Prostate
Peter A. Humphrey
I. NORMAL ANATOMY. The normal weight of the prostate is 20 g for ages 20 to 50, and 30 g for ages 60 to 80. Anatomically, the prostate gland comprises three zones: central zone, transition zone (where benign prostatic hyperplasia [BPH] occurs), and peripheral zone (where most carcinomas originate). (e-Fig. 29.1).* Microscopically, the normal adult prostate is a branching duct-acinar glandular system embedded in a dense fibromuscular stroma (e-Fig. 29.2). The epithelium has two layers: a luminal or secretory cell layer and a basal cell layer. Central zone epithelium can normally have architectural patterns that include cribriform and Roman bridge-like structures (e-Fig. 29.3).
II. GROSS EXAMINATION, TISSUE SAMPLING, AND HISTOLOGIC SLIDE PREPARATION. The most common prostatic parenchymal tissue samples examined in surgical pathology laboratories in the United States are, in order, 18-gauge needle cores, transurethral resection of prostate (TURP) chips, radical prostatectomy specimens, and fine needle aspirates.
A. Needle cores. Needle core biopsy sample handling and processing begins in the room where the procedure is performed. The needle biopsy tissue should be immediately placed into a container with fixative, which is usually 10% neutral buffered formalin, although a few laboratories prefer Bouin’s solution, Hollande’s solution, or IBF fixative. Bouin and Hollande’s solutions are picric acidbased fixatives that provide superior nuclear detail, but these strong oxidizing agents can react violently with combustible materials and reducing agents. Fixation in formalin should be for at least 6 hours. The number of cores received per container is highly variable, from 1 to >20. If the urologist and treating physician desire site-specific diagnosis, the core(s) should be placed in separate sitedesignated containers. Inking of cores to indicate site, with placement of cores marked with different colors into the same container, should not be performed because fragmentation renders site assignment impossible. Gross examination of prostate needle core tissue is not diagnostic, but is important for correlation with amount of tissue seen in the histologic sections and so it is vital to record, for each container, the size and number of tissue cores or fragments. It is recommended that no more than two cores be submitted per cassette for processing and embedding; some laboratories submit one core per cassette. Prostate cores can be marked with ink, which facilitates identification during embedding and the ability to see the cores in the paraffin blocks. Regardless, the cores should be placed into a cassette after being put into a fine mesh envelope, wrapped in lens paper, sandwiched between sponge pads, or double-embedded in agar-paraffin wax. After processing, the cores should be embedded in the same plane, in the same direction, with even spacing. From each paraffin block, three hematoxylin and eosin (H&E)-stained slides should be prepared, with three to four serial sections on each slide. Some laboratories cut interval, unstained sections on coated slides in case special studies such as immunohistochemistry are needed. Clinical requests for frozen section diagnosis of prostate needle cores are rare and should be restricted to patients with clinical evidence of metastatic cancer who are to undergo immediate treatment (usually orchiectomy) for pain relief.
B. TURP chips. The amount of prostate tissue resected in TURP procedures is variable, ranging from 5 to >75 g of tissue, with a mean of about 25 g. The gross description should include the aggregate weight of the chips. Recognizable gross features such as yellow coloration and induration can be recorded, but it has not been proven that chip color, size, or induration is linked to cancer presence, so gross selection of specific chips is not required. Although gross TURP chip sampling procedures are not standardized, one initial approach is to submit 12 g of chips or 6 to 8 blocks of tissue (with 1 to 2 g per cassette). For specimens >12 g, the initial 12 g are submitted, with one cassette for every additional 5 g (Arch Pathol Lab Med. 2009;133:1568). If the patient is younger than 60 years, all tissue should be submitted; all chip tissue should also be submitted if microscopic examination of partially submitted chips reveals carcinoma in <5% of tissue, or if high-grade prostatic intraepithelial neoplasia (PIN) or atypical glands (atypical small acinar proliferation [ASAP]) is found in sections of partially submitted chips. One H&E-stained slide, with one or two sections, is typically generated from each paraffin block of TURP chips.
C. Open suprapubic or retropubic simple prostatectomy (enucleation) tissue. The prostatic tissue from simple prostatectomies may be submitted to the pathology laboratory as a single mass or as pieces. The prostatectomy tissue should be weighed and sectioned at 3- to 5-mm intervals. The gross description for each piece should include size in three dimensions, weight, firmness, and coloration. Hard nodules should be sampled, and a total of eight cassettes or one cassette of tissue for each 5 g of tissue submitted. Additional tissue should be submitted if carcinoma is histologically detected in initial sections of partially submitted tissue, although no rules or recommendations exist on how many additional sections are required. One H&E-stained slide should be made per block.
D. Radical prostatectomy. The entire prostate gland is excised in prostate cancer surgery using open retropubic or perineal approaches, or using laparoscopic (including robotic) approaches. The prostate gland is also resected in toto in radical cystoprostatectomy for bladder cancer.
1. Pelvic lymph nodes. Pelvic lymphadenectomy may be performed as a separate procedure, often laparoscopic, or during the radical prostatectomy operation. Sentinel lymph node sampling is not routinely performed.
Frozen section analysis of the sampled lymph nodes may be requested for patients at risk for nodal metastasis, on the basis of serum prostatespecific antigen (PSA) level, needle biopsy Gleason score, and clinical stage. All grossly recognizable lymph nodes should be examined by frozen section; cytologic touch imprints can be made at the same time. Frozen section diagnosis of metastatic carcinoma in lymph nodes is highly specific, but fairly insensitive. The low sensitivity rate of 58% to 73% is due to sampling error.
Gross sampling of tissue after frozen section should entail submission of all grossly identifiable lymph node tissue and wide sampling of associated adipose tissue. The gross description of pelvic lymph nodes should include number, location, and size. One H&E-stained slide is made per paraffin block. Special studies to detect occult lymph node metastases, such as immunohistochemistry for cytokeratins or PSA, or reverse transcription-polymerase chain reaction (RT-PCR) for PSA RNA, are currently experimental and not performed in routine practice.
2. Prostate gland and seminal vesicles. The prostate gland and seminal vesicles from radical prostatectomy procedures may be received fresh or in fixative. All three dimensions and specimen weight should be recorded. Frozen section requests on fresh specimens are uncommon, and are usually made to evaluate margin status; this procedure has a high false-negative rate and is not standard practice. Fresh specimens are also used for tissue-banking protocols; after inking the entire outside of the specimen, tissue can be harvested from
inside the gland while preserving the inked periphery. Alternatively, after inking, margin sampling, and seminal vesicle amputation (see below), the whole unfixed gland can be sectioned with a large sharp knife from apex to base at 4-mm intervals perpendicular to the prostatic urethra; areas suspicious for carcinoma, as judged by palpation or visual inspection, may be sampled by imprints, scrapes, core biopsy, or small wedge sections.
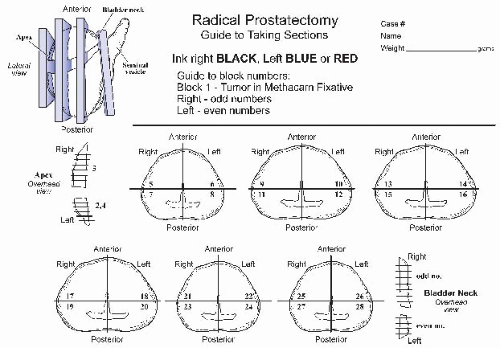
Fixation of the inked radical prostatectomy specimens (sectioned or unsectioned) is accomplished by at least overnight (or 24 to 48 hour) room temperature immersion in 10% neutral buffered formalin at 10 times the volume of the specimen. For gross examination of unsectioned glands after inking, the seminal vesicles are amputated (including the soft tissue and prostatic tissue at the base of the seminal vesicles) and submitted separately as right and left seminal vesicles. The prostate weight without the seminal vesicles is recorded, and distal apical (urethral) and bladder neck margins are taken if not already submitted separately by the surgeon. The distal apical margin is evaluated by amputating the distal 5 to 10 mm of the gland, dividing it into the right and left sides, and submitting radial sections (as for a cervical cone biopsy). The bladder neck margin can be assessed by a thin 2-mm shave margin or by conization; the latter is recommended. Ink on tumor cells is indicative of a positive margin for cone sections and the peripheral margin, whereas tumor anywhere in shave margin tissue indicates a positive margin. Vasa deferentia stumps may be sampled using en face sections, but this is not routine. The prostate gland is serially sectioned in a plane perpendicular to the urethra at 3- to 5-mm intervals using a long knife. The cut surfaces should be evaluated for gross evidence of BPH and carcinoma. Photographs or digital images can be used to document location and gross appearance of tissue in submitted cassettes. Diagrams (Fig. 29.1) or pictorial maps can also be used to indicate location of sections and any gross abnormalities.
Both complete and partial embedding methods are acceptable (Mod Pathol. 2011;24:6). Several protocols for partial submission have been published (Scand J Urol Nephrol Suppl. 2005;216:34; Mod Pathol. 2011;24:6). When there is grossly visible tumor, all lesions grossly suspicious for carcinoma should be submitted, along with distal apical and bladder neck margins and seminal vesicles. For cases with no grossly evident tumor, the posterior aspect of each transverse slice is submitted, as well as a mid-anterior block from each side, distal apical and bladder neck margins, and the seminal vesicles. Sections should be submitted as quarters or halves of the prostate, depending on gland size. Whole-amount sections are rarely made and do not provide additional morphologic information. If no or minimal tumor is seen in initial sections of a partially submitted gland, all remaining tissue should be embedded (including any frozen tissue sent to a tissue bank). If still no tumor is seen, basal cell and AMACR immunostains of atypical foci should be performed, and deeper sections should be obtained from block(s) from the region of the positive needle biopsy and areas of high-grade PIN. If cancer remains undetected, the tissue blocks should be flipped, and histologic sections prepared from the new tissue faces (Mod Pathol. 2011;24:6).
E. Cystoprostatectomy. The prostate gland in radical cystoprostatectomies performed for bladder cancer can be sampled by taking several sections of prostatic urethra and surrounding prostate tissue, any gross lesions, one block from the periphery of each side, and both seminal vesicles. The distal urethral shave margin is important in urothelial carcinoma cases.
III. DIAGNOSTIC FEATURES OF COMMON DISEASES OF THE PROSTATE
A. Inflammation and infection. Histopathologic identification of inflammatory cells in the prostate is common, but histologic identification of specific infectious agents is rare.
1. Asymptomatic inflammatory prostatitis, including acute neutrophilic inflammation and chronic lymphocytic, lymphoplasmacytic, or lymphohistiocytic inflammation, is common in all prostate tissue samples. Reporting this inflammation is optional; it may be useful to report if the inflammation is extensive or persistent in several needle core samples taken over time, because prostatic inflammation can raise the serum PSA. Inflammation can be associated with prostatic glandular atrophy, and reactive nuclear changes including prominent nucleoli. Inflammation is more commonly associated with benign epithelial conditions, especially atrophy and BPH, compared with high-grade PIN and carcinoma where usually only a small percentage of foci (around 10%) are inflamed.
2. Granulomatous prostatitis can clinically elevate the serum PSA and/or present as a palpable abnormality. The most common type is nonspecific granulomatous prostatitis, which is thought to be a response to prostatic secretions released into stroma by duct-acinar rupture. Microscopically, there is a lobulocentric noncaseating granulomatous inflammatory cell infiltrate with giant cells (e-Fig. 29.4). Variants include xanthogranulomatous prostatitis and prostatic “xanthoma.” Other types of granulomatous prostatitis include infectious and postbiopsy/postresection cases. Infectious granulomatous prostatitis is most often Bacille Calmette-Guérin (BCG)-related in patients treated for bladder urothelial carcinoma (e-Fig. 29.5). Fungal prostatitis is rare and usually seen in immunosuppressed patients. Postbiopsy/resection granulomas are most often identified in TURP chip tissue, and are characterized by a fibrinoid central zone surrounded by palisading histiocytes.
B. Atrophy of prostatic glands is the benign condition most likely to be misdiagnosed as prostatic carcinoma by light microscopy. It is a common, age-related process that could be related to inflammation, hormones, obstruction, or ischemia. It can also be treatment related, due to radiotherapy or hormonal therapy.
Histologically, atrophy is defined as cytoplasmic volume loss. It is not necessary to subtype atrophy, but it is important to recognize the existence of different histomorphologic patterns including simple atrophy (with or without cystic change) (e-Fig. 29.6), sclerotic atrophy, partial atrophy (e-Fig. 29.7), and postatrophic hyperplasia (or hyperplastic atrophy) (e-Figs. 29.8 and 29.9). Atrophy can be confused with carcinoma because it is usually a small gland lesion that can show a pseudoinfiltrative pattern of growth, stromal sclerosis, nuclear atypia, and closely packed acini (in postatrophic hyperplasia). Atrophy can also be noted in cystically dilated peripheral zone glands (e-Fig. 29.10) and in cystic change in BPH nodules. Another diagnostic pitfall is that atrophic glands can show a fragmented basal cell layer and even loss of basal cells in a few glands by immunohistochemical stains (such as 34betaE12 and p63) (Semin Diagn Pathol. 2005;22:88). In addition, the selective but not specific marker for neoplastic epithelial cells alpha-methylacyl coenzyme A racemase (AMACR) can be immunopositive in atrophy, particularly partial atrophy.
C. Metaplasia or change in cell type in benign prostatic epithelium can be squamous, transitional cell (urothelial), mucinous, and eosinophilic. These metaplasias are usually secondary to inflammation, therapy, or injury. They are not preneoplastic.
1. Squamous cell metaplasia is most often an incidental finding associated with inflammation and infarction in BPH nodules. Microscopically, small, solid nests or partially involved glands with a retained lumen are common (e-Fig. 29.11). Squamoid cytoplasm and intercellular bridges may be evident, but keratin pearls are rare. Nuclear atypia, including prominent nuclei and mitoses, may be present in squamous metaplasia adjacent to infarcts. Squamous metaplasia postradiation or hormonal therapy can be more diffuse and is frequently immature with less cytoplasm and an absence of keratinization.
2. Transitional cell or urothelial metaplasia should be distinguished from urothelial cells that normally line the prostatic urethra and central ducts of the prostate. This is usually a focal, incidental finding with small, solid nests or partial gland involvement by cytologically bland and uniform elongated cells, with some cells exhibiting nuclear grooves and cytoplasmic clearing (e-Fig. 29.12).
3. Mucinous metaplasia is replacement of benign luminal epithelium by benign mucin-secretory cells. This is a focal, incidental microscopic finding in which the constituent cells have a granular blue cytoplasmic appearance (e-Fig. 29.13). Goblet cells and luminal secretion of the mucin are uncommon.
4. Eosinophilic metaplasia is the designation for benign epithelium with large supranuclear eosinophilic granules, which represent exocrine differentiation with lysosome-like granules. This uncommon and typically focal change is more often seen in prostatic ductal epithelium and is associated with variable degrees of chronic inflammation and atrophy. Paneth cell-like change, which is also characterized by large eosinophilic cytoplasmic granules, is usually seen in PIN and carcinoma and reflects neuroendocrine differentiation. Paneth cell-like alteration may be found in nonneoplastic prostatic epithelium after radiation.
D. Hyperplasia. BPH is a clinical diagnosis. Histologically, BPH can be diagnosed in TURP chips and simple and radical prostatectomy specimens, but it should not be diagnosed in needle biopsy tissue.
1. Usual nodular epithelial and stromal hyperplasia is the most common morphologic presentation of BPH. Grossly, the nodules, which characteristically arise in the transition zone and periurethral area, are multiple and vary from solid white to spongy with cystic change.
a. Pure stromal nodules (nodular stromal hyperplasia) exist. Microscopically, the nodules can appear myxoid (e-Fig. 29.14), hyalinized, or
leiomyomatous, with spindled, ovoid, or stellate cells. Prominent thickwalled blood vessels and lymphocytes may be noted.
b. Mixed epithelial and stromal hyperplasia is most common, with variable admixtures of spindled stromal cells and complex benign glands with complex papillary and branching architecture (e-Fig. 29.15). Cystic change, inflammation, and basal cell hyperplasia are commonly detected in BPH nodules. Fibroadenomatoid features (e-Fig. 29.16) can rarely be seen.
c. Epithelial predominant BPH nodules (e-Fig. 29.17) are unusual.
d. Infarcts can be identified in larger BPH nodules and can elevate the serum PSA.
2. Basal cell hyperplasia is usually discovered in BPH nodules, but can also be found in peripheral zone needle biopsy tissue, often associated with inflammation. Microscopically, there are two or more layers of basal cells arranged in acinar, cribriform, and solid growth patterns (e-Fig. 29.18). In usual basal cell hyperplasia, the basal cells are uniform and cytologically bland, whereas in so-called atypical basal cell hyperplasia prominent nucleoli are discerned. The term “atypical” should be avoided because no form of basal cell hyperplasia is a known risk factor for neoplasia.
3. Cribriform hyperplasia (e-Fig. 29.19), which is completely benign and not a risk factor for neoplasia, is an infrequently seen variant of BPH. The luminal lining cells are cytologically bland and there is a prominent rim of basal cells.
4. Mesonephric remnant hyperplasia is a very rare prostatic proliferation displaying a vaguely lobular or infiltrative pattern of small tubules with cuboidal epithelium and intraluminal, eosinophilic secretions (Am J Surg Pathol. 2011;35:1054). Immunostains for high-molecular-weight cytokeratin (34betaE12) and/or p63 can be negative in some cases, and AMACR can be focally positive. These results may raise concern for prostatic adenocarcinoma, but helpful clues are negative PSA and prostate-specific acid phosphatase (PSAP) but positive PAX8 immunostains.
5. Verumontanum gland hyperplasia is a benign, small gland proliferation of the verumontanum and adjacent posterior urethra (e-Fig. 29.20). The closely packed glands can architecturally be alarming, but the lack of nuclear atypia and the presence of basal cells rules out carcinoma.
E. Atypical adenomatous hyperplasia (adenosis) is a nodular proliferation of closely packed small acini (e-Fig. 29.21). It is invariably an incidental histologic finding, most often found in transition zone tissue in TURP chips or prostatectomy specimens. The densely packed small pale acini are sometimes intermingled with larger, more complex glands. Nuclear atypia is absent to minimal. The basal cell layer is fragmented, and, on average, 50% of glands completely lack basal cells. Of note, AMACR is diffusely positive in about 8% of cases. Adenosis can be mistaken for well-differentiated Gleason score 2 to 4 adenocarcinoma. It does not have known premalignant potential.
F. Prostatic intraepithelial neoplasia (PIN) is a proliferation of atypical epithelial cells in preexisting ducts and acini (synonyms used in the past include atypical hyperplasia and dysplasia). Currently, PIN is graded as low-grade PIN and highgrade PIN (HG-PIN), although only HG-PIN has potential clinical significance and merits reporting. Isolated HG-PIN is diagnosed in about 5% to 10% of needle biopsies (J Urol. 2006;175:820). It is found in the vast majority of radical prostatectomy specimens with prostatic carcinoma. Microscopically, there are four major structural patterns of HG-PIN growth: tufting, micropapillary, cribriform, and flat (Mod Pathol. 2004;17:360) (Fig. 29.2 and e-Fig. 29.22). These patterns are often admixed. At high-power magnification, HG-PIN shows basal cells (which are typically reduced in number) and atypical luminal cells. Nuclear abnormalities that should be present to diagnose HG-PIN include increased nuclear size, increased chromatin clumping and content, and prominent nucleoli.
The diagnosis can usually be made on H&E-stained sections. Immunostains for basal cells (34betaE12 and p63) and AMACR can be useful when the differential diagnosis is HG-PIN with outpouching versus HG-PIN with associated invasive adenocarcinoma (Am J Surg Pathol. 2005;29:529). Isolated HG-PIN in needle biopsy and TURP chips has been considered a risk factor for subsequent detection of carcinoma on rebiopsy, although the level of risk has decreased with increased 10- to 12-core sampling of the prostate (Am J Surg Pathol. 2005;29: 1201; Urology. 2005;65: 538), such that not all patients necessarily need to undergo rebiopsy in the first year following diagnosis of isolated HG-PIN (J Urol. 2006;175: 820). However, patients with two or more cores with HG-PIN do appear to be at increased risk for subsequent detection of carcinoma and should be considered candidates for rebiopsy (J Urol. 2009;182: 485).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree