete chemistry profile, coagulation parameters, serum lactate, procalcitonin (PCT), and arterial blood gases.
TREATMENT
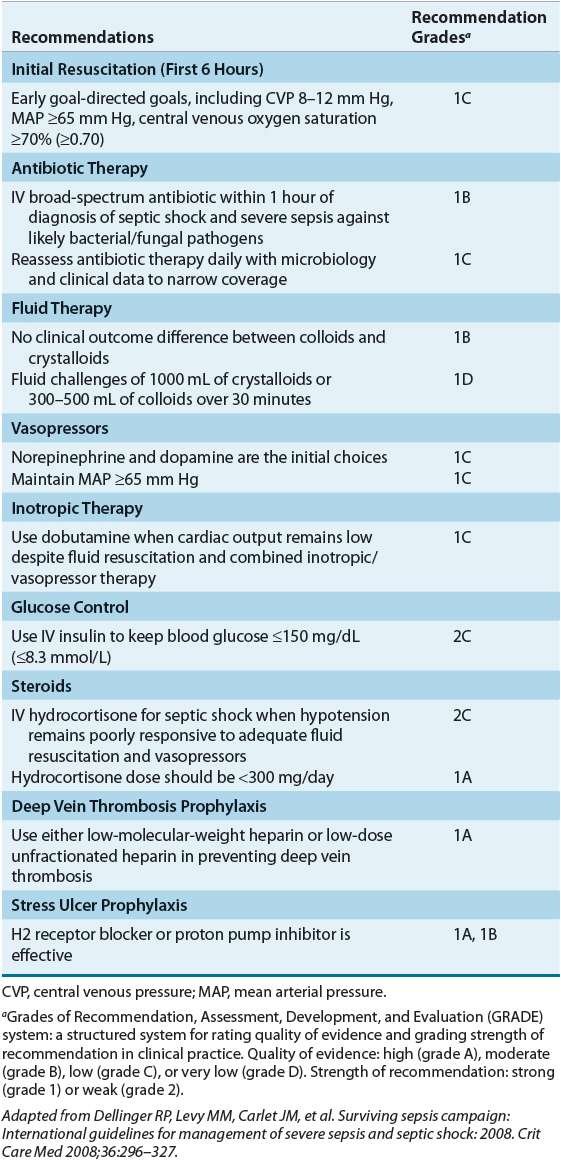
In 2008, a “surviving sepsis” campaign guideline for management of severe sepsis and septic shock was published as an international effort to increase awareness and improve outcome in severe sepsis.32 The primary goals of therapy for patients with sepsis are (a) timely diagnosis and identification of the pathogen, (b) rapid elimination of the source of infection medically and/or surgically, (c) early initiation of aggressive antimicrobial therapy, (d) interruption of pathogenic sequence leading to septic shock, and (e) avoidance of organ failure. Supportive care such as stress ulcer prophylaxis and nutritional support is important to prevent complications during the stay in the ICU. Table 97–3 describes the summary of the surviving sepsis campaign treatment recommendations.
TABLE 97-3 Evidence-based Treatment Recommendations for Sepsis and Septic Shock
Elimination of the Source of Infection
After the source of infection is identified, prompt efforts to eradicate that source should be made.32 With an infected intravascular catheter, the catheter should be removed and cultured. Urinary tract catheters should be removed if association with sepsis is suspected. Suspicion of soft tissue (cellulitis or wound infection) or bone involvement should lead to aggressive debridement of the affected area. Evidence of an abscess or sepsis associated with any intraabdominal pathology should prompt surgical intervention.
Antimicrobial Therapy
![]() The most recent guidelines from the Surviving Sepsis Campaign recommended starting IV antibiotic therapy as early as possible because early administration of broad-spectrum antibiotics is critical in decreasing the risk of mortality.32 Early administration (within 1 hour vs. 6 hours of diagnosis) of broad-spectrum antibiotics was independently associated with lower hospital mortality in patients with severe sepsis.36,37
The most recent guidelines from the Surviving Sepsis Campaign recommended starting IV antibiotic therapy as early as possible because early administration of broad-spectrum antibiotics is critical in decreasing the risk of mortality.32 Early administration (within 1 hour vs. 6 hours of diagnosis) of broad-spectrum antibiotics was independently associated with lower hospital mortality in patients with severe sepsis.36,37
Delays in the initiation of effective antimicrobial therapy after the onset of hypotension were significant predictors of mortality.38 In a large retrospective study, inappropriate initial antimicrobial therapy occurred in about 20% of patients with septic shock, and was associated with a fivefold reduction in survival in comparison to those who received appropriate therapy (52.0% vs. 10.3%, respectively).12 Therefore, early administration of appropriate antimicrobial therapy is critical in the treatment of severe sepsis and septic shock.
Pharmacokinetics of Antimicrobial Agents in Critically Ill Patients
Pathophysiologic changes have been reported in sepsis that can affect drug distribution, and adjusted dosing regimens are required in critically ill patients with sepsis.39 Initially high creatinine clearance can be seen in patients with normal serum creatinine because of increased renal preload. Volume of distribution can increase because of fluid accumulation from leaky capillaries and/or altered protein binding. Consequently, some antimicrobial agents, especially for hydrophilic antimicrobials including aminoglycosides, β-lactams, carbapenems, and vancomycin can result in lower peak serum concentrations with usual doses.40 However, as sepsis progresses, organ perfusion decreases because of significant myocardial depression and leads to multiple organ dysfunction. Consequently, clearance of antimicrobial agents is decreased, prolonging the elimination half-life and accumulation of metabolites. Hence, in addition to selecting the most appropriate antimicrobial agents, a clinician must ensure effective antibiotic usage, such as proper dosing, interval of administration, optimal duration of treatment, monitoring of drug levels when appropriate, and avoidance of unwanted drug interactions. The lack of adherence to these requirements can lead to suboptimal or excessive tissue concentrations that can promote antibiotic resistance, toxicity, and inadequate efficacy despite appropriate antibiotic selection.
Selection of Antimicrobial Agents
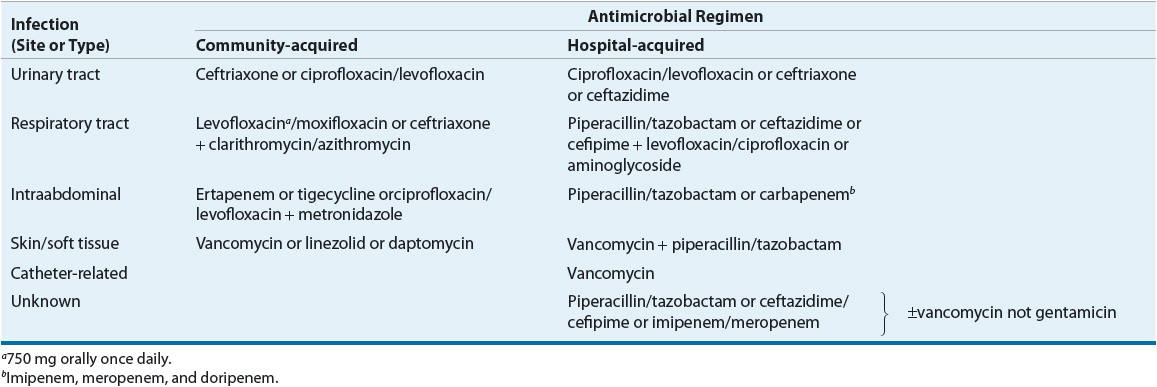
The selection of an empiric regimen should be based on the suspected site of infection, the most likely pathogens, acquisition of the organism from the community or hospital, the patient’s immune status, and the antibiotic susceptibility and resistance profile for the institution. All patients should be treated initially with parenteral antibiotics for optimal drug concentrations within the first hour of recognition of severe sepsis after appropriate cultures have been taken.32 Empiric therapy for an immunocompromised patient should be broad enough to cover likely pathogens and penetrate adequately into the presumed infection site. Once the pathogen and its susceptibility pattern are known, the antimicrobial regimen should be modified accordingly.
Table 97–4 lists antimicrobial regimens that can be used empirically based on the possible source of infection. In the nonneutropenic patient with a urinary tract infection, ceftriaxone and fluoroquinolones are generally recommended. When there is increased risk of P. aeruginosa in sepsis or hospital-acquired infections, an antipseudomonal antibiotic, such as ceftazidime, is recommended.41
TABLE 97-4 Empiric Antimicrobial Regimens in Sepsis