Rectal Prolapse: the Open Abdominal Approach
Philip H. Gordon
Introduction
Rectal prolapse is the protrusion of the full thickness of the rectal wall through the anal sphincter. More than 100 techniques for its repair have been described indicating that none are entirely satisfactory. This chapter presents the current information about the more popularly used techniques in the repair of complete rectal procidentia. Each procedure has advantages and disadvantages. The particular method of repair used by a surgeon depends on his previous training and exposure to a certain technique, which is modified by his own personal experience.
There are two theories regarding the etiology of rectal procidentia—a sliding hernia through a defect in the pelvic fascia and an intussusception. Cineradiographic studies support the latter belief, that is, that rectal prolapse is the result of intussusception, or infolding of the rectum, usually beginning 6 to 8 cm from the anal verge. However, the factors that cause intussusception are still unknown. Excessive straining at defecation appears to be an important factor in the pathogenesis of rectal prolapse. The anatomic abnormalities commonly found in patients with this condition are the results of prolonged rectal prolapse, and not the cause. Pathological anatomy of patients with rectal procidentia include a deep cul-de-sac, a redundant sigmoid colon, diastasis of the levator ani muscle, a patulous anal sphincter, and loss of the posterior sacral curve of the rectum and its sacral attachments.
It was once thought that weakness of the pelvic floor muscle was the main cause of rectal prolapse. However, electromyography shows overactive pelvic floor muscle in some patients and weakness in others. Fecal incontinence in rectal prolapse is the result of neuropathy of the pudendal nerve due to prolonged stretching, and not from mechanical stretching of the external sphincter by the prolapse, as previously believed.
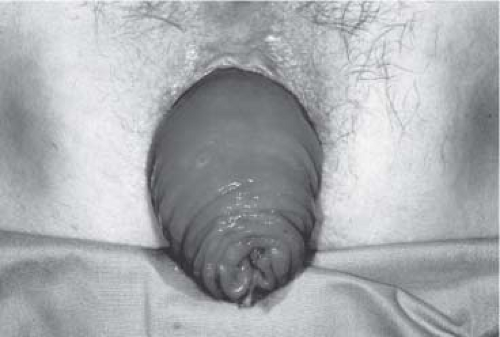
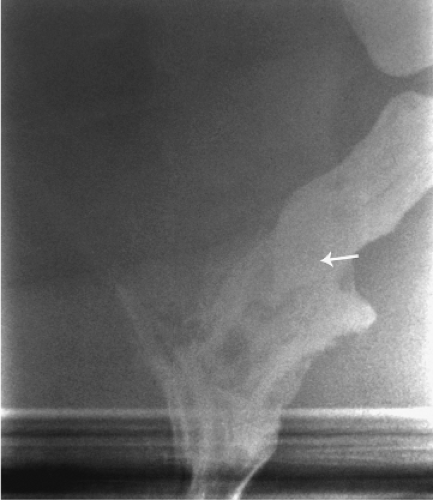
These patients usually complain of mucous leakage into their underwear and around the anus that causes variable degrees of perineal skin irritation. Dull perineal pain or pressure and a feeling of incomplete evacuation are common. Difficult defecation is another common feature. By the time the patients seek medical consultation, about 50% of them already have fecal incontinence. All patients in whom rectal prolapse is suspected should be examined while straining on the toilet. The diagnosis of rectal prolapse is easy if the protrusion comes through the anus (Fig. 1). Hidden prolapse, in which the intussusception is above the anal canal, can be diagnosed only by defecating proctography (Fig. 2). When the prolapse comes out but not through the anus, it can be easily misdiagnosed as mucosal prolapse or prolapsed hemorrhoids, but the latter are characterized by radial folds rather than circular folds.
Bachoo et al. tried to determine the best choice of operation for rectal procidentia. All randomized or quasi-randomized trials of operation in the management of rectal prolapse were sought. The three primary outcome measures were number of patients with recurrent rectal prolapse, and residual mucosal prolapse or fecal incontinence. Eight trials were included with a total of 264 participants. There were no detectable differences in recurrent prolapses between abdominal and perineal approaches, although there was a suggestion that fecal incontinence was less common after abdominal procedures. There were no detectable differences between the methods used for fixation during rectopexy. Division rather than preservation of the lateral ligaments was associated with less recurrent prolapse but more postoperative constipation, although these findings were found in small numbers.
Ideally, the repair should be chosen not only to correct the prolapse, but also to restore defecatory function and improve fecal incontinence. By and large, the major consideration is the patient’s general state of health. At present, for the good-risk patient, an abdominal approach is still favored. The recommended procedure would be a pelvic fixation, in which the rectum is not completely surrounded anteriorly by a synthetic material. However, a resection might be recommended instead of one of the fixation procedures for an individual who has symptomatic diverticular disease or who suffers from severe constipation in association with a very redundant colon. Certainly one would hesitate to insert a foreign body in the pelvis in association with the resection.
For poor-risk patients, a perineal approach would be more appropriate, and the Delorme operation or possibly a perineal rectosigmoidectomy (Altemeier procedure) is recommended. For intermediate-risk patients, a perineal rectosigmoidectomy should be considered. For patients confined to a nursing home, amputation of a massive prolapse definitely would facilitate their care.
All patients with rectal prolapse should at least be examined with a flexible sigmoidoscope to rule out the associated abnormalities, especially polyps, cancer, and inflammatory bowel disease. Occasionally, a solitary rectal ulcer is found. If indicated, a total colonoscopy should be performed. Defecating proctography is indicated, if hidden prolapse is suspected.
Anal manometry will not change the surgical approach and is not helpful to predict postoperative functional results. Electromyography and measurement of the pudendal nerve terminal motor latency are useful to confirm the nerve damage to the external anal sphincter and the pelvic floor muscles. Patients with damage to the pudendal nerves usually continue to have fecal incontinence after successful repair of the rectal prolapse.
Bowel Preparation
Despite ongoing controversy, the author continues to prepare patients with a mechanical bowel cleansing. A bowel devoid of stool is easier to handle especially if a resection is anticipated. At present, two sachets of oral Pico-Salax are used the day prior to operation. Antibiotics are also administered preoperatively and our current choice is Timentin 3.1-g i.v. within 1 hour of incision and two postoperative doses q8h.
Well’s Repair
The anterior mesh sling or Ripstein repair has long been the most popular repair for rectal prolapse in the United States. This technique has been plagued with complications and Ripstein himself abandoned the technique in favor of the posterior sling. The complication rate might be reduced by half, if the sling were placed posterior to the rectum leaving the anterior one-fourth to one-third of the circumference of the rectum free to expand, a principle expounded by Wells.
Ivalon sponge (polyvinyl alcohol), a popular material used in the United Kingdom, I understand is not approved by the U.S. Food and Drug Administration. I have successfully used this material for many years. Marlex (polypropylene) and Teflon (polycarbon fluoride) are the most common materials used. Other materials such as Gor-Tex (polytetrafluoroethylene), Prolene (polypropylene, similar to Marlex), and Vicryl (polyglycolic acid) have also been employed.
Technique
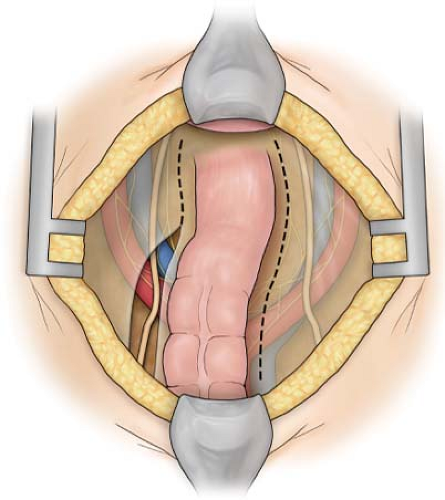
With the patient in the supine position, after the bladder has been catheterized, the skin is prepared with a suitable antiseptic. The abdomen is entered through an infraumbilical transverse incision. With the patient placed in a 20-degree Trendelenburg position, the abdominal viscera are packed away in the upper abdomen with a self-retraining retractor. Dissection is begun by incising the peritoneum on each side of the rectosigmoid, commencing about 5 cm above the pelvic brim (Fig. 3). Care is taken to protect the ureters. The
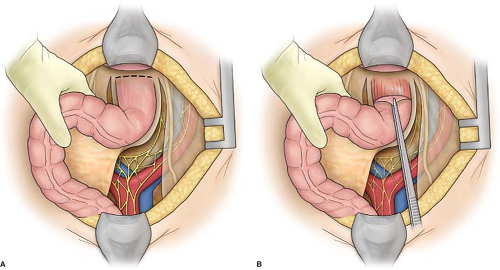
presacral space is entered at the level of the sacral promontory, and the rectum is gently mobilized from the sacral hollow (Fig. 4). At this point, great care must be taken to ensure that the mesorectum is anterior to the plane of dissection or the intestine may be devascularized and there may be annoying bleeding. At the same time, one should avoid the presacral nerves. Dissection in the presacral plane should be performed with cautery taking care not to injure the rectal wall. Any fibrous bands are cut with scissors or cautery, since tearing will result in troublesome bleeding. Sweeping the hand to each side of the rectum will make the lateral stalks more prominent. The rectum is fully mobilized to the level of the coccyx. Anterior mobilization is begun by continuing the lateral peritoneal incisions distally until they meet in the deepest portion of the cul-de-sac (Fig. 5). With the rectum grasped in the left hand, this plane can often be dissected with cautery. Extensive dissection in this plane is not necessary because a deep cul-de-sac is a characteristic anatomic feature, in most instances, of complete rectal procidentia. The seminal vesicles or vaginal vault is easily reached. The lateral stalks, or at least the upper portion of the lateral stalks on each side, are divided between clamps and ligated. If the rectum is not fully mobilized, redundant rectum remaining below the point of fixation may lead to recurrence of prolapse. Throughout the procedure, meticulous hemostasis is essential, since hematoma formation may predispose to infection.
presacral space is entered at the level of the sacral promontory, and the rectum is gently mobilized from the sacral hollow (Fig. 4). At this point, great care must be taken to ensure that the mesorectum is anterior to the plane of dissection or the intestine may be devascularized and there may be annoying bleeding. At the same time, one should avoid the presacral nerves. Dissection in the presacral plane should be performed with cautery taking care not to injure the rectal wall. Any fibrous bands are cut with scissors or cautery, since tearing will result in troublesome bleeding. Sweeping the hand to each side of the rectum will make the lateral stalks more prominent. The rectum is fully mobilized to the level of the coccyx. Anterior mobilization is begun by continuing the lateral peritoneal incisions distally until they meet in the deepest portion of the cul-de-sac (Fig. 5). With the rectum grasped in the left hand, this plane can often be dissected with cautery. Extensive dissection in this plane is not necessary because a deep cul-de-sac is a characteristic anatomic feature, in most instances, of complete rectal procidentia. The seminal vesicles or vaginal vault is easily reached. The lateral stalks, or at least the upper portion of the lateral stalks on each side, are divided between clamps and ligated. If the rectum is not fully mobilized, redundant rectum remaining below the point of fixation may lead to recurrence of prolapse. Throughout the procedure, meticulous hemostasis is essential, since hematoma formation may predispose to infection.
 Fig. 4. A: Mobilization of rectum accomplished by cautery dissection, avoiding presacral plexus of veins. B: Sagittal view: dissection carried down toward tip of coccyx, preserving mesorectum. |
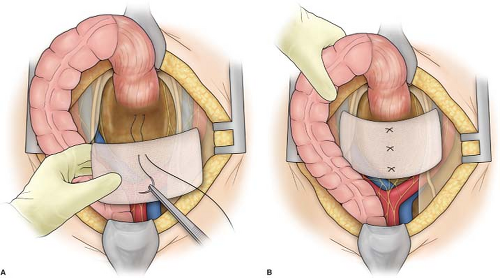
A rectangular sheet of previously sterilized Ivalon (polyvinyl alcohol) sponge, 3 mm thick and 22 by 10 cm, is then moistened in normal saline solution to make it pliable and easy to handle. Three sutures of a nonabsorbable monofilament material such as 00 polypropylene on a noncutting needle are then passed through the Ivalon sponge, the presacral fascia, and back through the Ivalon sponge (Fig. 6A). The distal suture should be placed as low as possible deep in the pelvis. The sponge is then “rail-roaded” into the pelvis, and the sutures are tied (Fig. 6B).
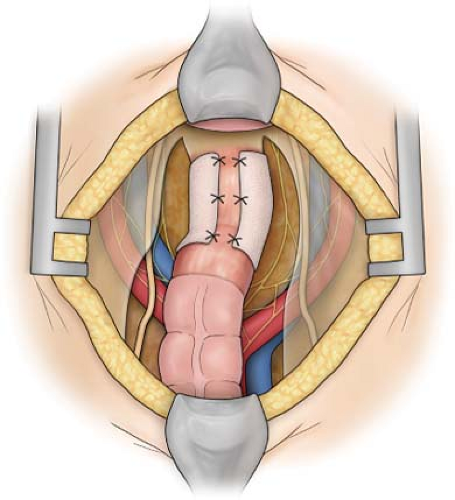 Fig. 7. Ivalon sheet applied to posterior three-fourths of circumference and sutured in place with three 000 polypropylene sutures on each side.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|