Pancreaticoduodenectomy (Whipple Operation) and Total Pancreatectomy for Cancer
Douglas B. Evans
Kathleen K. Christians
W. Dennis Foley
Introduction
Pancreaticoduodenectomy was first formally described by Whipple, Parsons, and Mullins in 1935 in the Annals of Surgery. The original operation was completed in two stages. The first stage consisted of a gastroenterostomy, ligation of the common duct, and cholecystogastrostomy. Three to 4 weeks later, the descending portion of the duodenum and a portion of the pancreatic head were resected and the ducts of Wirsung and Santorini were ligated. The pancreas was not internally drained because pancreatic juice was not considered essential to life and as stated in their manuscript, “activation of the pancreatic ferments by duodenal contents compromised any type of anastomosis in the human subject.” Between 1927 and 1935, 80 cases were reported and were associated with an operative mortality rate of 35%. By 1945, modifications of the two-stage procedure were numerous, and Whipple himself “condemned” the operation advocating, instead, the one-stage procedure with choledochojejunostomy and implantation of the pancreatic duct into the jejunum. Waugh and Clagett from the Mayo Clinic subsequently reported that a pancreaticojejunostomy, choledochojejunostomy, and a retrocolic, antiperistaltic gastrojejunostomy distal to the biliary and pancreatic anastomoses was the most effective operation; little has been altered since their report in 1946. Waugh and Clagett stated that the objective of an operation for the cure of malignant lesions in any location should be constant: (a) the procedure should be radical enough to give reasonable opportunity for cure, (b) the operative mortality should not outweigh the prospects for cure, and (c) the patient should be left in as normal a condition as possible. These basic principles still stand true today.
Recent advances in surgical technique, anesthesia, and critical care have resulted in a 30-day in-hospital mortality rate of <2% for pancreaticoduodenectomies performed at major referral centers by experienced surgeons. Achieving such a low surgery-related mortality rate is a function of the level of experience of the individual surgeon as well as the institution. Although the relationship between volume and outcome is not fully characterized, high-volume hospitals tend to have more surgeons specializing in specific procedures, more consistent postoperative care, better-staffed ICUs, and greater resources to deal with postoperative complications. Patient selection, preoperative medical evaluation, surgical technique, and postoperative care are all critically important in minimizing patient morbidity and mortality and optimizing long-term oncologic outcome. A reasonable level of experience with major pancreatic resection is thus necessary to achieve good results.
Current treatment strategies for patients with localized pancreatic cancer are based on knowledge of the disease’s natural history and patterns of treatment failure. Patients who undergo multimodality therapy that includes pancreaticoduodenectomy for adenocarcinoma of the pancreatic head have a long-term survival rate approaching 30% and a median survival of 2 to 3 years. While vastly improved over the past decade, this relatively modest median survival mandates that treatment-related morbidity be low and treatment-related mortality be rare. Accurate preoperative imaging and proper surgical technique can minimize local recurrence. Routine use of multimodality therapy (chemotherapy with or without chemoradiation) maximizes survival duration by treating the microscopic metastatic disease present in most patients at the time of surgery. Operations resulting in grossly positive margins (R2 resections) offer no survival benefit over palliative chemotherapy and radiation. Therefore, pancreaticoduodenectomy should only be performed when there is a reasonable expectation that all gross disease can be removed. In addition, the development of endoscopic and percutaneously placed stents has allowed most patients with locally advanced, unresectable disease to avoid laparotomy for biliary or gastric decompression. This chapter focuses on the proper use of preoperative diagnostic studies and the technical maneuvers involved in the performance of pancreaticoduodenectomy and total pancreatectomy. Investigational techniques for early diagnosis and details of multimodality therapy are beyond the scope of this chapter but have been reviewed in recent publications listed in selected references.
Advanced imaging technology has improved preoperative clinical staging accuracy to the degree that “exploratory surgery” to determine resectability for known or suspected pancreatic or periampullary cancer is no longer necessary.
Preoperative evaluation should include a detailed history and physical examination (including functional status), chest imaging, laboratory studies including tumor markers (CA19-9, CEA), contrast-enhanced pancreas-protocol computed tomography (CT) of the abdomen, and further evaluation of comorbid conditions as indicated. The CT allows assessment of the tumor’s relationship to the superior mesenteric artery (SMA), the superior mesenteric vein (SMV) and SMV-portal vein confluence (SMV-PV), and the celiac axis and hepatic artery (HA). CT also defines any arterial or venous aberrations (replaced left or right HA, IMV draining directly into the SMV, first jejunal branch draining anterior to the SMA, etc.) and highlights potential nodal or extrapancreatic metastases. Clinicians can then place the patient into one of four categories based on appropriately performed CT imaging: (a) resectable, (b) borderline resectable, (c) locally advanced, and (d) metastatic. These categories help guide multidisciplinary management as will be discussed later in this chapter.
The aim of a pancreas-protocol CT is to obtain three-dimensional submillimeter isotropic voxel resolution of the organ during optimum phases of both pancreatic and peripancreatic vascular enhancement. To achieve this objective, detector elements with projected resolution of 0.4 to 0.7 mm at isocenter with submillimeter detector rows of 0.5 to 0.75 mm are required. This can be achieved with 16-channel, 64-channel, or greater devices. The advantage of 64-channel or greater is rapid image acquisition in the z or longitudinal axis. Contrast-enhancement depends upon iodine delivery and linking of image acquisition to defined circulation phases. Contrast delivery should be on the order of 4 to 6 mL/s (350–370 mg of iodine/mL), preferably via an 18 to 20 gauge venous catheter placed in an antecubital vein and delivered via a programmed
power injector. Individual patient circulation times vary considerably and it is critical that the contrast delivery be tailored to each individual patient. This can be done either with a preliminary mini bolus or bolus tracking software. For pancreatic imaging, the stomach and duodenum are distended with 800 mL of water administered 15 to 30 minutes prior to study and an additional 200 mL of water immediately prior to scan.
power injector. Individual patient circulation times vary considerably and it is critical that the contrast delivery be tailored to each individual patient. This can be done either with a preliminary mini bolus or bolus tracking software. For pancreatic imaging, the stomach and duodenum are distended with 800 mL of water administered 15 to 30 minutes prior to study and an additional 200 mL of water immediately prior to scan.
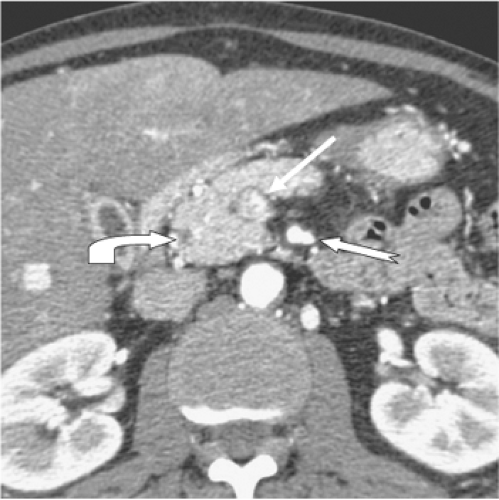
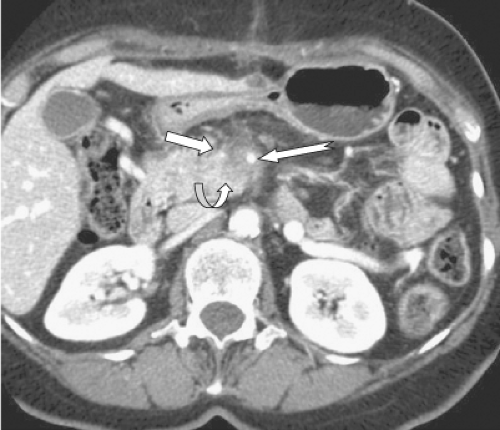
The “pancreatic phase,” the phase of optimum pancreatic parenchymal enhancement, is a capillary phase acquisition occurring 15 seconds after aortic arrival of the contrast medium following the peripheral intravenous bolus injection. Antecubital vein to abdominal aortic circulation time can vary between 12 and 30 seconds in individual patients, thus explaining the critical role of circulation timing in providing consistent reproducible pancreatic contrast-enhancement. The pancreatic phase, also labeled as a “late arterial phase” also corresponds in timing to the initial opacification of the superior mesenteric and portal vein which predictably occurs 10 to 15 seconds after aortic arrival of contrast material in the SMA. Thus, while this phase provides optimum delineation of the pancreas and peripancreatic arteries (Fig. 1), the opacification of the splenic, superior mesenteric, and portal veins is not yet at peak.
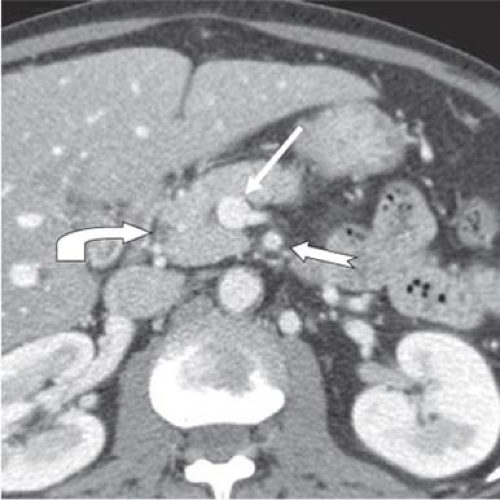
A second phase, commonly delineated as the “portal venous phase,” occurs 45 seconds after the pancreatic phase (Fig. 2). In this phase, the spleen that was inhomogeneous in the pancreatic phase now becomes uniformly homogeneous and the hepatic veins that were not enhanced in the late arterial phase now become uniformly enhanced. In addition, the liver that was less enhanced than the pancreas in the pancreatic phase now becomes more enhanced than the pancreas due to contrast material inflow from the portal vein into the hepatic sinusoids.
The two pancreatic phases thus defined have different roles in evaluation of focal pancreatic pathology. The pancreatic phase provides maximum contrast differentiation between relatively hypovascular, hypoattenuating tumors (most commonly pancreatic adenocarcinoma) and normal pancreatic parenchyma, or the converse, between hypervascular hyperattenuating tumors (most commonly neuroendocrine tumors) and background pancreatic parenchyma. In addition, the pancreatic and intrapancreatic common ducts are best defined in
this phase. In the pancreatic phase, boundaries of pancreatic neoplasms in relation to critical arterial structures (celiac, CH, splenic, and SMA) are delineated. Aberrant or replaced right and left hepatic arteries are also displayed in this phase.
this phase. In the pancreatic phase, boundaries of pancreatic neoplasms in relation to critical arterial structures (celiac, CH, splenic, and SMA) are delineated. Aberrant or replaced right and left hepatic arteries are also displayed in this phase.
The portal venous or hepatic phase best delineates the extra hepatic portal venous system and in conjunction with the late arterial phase delineates tumor margin in relationship to the SMV and its proximal tributaries including gastroepiploic, middle colic and jejunal veins, the splenic vein, the inferior mesenteric vein (IMV) whether inserted into the splenic or proximal SMV, and the portal vein. Peripancreatic lymphadenopathy and hepatic metastases are generally best defined in the portal venous or hepatic phase. However, the liver is also completely imaged in the pancreatic phase and metastases may be recognized as ring-enhancing lesions that become uniformly hypoattenuating in the hepatic phase.
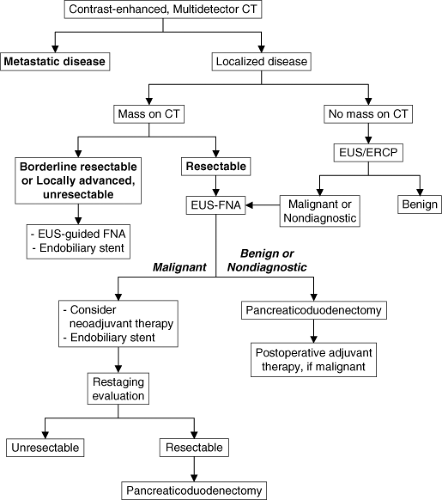
Our current algorithm for the diagnosis and treatment of patients with presumed or biopsy-proven adenocarcinoma of the pancreatic head or periampullary region is illustrated in Figure 3. CT imaging should always be performed prior to diagnostic procedures to avoid inaccurate reads in the event of procedural-induced pancreatitis. Patients with a visible mass on pancreas-protocol CT will undergo endoscopic ultrasound-guided fine-needle aspiration biopsy (EUS-FNA) for tissue diagnosis. FNA is not necessary if the results will not influence the treatment plan. Negative results from EUS-FNA should not be considered definitive proof of benign disease. Cytopathologists should be present in the endoscopy area at the time of the procedure to process tissue samples. Their presence at the time of EUS-FNA has considerably improved the diagnostic yield of this procedure to over 90%. Patients who are jaundiced will then undergo endoscopic retrograde cholangiopancreatography (ERCP) with placement of an endobiliary stent (preferably as a combined procedure with EUS). Those patients with a low-density mass in the pancreatic head on CT imaging and a positive tissue diagnosis are eligible for neoadjuvant therapy (hopefully as part of a clinical trial). Those with an obvious cancer and/or positive tissue diagnosis have metal stents placed (vs. plastic) to avoid stent occlusion during induction therapy. Patients with extrahepatic biliary obstruction without a CT-defined mass in the pancreas undergo upper endoscopy and EUS. Endoscopic evaluation may discover an ampullary tumor or related pathology, and EUS may define a mass in the pancreas or distal bile duct not seen on CT. A double-duct sign indicates proximal obstruction of the common bile duct and pancreatic ducts. If the etiology of biliary obstruction remains unclear, ERCP (or magnetic resonance cholangiopancreatography [MRCP]) is usually necessary. At the time of ERCP, a malignant obstruction of the intrapancreatic portion of the common bile duct is characterized by an abrupt cutoff, or irregular stenosis of the common bile duct. This is in contradistinction to a long, smooth tapering stricture seen in chronic pancreatitis or a filling defect or meniscus found in choledocholithiasis. Endoscopic stents are placed to prevent cholangitis in patients with extrahepatic biliary obstruction undergoing diagnostic ERCP.
In a patient who presents with extrahepatic biliary obstruction, a malignant-appearing stricture of the intrapancreatic portion of the common bile duct, and no history of recurrent pancreatitis or alcohol abuse, the absence of a mass on CT or EUS images does not rule out carcinoma of the pancreas or bile duct. Instead, the results of EUS, with or without FNA, should be considered within the clinical context, as a complement to CT and ERCP findings. FNA biopsy should only be performed if a mass is identified unless an experienced endoscopic ultrasonographer is capable of directing the biopsy at the point of the pancreatic or bile duct stricture. Blind biopsy of
the pancreas should be avoided. If tissue diagnosis is not obtainable, these patients are taken to the operating room for pancreaticoduodenectomy based on the high index of clinical suspicion.
the pancreas should be avoided. If tissue diagnosis is not obtainable, these patients are taken to the operating room for pancreaticoduodenectomy based on the high index of clinical suspicion.
For patients with a pancreatic head mass, CT imaging is used to define the extent of disease. Classifications include resectable, borderline resectable, locally advanced, or metastatic. Only patients characterized as resectable or borderline resectable are considered eligible for pancreaticoduodenectomy.
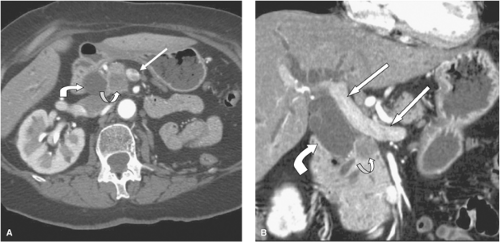
CT criteria for a resectable tumor include: (a) the absence of extrapancreatic disease, (b) a patent SMV-PV confluence, and (c) no direct tumor extension to the HA, celiac axis, or SMA. The AHPBA/SSO consensus statement took a more conservative view of venous involvement and defined resectable tumors as those with no radiographic evidence of SMV and PV abutment, distortion, tumor thrombus, or venous encasement (Fig. 4). A clear soft tissue plane must also be present around the celiac axis, HA, and SMA. There is evolving consensus that a surgery-first strategy should be considered only for pancreatic tumors with no radiographic evidence of extension to adjacent vascular structures.
Borderline resectable patients as described by Katz encompass three types: Type A patients have tumor-artery abutment of ≤180 degree of the circumference of the SMA or celiac axis, tumor abutment or encasement (>180 degree) of a short segment of the HA (usually at the origin of the gastroduodenal artery [GDA]), or short segment occlusion of the SMV, PV, or SMV-PV confluence with a suitable PV above and SMV below for reconstruction. Type B patients have CT findings suspicious for, but not diagnostic of, metastatic disease. These patient’s tumors may be resectable or borderline resectable. Type C patients are borderline resectable due to a marginal performance status as well as those with severe preexisting comorbidities requiring protracted evaluation that precludes immediate operation. This category does not include those with no potential for eventual operation (i.e., critical aortic stenosis). Colon and/or mesocolon invasion is not included in the definition of borderline resectable cancer, both because it is not quantifiable and because removal of the mesocolon or colon is unrelated to the risk for a margin-positive resection.
Patients with borderline resectable tumors as defined above are not taken directly to surgery. The rationale for delivering neoadjuvant therapy to patients with borderline resectable tumors is based on: (a) providing a time interval to gauge the aggressiveness of the cancer in order to select patients for surgery who have stable or responding disease. Those with disease stability or treatment response have the greatest likelihood of a favorable oncologic outcome—an important factor given that the morbidity of the operation is potentially high and the anticipated survival duration modest; (b) the early treatment of micrometastatic disease, which exists in the majority of patients; (c) giving “adjuvant” therapy in a “neoadjuvant” setting, when it is given to a well-vascularized tumor, is potentially more effective and better tolerated, and (d) the potential for tumor downstaging (tumor destruction, particularly at the periphery) to maximize the potential for an R0/R1 resection.
Locally advanced tumors are defined as those with encasement (>180 degree) of adjacent arteries (Fig. 5) or an occluded SMV-PV with no technical option for resection and reconstruction. Locally advanced (Stage III) tumors should not be considered for operation. Contemporary chemotherapy and radiation will not convert a tumor with CT evidence of arterial encasement into a resectable tumor. In contrast to the experience with venous resection, the limited experience with arterial resection and reconstruction as part of a regional pancreatectomy for pancreatic adenocarcinoma has been associated with unacceptably high perioperative morbidity
and mortality. In addition, the celiac axis and SMA are surrounded by a dense perineural plexus (not present around the SMV or PV) and once the tumor has access to this perineural plexus, retroperitoneal tumor extension is present, and even with arterial resection, it is unlikely that negative resection margins can be achieved. Therefore, surgical resection in patients with clear evidence of arterial encasement on preoperative CT scans is not recommended. Because of the accuracy of CT in predicting resectability, the majority of patients with disease determined to be unresectable by CT based on arterial encasement can be spared laparotomy. As mentioned previously, biliary and gastric decompression can be performed endoscopically if necessary.
and mortality. In addition, the celiac axis and SMA are surrounded by a dense perineural plexus (not present around the SMV or PV) and once the tumor has access to this perineural plexus, retroperitoneal tumor extension is present, and even with arterial resection, it is unlikely that negative resection margins can be achieved. Therefore, surgical resection in patients with clear evidence of arterial encasement on preoperative CT scans is not recommended. Because of the accuracy of CT in predicting resectability, the majority of patients with disease determined to be unresectable by CT based on arterial encasement can be spared laparotomy. As mentioned previously, biliary and gastric decompression can be performed endoscopically if necessary.
CT is not as accurate in predicting the need for venous resection. However, the loss of the normal tissue plane between the tumor and the SMV, SMV-PV distortion, or tumor abutment should alert the surgeon preoperatively to the potential for direct tumor invasion of the vessel wall. If any of these findings are noted on preoperative CT imaging, surgery should not be undertaken unless the surgeon has a technical strategy for the intraoperative management of venous involvement. Accurate preoperative assessment of resectability helps limit intraoperative indecision, decreases operative time, and lessens the patient’s risk of morbidity/mortality related to technical misadventures. A high rate of resectability (80%) and a low rate of margin positivity (10% to 20%) can be achieved by utilizing CT criteria to determine whether a laparotomy is indicated.
Laparoscopy has been used in patients with radiographic evidence of localized pancreatic cancer to detect extrapancreatic tumor not seen on CT imaging, thereby allowing laparotomy to be limited to patients with localized disease. It is important to remember that 70% to 80% of patients with pancreatic cancer present with locally advanced or metastatic disease; if laparoscopy is done early in the diagnostic sequence, it will have a very high yield of positive findings. In contrast, if laparoscopy is performed after high-quality CT, the yield will likely be much lower. Studies have suggested that CT occult extrapancreatic disease is uncommon, being found in no more than 10% to 15% of patients with tumors considered resectable following high-quality CT. Laparoscopy prior to laparotomy (under a single anesthesia induction) is routinely used by the authors in patients with biopsy-proven or suspected potentially resectable pancreatic cancer in whom a decision has been made to proceed with pancreaticoduodenectomy. Data are not available to support the cost-effectiveness of laparoscopy’s routine use as a staging procedure under a separate anesthesia induction prior to treatment planning. In addition, laparoscopy should not be a substitute for inadequate CT imaging.
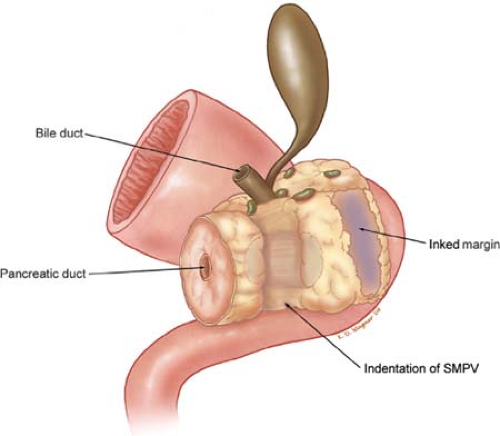
The SMA margin of resection is the soft tissue margin adjacent to the proximal 3 to 4 cm of the SMA (Fig. 6). This margin has previously been referred to as the retroperitoneal, uncinate, or the mesenteric margin of resection. The surgical technique described
herein emphasizes the direct identification of the SMA and removal of all perivascular and lymphatic tissue to the right of this vessel. This more aggressive retroperitoneal/mesenteric dissection combined with the other components of multimodality therapy improves local disease control, alters patterns of recurrence, and represents a logical strategy to maximize length and quality of patient survival.
herein emphasizes the direct identification of the SMA and removal of all perivascular and lymphatic tissue to the right of this vessel. This more aggressive retroperitoneal/mesenteric dissection combined with the other components of multimodality therapy improves local disease control, alters patterns of recurrence, and represents a logical strategy to maximize length and quality of patient survival.
Pancreaticoduodenectomy
Pancreaticoduodenectomy involves removal of the pancreatic head, duodenum, gallbladder, and bile duct with or without removal of the gastric antrum. It represents the standard surgical procedure for neoplasms of the pancreatic head and periampullary region. At the time of surgical exploration for pancreatic head cancer, a Kocher maneuver was commonly taught to be the first step performed to allow assessment of the relationship of the tumor to the SMA by palpation. By mobilizing the pancreatic head and duodenum from their retroperitoneal attachments, the surgeon attempts to palpate a plane of normal tissue between the firm tumor and the posterior pulsation of the SMA. In reality, this step is a crude assessment subject to individual surgeon bias. The relationship of the tumor to the right lateral wall of the SMA is the most critical aspect of the pretreatment staging evaluation. This tumor–vessel relationship must be accurately evaluated prior to taking the patient to the operating room and cannot be accurately assessed intraoperatively following a Kocher maneuver. In addition, for larger tumors, for those tumors containing significant peritumoral fibrosis, or for reoperative cases, palpation of the relationship of the primary tumor to the SMA after Kocher maneuver with any reasonable degree of accuracy is usually impossible.
The second maneuver traditionally performed to assess resectability is to develop a plane of dissection between the anterior surface of the SMV-PV confluence and the posterior surface of the neck of the pancreas to exclude tumor involvement of the SMV or SMV-PV confluence (Fig. 7); such tumor involvement, in the opinion of most surgeons, precludes resection. However, the rationale for this maneuver early in the operation is also unclear. Tumors of the pancreatic head or uncinate process are prone to invade the lateral or posterior wall of the SMV-PV confluence, not the anterior wall. The anterior wall is rarely involved in the absence of encasement of the celiac axis or SMA origin (as seen with locally advanced tumors of the pancreatic neck or body), and this finding should be visible on preoperative CT images. In addition, the relationship of a pancreatic head tumor to the lateral and posterior walls of the SMV-PV confluence (and the SMA) can be directly inspected only after gastroduodenal and pancreatic transection, by which point the surgeon has already committed the patient to resection. Therein lies the rationale for detailed preoperative imaging using high-quality, contrast-enhanced CT, with laparotomy undertaken only in patients who have CT-defined resectable tumors. Both of the above described maneuvers are founded in surgical tradition rather than surgical anatomy—they are not accurate techniques for the assessment of resectability of a primary carcinoma of the pancreatic head.
Pancreatic Resection
Our recommended technique for pancreaticoduodenectomy utilizes a midline upper abdominal incision (after routine laparoscopic exploration confirms the absence of extrapancreatic metastatic disease). Lymph node biopsy for frozen-section analysis is controversial. Positive lymph nodes are a prognostic factor (along with poorly differentiated histology, microscopically positive resection margins, etc.), predictive of decreased survival duration. However, the majority (60% to 90%) of resected specimens contain microscopic metastases in regional lymph nodes. In a good-risk patient with localized, resectable pancreatic cancer, we do not view lymph node metastases as an absolute contraindication to pancreaticoduodenectomy when performed as part of a multimodality approach to pancreatic cancer. Therefore, in general, we do not perform random lymph node sampling for frozen-section analysis at the time of pancreaticoduodenectomy. However, each case should be considered individually such as in the case of a high-risk patient (due to medical comorbidities or oncologic concerns) with suspicious adenopathy, a positive regional lymph node may be viewed as a contraindication to proceeding with pancreaticoduodenectomy.
The surgical resection is divided into the following clearly defined steps:
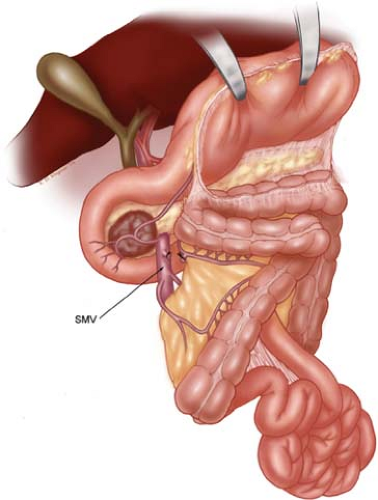
The purpose of step 1 is to isolate the infrapancreatic SMV and separate the colon (and its mesentery) from the duodenum and pancreatic head (Fig. 8). The lesser sac is entered by taking the greater omentum off of the transverse colon; and the loose attachments of the posterior gastric wall to the anterior surface of the pancreas. The hepatic flexure of
the colon is mobilized from its retroperitoneal attachments. The visceral peritoneum along the inferior border of the pancreas from a point medial to (to the patient’s left of) the middle colic vessels is incised and carried out laterally (to the patient’s right) and inferiorly to expose the junction of the middle colic vein and the SMV. The middle colic vein may enter directly into the anterior surface of the infrapancreatic SMV or arise as a common trunk with the gastroepiploic vein (gastrocolic trunk). If the middle colic and gastroepiploic veins share a common trunk, the common trunk can be preserved (and the gastroepiploic vein divided) or the entire trunk divided; otherwise, the gastroepiploic vein is left intact and divided later in the operation following pancreatic transection. When necessary, the middle colic vein can be divided proximal to its junction with the SMV; this is often needed when operating on obese patients where a traction injury of the middle colic vein may occur if the vein is not divided. Division of the middle colic vein is especially important when dealing with large tumors or tumors extending inferiorly from the uncinate process. In fact, the base of the transverse mesocolon and the anterior leaf of the small-bowel mesentery can be left attached to the tumor and resected en bloc with the pancreatic head. Such caudal or inferior tumor extension is not a contraindication to pancreaticoduodenectomy but does add complexity during this step when the infrapancreatic SMV needs to be identified.
Mobilization of the retroperitoneal attachments of the small-bowel- and right colon mesentery is performed in patients with uncinate tumors extending into the small-bowel mesentery and in some patients who require venous resection and reconstruction. This maneuver was first described by Cattell and Braasch for exposure of the duodenum. The visceral peritoneum of the small-bowel mesentery is incised to the ligament of Treitz, thereby mobilizing the small-bowel mesentery. When complete, this maneuver allows cephalad retraction of the right colon and small bowel, exposing the third and fourth portions of the duodenum.
Occasionally, when there is extensive inflammatory change or scarring at the root of mesentery usually due to a prior attempt at resection, we may not identify the SMV early in the operation. In such cases, the SMV will be exposed during step 6 when the pancreas is divided into a caudal direction from the level of the portal vein. Due to lack of vascular control, this can be dangerous but it is a valuable technique for the experienced surgeon who deals with more complicated pancreatic resections especially in a reoperative setting.
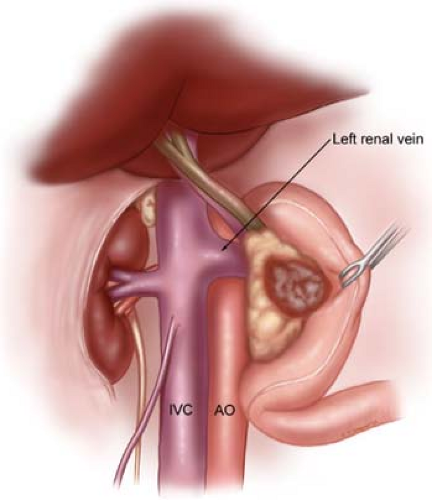
The Kocher maneuver is begun at the transverse portion (third portion) of the duodenum by identifying the inferior vena cava. All fibrofatty and lymphatic tissues medial to right gonadal vein and anterior to the inferior vena cava are elevated, along with the pancreatic head and duodenum (Fig. 9). The Kocher maneuver is continued to the left lateral edge of the aorta, with exposure of the anterior surface of the left renal vein. The right gonadal vein serves as a good landmark coursing anterior/medial to the right ureter, thus helping prevent inadvertent ureteric injury. A complete Kocher maneuver is necessary for the subsequent dissection of the pancreatic head from the SMA (step 6). Particularly important is the division of the leaf of peritoneum that is posterior to the mesenteric vessels; incision of this portion of peritoneum, as it extends from the retroperitoneum to the right lateral aspect of the root of the small-bowel mesentery, is necessary prior to performing the SMA dissection.
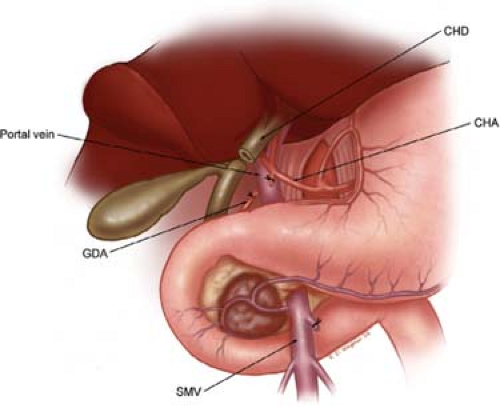
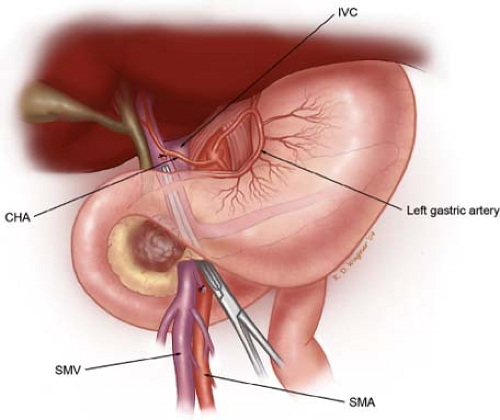
Fig. 10. Illustration of step 3. Dissection of the porta hepatis begins with identification of the common hepatic artery (CHA), by removal of the large lymph node, which commonly sits anterior to this vessel. The CHA is then followed distally to allow identification and ligation and division of the right gastric artery (not shown) and the gastroduodenal artery (GDA). This allows the CHA-proper hepatic artery to be mobilized cephalad and medial off of the underlying anterior surface of the portal vein (PV). The PV is always identified prior to division of the common hepatic duct (CHD). Careful palpation of the porta hepatis prior to division of the bile duct should alert one to the possibility of an accessory or replaced right hepatic artery traveling posterior to the bile duct and lateral to the portal vein. The CHD is divided at the level of the cystic duct. SMV, superior mesenteric vein.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access