Chapter 10
Neuroanatomy of the Autonomic Nervous System
Sympathetic, Parasympathetic, and Enteric Divisions
Innervation of Autonomic Effectors
Innervation of Peripheral Effectors
Innervation of the Heart and Lungs
Innervation of the Lower Urinary Tract
Efferent Response and the Cholinergic Anti-inflammatory Pathway
Sympathetic Innervation of Lymphoid Tissue
Neurotransmission Occurring at the Autonomic Ganglia and Neuroeffector Junctions
Control of Autonomic Efferents: Central Autonomic Network
Sympathetic, Parasympathetic, and Enteric Divisions
The ANS is composed of a sympathetic and a parasympathetic division. These two divisions are discussed here, followed by a description of a third division of the ANS. This third division is the enteric nervous system, which is a complex network of neurons located within the wall of the gut. Typically the anatomy and functions of the parasympathetic and sympathetic divisions are described separately. This method of introducing the basic concepts and components of the divisions may lead to the misconception that they function antagonistically and separately. This is certainly not the case. In fact, the fibers of both divisions are tonically active, and in most cases do not function as either “on” or “off.” Although some tissues are predominantly or exclusively innervated by one division (e.g., sympathetic peripheral effector tissues), many tissues have a dual innervation. A balance of activity exists between the division fibers in these cases, such that when one division increases its activity, the other division decreases its activity. The central nervous system (CNS) has the ability to alter the balance of activity between these divisions. In addition, the sympathetic and parasympathetic divisions interact with the somatic nervous system and different hormonal systems to stabilize the internal environment and maintain homeostasis during normal conditions or emergency situations.
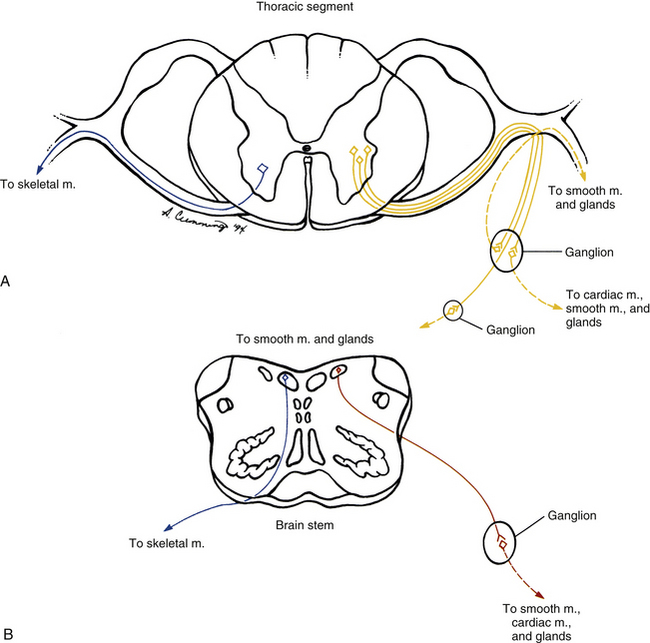
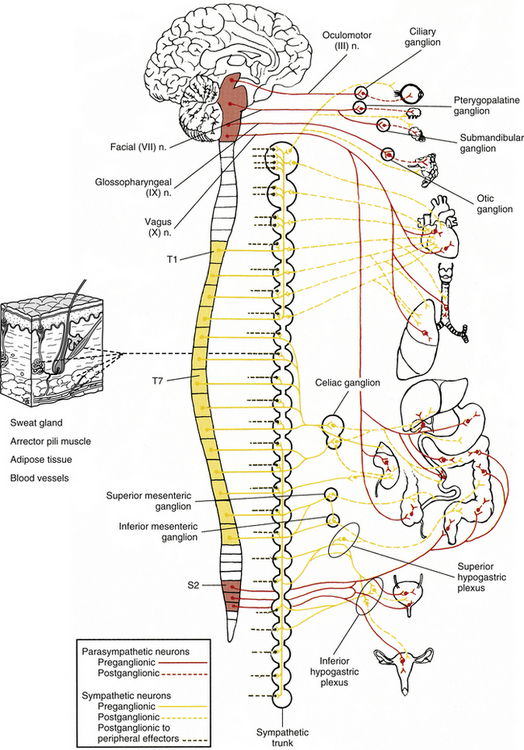
The ANS can be described best after certain characteristics common to both the sympathetic and parasympathetic divisions have been reviewed. The parasympathetic and sympathetic innervation of autonomic effectors (i.e., organs, vessels, glands) is organized differently than the innervation of skeletal muscle (Fig. 10-1). Although the axons of alpha and gamma motor neurons course directly to skeletal muscles, the innervation of ANS effectors requires a chain of two neurons (see Fig. 10-1), called the preganglionic and postganglionic neurons. The cell body of the preganglionic neuron always is located in the CNS—either in the spinal cord or in the brain stem (Fig. 10-2). The axon is thinly myelinated and immediately leaves the CNS within a specific ventral root of the spinal cord or within certain cranial nerves exiting the brain stem. The cell body of the postganglionic neuron is located in an autonomic ganglion that may be found in numerous places outside the CNS (see Fig. 10-2). The preganglionic neuron synapses with the postganglionic neuron within this ganglion. The axon of the postganglionic neuron is unmyelinated and innervates the effector. Both the preganglionic and postganglionic neurons frequently travel in components of the peripheral nervous system (PNS) (i.e., spinal nerves, cranial nerves) and are intermingled with afferents and somatic motor neurons of peripheral nerves. As stated, most effectors are innervated by both sympathetic and parasympathetic fibers (Table 10-1; see also Fig. 10-2). These fibers produce antagonistic but coordinated responses in the effectors. Descending input from higher integrative centers such as the hypothalamus and areas of the brain stem reaches the cell bodies of preganglionic fibers to regulate and adjust their activity. This descending input is a part of several specific visceral reflex pathways and also is used by higher centers to institute widespread bodily changes.
Table 10-1
Functions of the Sympathetic and Parasympathetic Divisions
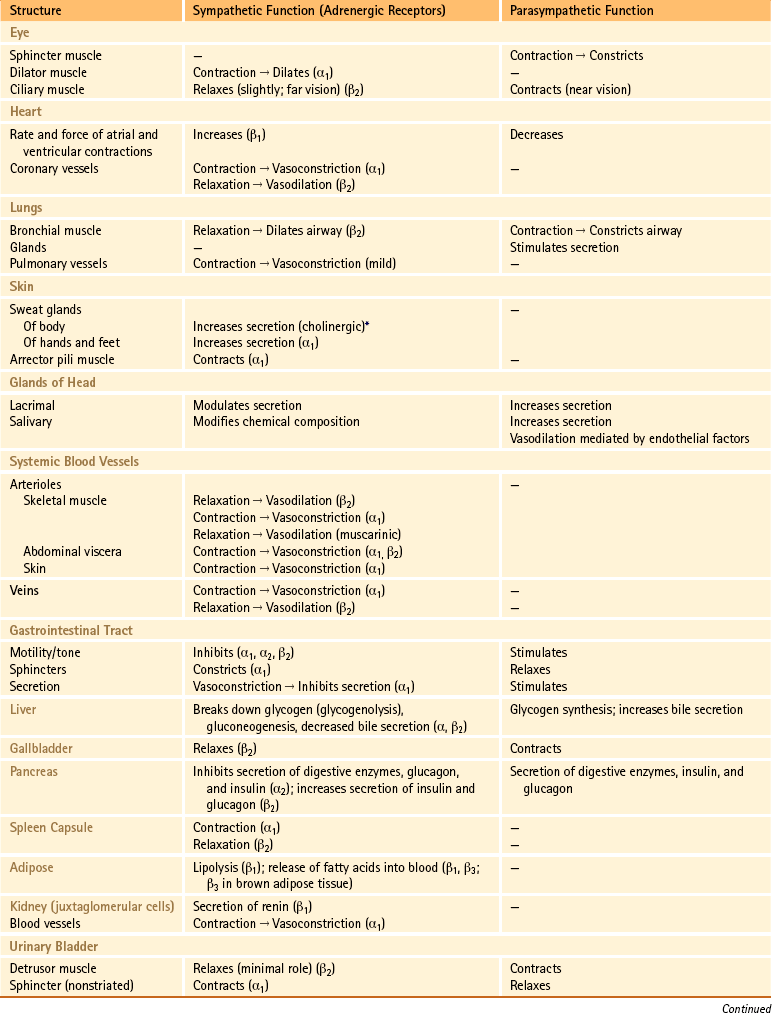

∗Sweat glands are an exception; their sympathetic fibers release ACh, which binds to cholinergic receptors.
From Benarroch EE et al. (1999). Medical neurosciences (4th ed.). New York: Lippincott Williams & Wilkins; Bray JJ et al. (1994). Lecture notes on human physiology (3rd ed.). Cambridge, UK: Blackwell Science; FitzGerald MJT & Folan-Curran J. (2002). Clinical neuroanatomy and related neuroscience (4th ed.). Philadelphia: WB Saunders; Tortora GJ & Derrickson B. (2009). Principles of anatomy and physiology (12th ed.). Hoboken, NJ: John Wiley & Sons; Waxman SG. (2003). Clinical neuroanatomy (25th ed.). Chicago: McGraw-Hill.
Sympathetic Division
Preganglionic Sympathetic Neurons
The cell bodies of the preganglionic sympathetic neurons are located in the spinal cord in all thoracic segments and in the upper two or three lumbar segments (see Fig. 10-2). Because of the distribution of these preganglionic cell bodies, the sympathetic division of the ANS often is called the thoracolumbar division. These preganglionic neurons comprise a heterogeneous population within the spinal cord. The dendritic arrangement of these neurons ranges from simple to complex arborizations. The cell bodies are of different shapes, and their size falls in a range between the size of smaller dorsal horn neurons and that of larger somatic motor neurons. Of the total membranous surface area of these neurons, the cell body of each composes a maximum of 15%, which likely indicates the importance of the dendritic surface area of that neuron (Cabot, 1990).
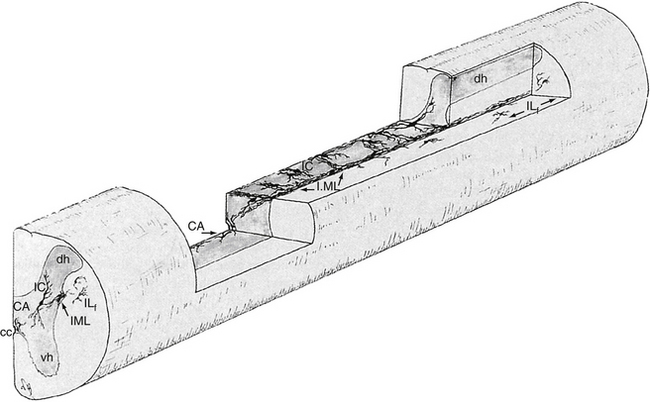
The cell bodies of the preganglionic neurons are found in four nuclei within the intermediate gray matter of the spinal cord (Fig. 10-3) (Cabot, 1990). The largest group of these cell bodies is the intermediolateral (IML) cell column that forms the lateral horn. Throughout this column are clusters of 20 to 100 neurons that are separated by distances ranging from 200 to 500 µm in the thoracic region and from 100 to 300 µm in the lumbar region. The cell bodies are approximately 12 to 13 µm in diameter and histologically are similar to motor neurons (Harati, 1993). The diameters of the axons range from 2 to 5 µm, and their speed of conduction is approximately 3 to 15 m/sec. These fibers often are classified in the B group (see Chapter 9). At the T6 and T7 levels, the mean number of these cells is approximately 5000, but it has been shown that the number decreases with age at the rate of approximately 8% per decade (Harati, 1993).
The other three nuclear groups of preganglionic neurons have been described by Cabot (1990) and are the lateral funicular area (located lateral and dorsal to the intermediolateral group), the intercalated cell group (located medial to the IML column and possibly the same cluster of neurons typically called the intermediomedial group), and the central autonomic nucleus (located lateral and dorsal to the central canal). The combination of these groups forms a ladderlike structure in longitudinal sections in which the paired IML cell columns form the sides of the ladder and the interconnected central autonomic nucleus and intercalated cell group form the rungs (Fig. 10-3). The IML cell column is the origin of the vast majority of preganglionic fibers, but the other three nuclei also give rise to some preganglionic fibers. The four nuclei are the recipients of extensive input from higher centers such as the hypothalamus and brain stem nuclei. These sources release various neurotransmitters that have been identified as monoamines, neuropeptides, and amino acids. Although the anatomic characteristics of these four nuclei have been described, the exact functions of each specific nucleus still remain unclear.
Postganglionic Sympathetic Neurons
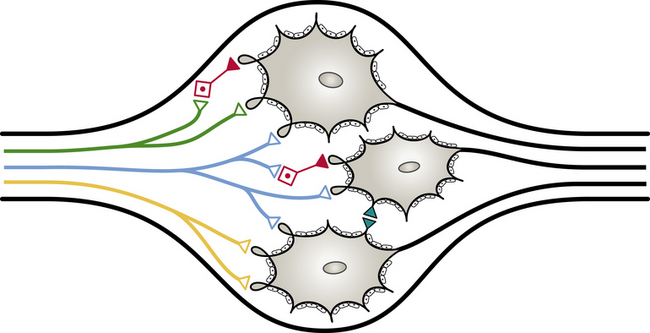
According to the general rule of organization of the ANS, two neurons are necessary for the impulse to reach the effector. One is the preganglionic neuron, just discussed. The second neuron in the pathway to an autonomic effector is the postganglionic neuron. This neuron’s axon is classified as a group C fiber (see Chapter 9). Generally it is described as unmyelinated, with a diameter ranging from 0.3 to 1.3 µm and a slow conduction speed ranging from 0.7 to 2.3 m/sec (Carpenter & Sutin, 1983). The cell body is located outside the CNS in an autonomic ganglion. Unlike a sensory ganglion of cranial nerves and a dorsal root ganglion of spinal nerves, in which no synapses occur, an autonomic ganglion is the location of the synapse between the preganglionic and postganglionic neurons. Preganglionic fibers disseminate their information by diverging and synapsing on numerous postganglionic fibers. This principle of divergence is based on studies of the superior cervical ganglion of mammals. Results of different studies show preganglionic to postganglionic ratios of 1:4 (Loewy, 1990a), 1:15 to 1:20, and 1:196 in a human superior cervical ganglion (Ebbesson, 1968; Standring et al., 2008). (The parasympathetic division also has been found to exhibit divergence, but to a lesser degree.) This divergence may allow the effects of sympathetic stimulation to be more widespread throughout the body and to be of greater magnitude.
The autonomic ganglion in which the synapse occurs may be one of a chain of ganglia (called the sympathetic chain, sympathetic trunk, or paravertebral ganglia) located near the vertebral bodies of the spinal column, or it may be a prevertebral ganglion, such as the celiac ganglion (see Fig. 10-2), found within one of the autonomic nerve plexuses.
These plexuses surround the large arteries in the abdominal and pelvic cavities. The ganglion, regardless of location, is encapsulated by connective tissue. The connective tissue capsule is continuous with the epineurium of the bundle of entering preganglionic neurons and the bundle of exiting postganglionic neurons. Within the capsule are predominantly multipolar, spheroidal-shaped postganglionic neurons (Fig. 10-4). These neurons consist of cell bodies that have diameters ranging from 25 to 50 µm and dendrites that branch in a complex pattern. These dendrites are the location where preganglionic neuron axons commonly synapse. Satellite cells (neuroglial cells) that are similar to those found in the dorsal root ganglia surround the cell bodies and dendrites. These cells provide support and help maintain the chemical environment. Interneurons are also located in the ganglion. One type, called small (cell bodies ranging in diameter from 15 to 20 µm) intensely fluorescent cells (SIFs), is present singly or in clusters. These cells contain the neurotransmitters epinephrine, serotonin, and dopamine (Hamill, 1996), which, when released, bind to and modulate the activity of postganglionic neurons. Another type of interneuron found in the ganglion is the small chromaffin cell, which also contains catecholamines. The exact difference between the chromaffin cell and SIF is unclear (Carpenter & Sutin, 1983; Harati, 1993; Standring et al., 2008).
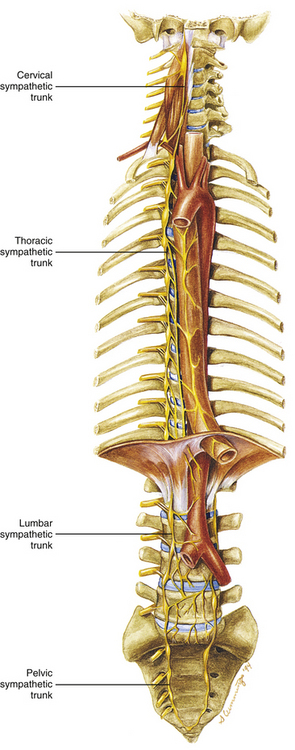
Sympathetic Trunk
Two sympathetic trunks are located in the body, each of which lies on the anterolateral side of the vertebral column (Fig. 10-5). They both extend from the base of the skull to the coccyx. The ganglia of the sympathetic trunks are also called the paravertebral ganglia because they lie next to the vertebral column. Inferiorly the two trunks join in the midline and terminate on the anterior surface of the coccyx as the ganglion impar.
Each sympathetic trunk shares important anatomic relationships with surrounding structures. In the neck it lies between the carotid sheath and prevertebral muscles, which cover the transverse processes (TPs) of the cervical vertebrae. It is found anterior to the heads of the ribs in the thorax, anterolateral to the bodies of the lumbar vertebrae in the abdomen, and medial to the anterior sacral foramina in the pelvis (Standring et al., 2008). As the name sympathetic chain ganglia implies, this structure consists of approximately 22 ganglia that are linked together by connective tissue surrounding ascending and descending fibers. The total number of ganglia does not correspond exactly to the number of spinal nerves because some of the ganglia have fused with one another. This fusion is most evident in the cervical region, where there are only three cervical ganglia (see Cervical Sympathetic Trunk later in this chapter and Cervical Sympathetics in Chapter 5). The thoracic portion of the sympathetic trunk includes 10 to 12 ganglia (70% of the time there are 11), the lumbar region exhibits 4 ganglia (although this number may vary), and 4 or 5 ganglia appear in the sacral region of the trunk. The union of the two sympathetic trunks forms the one coccygeal ganglion.
The preganglionic fibers exit the spinal cord in the ventral roots of cord segments T1 to L2 or L3 to reach the postganglionic neurons. Therefore at these particular levels, the ventral roots include both preganglionic sympathetic fibers and fibers to skeletal muscle (i.e., alpha and gamma motor neurons). The preganglionic fibers continue into the spinal nerve, and at the division of the spinal nerve into its dorsal and ventral rami (posterior and anterior primary divisions, respectively), the myelinated preganglionic fibers exit, forming the white (myelin is a white substance) ramus communicans, and then continue into the sympathetic trunk. (There are only 14 or 15 white rami on each side because there are only 14 or 15 spinal cord segments [T1 to L2-3] that provide preganglionic sympathetic fibers.)
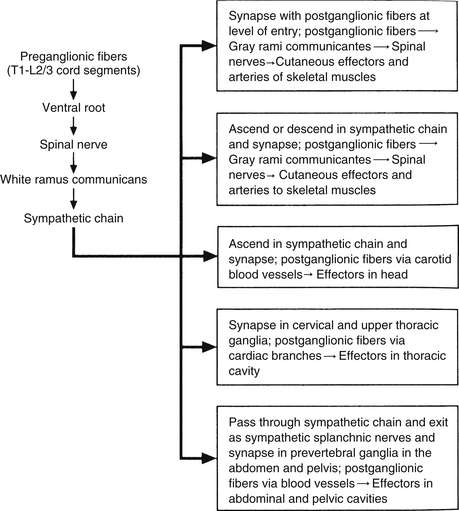
Autonomic fibers innervating peripheral blood vessels (including those in the skeletal muscles and skin), sweat glands, and arrector pili muscles of hair follicles travel in spinal nerves and subsequently peripheral nerves to innervate the appropriate effectors. These effectors are located in the area of distribution of each of the peripheral nerves. After entering the sympathetic trunk, preganglionic fibers associated with these effectors do one of three things (Fig. 10-6): ascend to synapse with postganglionic neurons in ganglia above T1 (for cervical nerves); synapse with postganglionic neurons at the level of entry into the trunk (i.e., T1 to L2 or L3 for those corresponding nerves); or descend to synapse with postganglionic neurons in ganglia below L2-3 (for lumbar and sacral nerves). From the sympathetic trunk the postganglionic fibers course through gray (these are unmyelinated fibers) rami communicantes (usually located proximal to the white rami), enter the spinal nerve at the location of its division into dorsal and ventral rami, and continue to the ANS effectors. Therefore the dorsal and ventral rami and subsequently formed peripheral nerves include sensory afferent fibers, motor neurons to skeletal muscle, and postganglionic sympathetic fibers. The ventral roots of T1 to L2-3 cord segments are unique in that they contain motor neurons to skeletal muscle and also preganglionic sympathetic fibers.
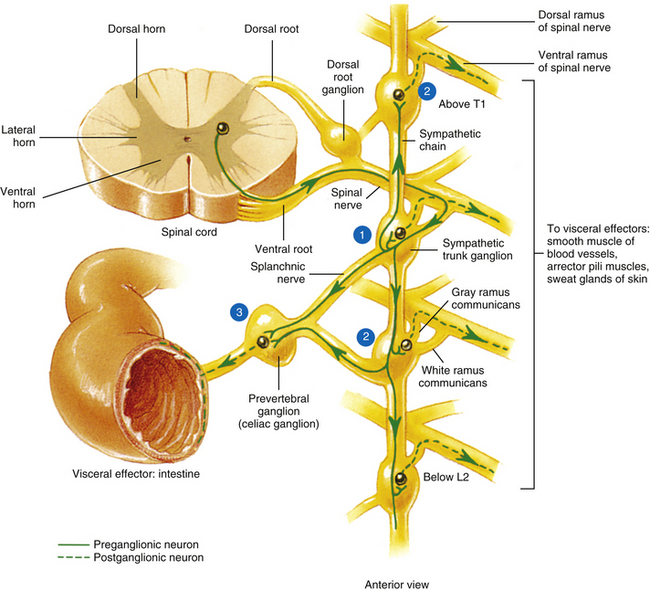
FIG. 10-6 Summary of synapses of sympathetic preganglionic and postganglionic neurons. 1, Preganglionic fibers enter the chain via the white ramus communicans and may synapse at that level, ascend to a more superior ganglion and synapse, descend to a more inferior ganglion and synapse, or pass through the chain. 2, Postganglionic fibers, destined to innervate peripheral effectors, exit the sympathetic chain via the gray ramus communicans (at least one for every spinal nerve) and enter into a ventral (anterior) ramus (they may enter a dorsal [posterior] ramus). 3, Preganglionic fibers, destined to innervate viscera in the abdominal and pelvic cavities, exit the sympathetic chain without synapsing and travel to a prevertebral ganglion and synapse on postganglionic neurons. (From Tortora GJ & Grabowski SR. [2003]. Principles of anatomy and physiology [10th ed.]. New York: John Wiley & Sons, Inc.]
Sympathetic Preganglionic and Postganglionic Fibers
Sympathetic preganglionic fibers sending nerve impulses to effectors in the head enter the sympathetic trunk, ascend to the superior cervical ganglion, and synapse with postganglionic neurons. The postganglionic fibers course with large blood vessels to reach effectors located in the head region (Fig. 10-7, A). Such effectors include glands, the smooth muscle of blood vessels, and the smooth muscle of the eye. Some preganglionic fibers sending impulses to smooth muscle, cardiac muscle, and glands of the thorax also ascend on entering the trunk and synapse at rostral levels, whereas others synapse with postganglionic fibers at the level of entry. These postganglionic fibers leave the chain as branches that merge with other nerve fibers, including parasympathetic vagal fibers, to form plexuses innervating the heart and lungs. Abdominal and pelvic effectors are innervated in a different manner than the effectors of the head, thorax, and cutaneous regions. Preganglionic fibers enter the sympathetic trunk via white rami communicantes but do not synapse in the chain ganglia. Instead they pass through the chain ganglia and emerge as a collection of fibers coursing in splanchnic (referring to the viscera) nerves. These fibers within the splanchnic nerves course inferiorly in an anteromedial direction, pass through the diaphragm, and terminate in various prevertebral ganglia. Here they synapse on postganglionic neurons that then continue to the effectors of the abdominal and pelvic cavities (Fig. 10-7, B). The sympathetic prevertebral ganglia are enmeshed in plexuses of sympathetic and parasympathetic fibers and are located near large arteries found in the abdominal cavity. Examples are the celiac, superior mesenteric, aorticorenal, and inferior mesenteric ganglia.
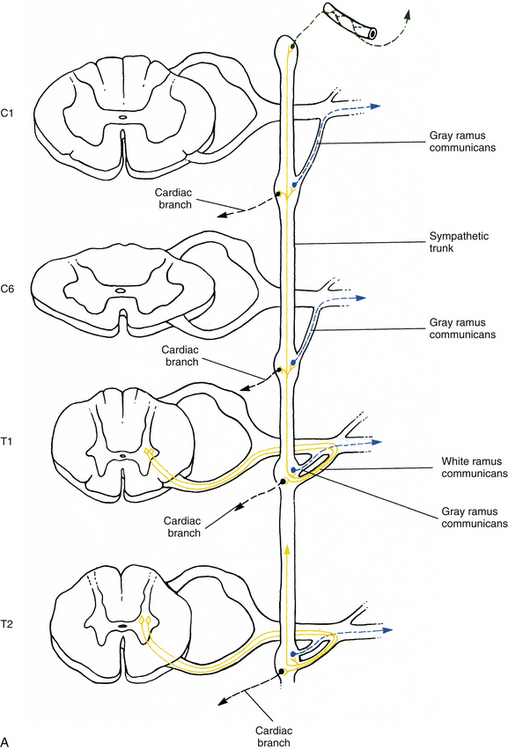
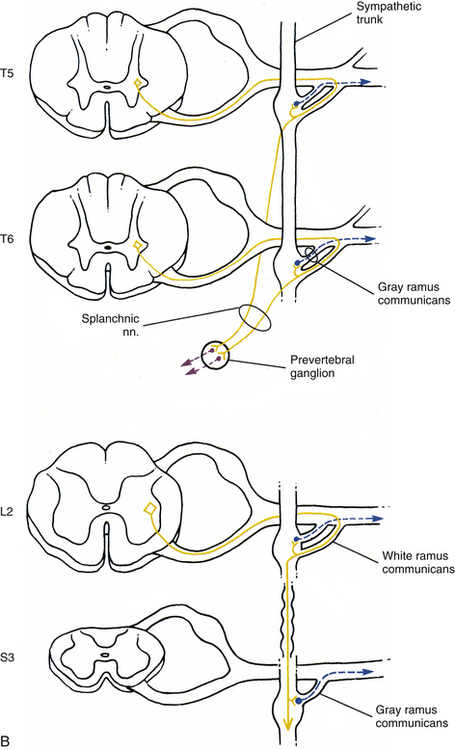
FIG. 10-7 Diagrammatic scheme showing the options of sympathetic neurons. A, This shows that once preganglionic fibers (yellow) have entered the chain, they may ascend to higher levels and synapse with postganglionic fibers that may enter gray rami (blue) or travel on blood vessels (green); synapse in numerous ganglia with postganglionic neurons that leave the chain as cardiac branches (black); or synapse at the level of entry with postganglionic neurons that enter gray rami. B, This shows that once preganglionic fibers (yellow) have entered the chain they may synapse at the level of entry with postganglionic neurons that enter gray rami (blue); descend to lower ganglia and synapse on postganglionic neurons that enter gray rami; or pass through the chain without synapsing and travel to prevertebral ganglia, where they synapse with postganglionic neurons (purple), the axons of which course to effectors in the abdominal and pelvic cavities.
On entering the sympathetic trunk, a typical preganglionic neuron (other than those forming splanchnic nerves) may either ascend or descend and, in each case, subsequently synapse in more than one ganglion. A preganglionic neuron also may synapse at the entry level and send collateral branches up or down to other ganglia. However, less than 2% of the neurons send a branch both up and down (Cabot, 1990). In all cases described thus far, a preganglionic neuron has synapsed with a postganglionic neuron. However, a notable exception is the innervation of the medulla of the adrenal gland which is directly innervated by preganglionic sympathetic neurons. The adrenal medulla develops from the same embryonic neural crest as postganglionic neurons. Although the medullary chromaffin cells do not resemble postganglionic neurons in appearance, they do function in a similar manner. Preganglionic neurons innervate the medulla directly, which in turn releases epinephrine and some norepinephrine into the bloodstream. These neurotransmitters circulate throughout the body, stimulating effectors and assisting in the overall sympathetic response.
A summary of the various sympathetic nerve pathways is provided in Figures 10-6, 10-7, and 10-8.
Specific Regions of the Sympathetic Trunk
Cervical sympathetic trunk: This part of the sympathetic trunk is located anterior to the TPs of the cervical vertebrae, behind and medial to the carotid sheath, which houses the vagus nerve, internal jugular vein, and internal carotid artery. It lies anteriorly on the prevertebral fascia covering the longus capitus and colli muscles. At the level of the C6 vertebra the diameter of the trunk has been measured to be between 1.0 and 4.7 mm with a mean of 2.8 mm (Saylam et al., 2009). A common surgical approach to the cervical spine is from an anterior direction and involves retracting the carotid sheath and/or the longus colli muscle (the longest and most medial of the prevertebral muscles) to expose the lateral structures of the cervical vertebral bodies. Understanding the anatomy of the sympathetic trunk and its ganglia is necessary in order to reduce the risk of injury during these surgical procedures. The fusion of the eight cervical ganglia results in three distinct ganglia in the region of the neck (Figs. 10-9, A, and 10-10; see also Fig. 10-5). These are known as the superior, middle, and cervicothoracic (stellate) ganglia. (Approximately 20% of the time the T1 ganglion is separate, in which case the cervicothoracic ganglion is called the inferior cervical ganglion [Pather et al., 2006].) The superior ganglion (Fig. 10-11, A; see also Figs. 10-9, A, and 10-10) is the largest of the three and lies high in the neck on the TPs of vertebrae C2 and C3, anterior to the longus capitis muscle and posterior to the cervical part of the internal carotid artery. It is also in the vicinity of the internal jugular vein and the glossopharyngeal, vagus, spinal accessory, and hypoglossal cranial nerves (Standring et al., 2008). The proximity of the ganglion to these nerves may account for the autonomic effects seen when these nerves are lesioned in this location (Cross, 1993b). The ganglion is formed by the fusion of the first four cervical ganglia. Its mean length is 3.3 cm and its mean width is 0.8 cm (Saylam et al., 2009), and it includes more than 1 million neurons (Carpenter & Sutin, 1983; Harati, 1993). Postganglionic fibers leaving this ganglion course to various regions. Some ascend as perivascular plexuses on the internal and external carotid arteries. A large branch (internal carotid nerve) from the superior cervical ganglion ascends with the internal carotid artery and divides into branches that form the internal carotid plexus (see Fig. 10-9, A) (Standring et al., 2008). This plexus, which surrounds the artery and innervates its wall, continues to travel with that artery, and within the cranial cavity the fibers innervate the autonomic effectors. Examples of these effectors are the dilator pupillae muscle of the eye, the superior tarsal muscle (Müller’s muscle) of the eyelid, and sweat glands in the medial part of the forehead (Watson & Vijayan, 1995; Salvesen, 2001). In addition, some are sympathetic vasoconstrictor fibers and innervate cerebral branches of the internal carotid artery. Other postganglionic fibers leave the ganglion as medial, lateral, and anterior branches and course directly to effectors. The lateral branches include slender filaments that communicate with the inferior vagal ganglion, hypoglossal nerve, the superior jugular bulb, and jugular glomus of the internal jugular vein, and the meninges of the posterior cranial fossa. One branch, the jugular nerve, branches at the cranial base to join the inferior glossopharyngeal ganglion and the superior vagal ganglion (Standring et al., 2008). Other lateral branches are gray rami that join the first four cervical spinal nerves. The latter travel with those spinal nerves to effectors in the areas of distribution of the nerves. The medial branches include the laryngopharyngeal branches, which supply the carotid body and help form the pharyngeal plexus and cardiac branches. The cardiac branches contain efferent fibers and no nociceptive cardiac afferent fibers. The anterior branches travel with the common and external carotid arteries. The fibers surround the arteries as delicate plexuses within which small ganglia are sometimes found. An external carotid plexus continues with branches of the external carotid artery to innervate such structures as the arrector pili muscles, cutaneous blood vessels, and facial sweat glands not innervated by the internal carotid plexus. These fibers travel with terminal branches of the trigeminal nerve (cranial nerve [CN] V) (Watson & Vijayan, 1995; Salvesen, 2001; Standring, 2008).
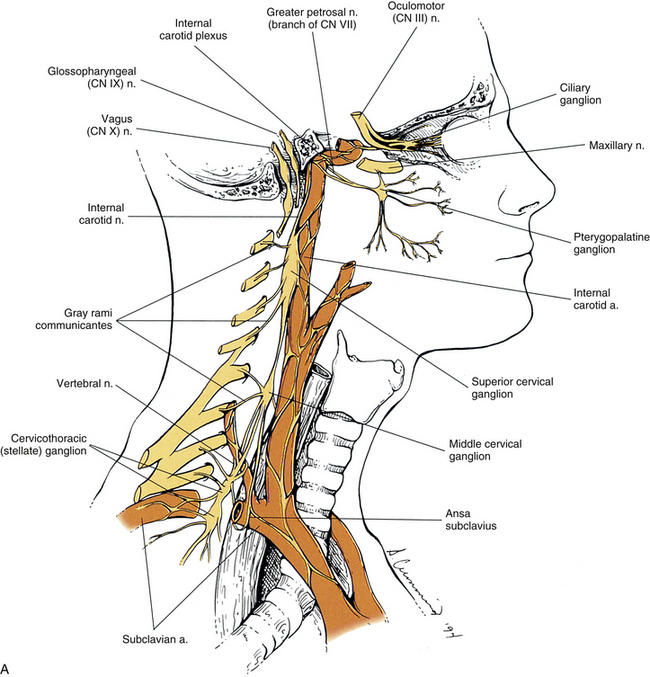
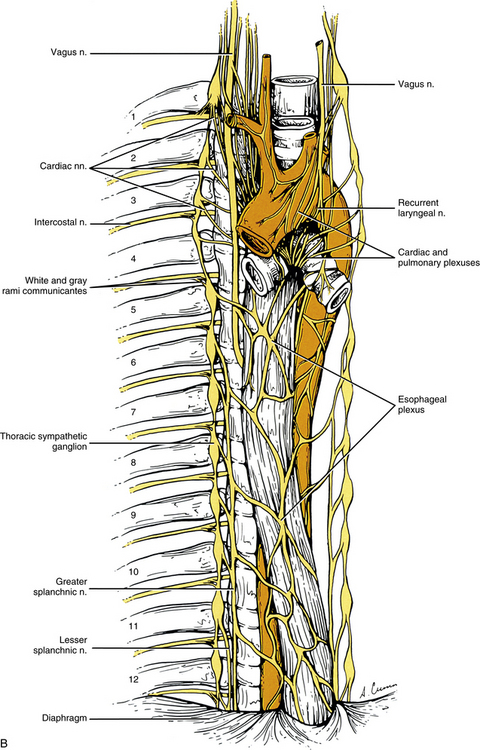
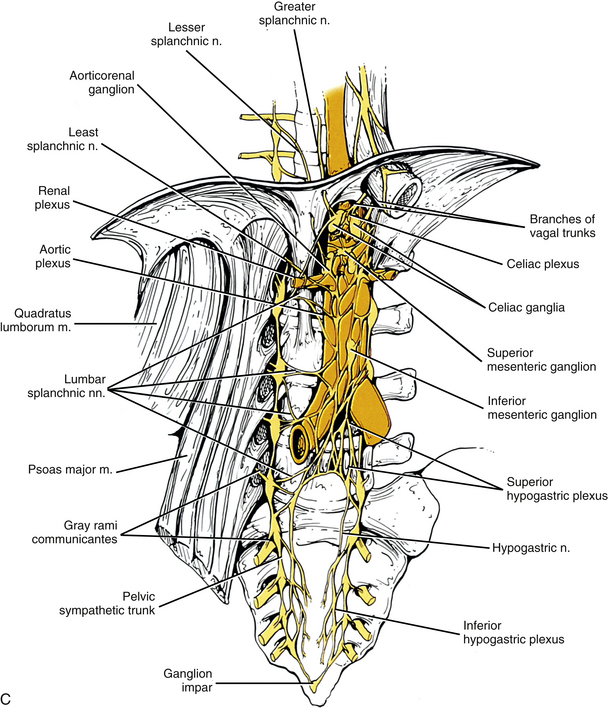
FIG. 10-9 A, Cervical sympathetic trunk and the continuation of autonomic fibers to effectors in the head. Note the relationship of the superior cervical ganglion to the vagus and glossopharyngeal cranial nerves and the internal carotid artery. Gray (not white) rami communicantes course from the cervical chain to the cervical spinal nerves. Leaving the cervicothoracic ganglion are the vertebral nerve and plexus that travel with the vertebral artery. Fibers of some postganglionic neuron cell bodies located in the superior cervical ganglion initially form the internal carotid nerve, which travels with the internal carotid artery, and subsequently branches to form the internal carotid plexus. Fibers of other postganglionic neuron cell bodies located in the superior cervical ganglion course with branches of the external carotid artery to sweat glands (not shown). Note that postganglionic fibers leave the blood vessels and travel with branches of cranial nerves. On the way to their destination, the sympathetic fibers may pass through, but do not synapse in, parasympathetic ganglia (e.g., ciliary and pterygopalatine). B, The thoracic sympathetic trunk shows that gray (medial) and white (lateral) rami communicantes are present. Thoracic autonomic plexuses (e.g., cardiac, pulmonary, and esophageal), which are formed by postganglionic sympathetic fibers and vagal preganglionic fibers, are shown. Cardiac nerves and greater and lesser splanchnic nerves are also illustrated.
C, Lumbar and pelvic sympathetic trunks and autonomic plexuses. The psoas major muscle has been reflected laterally to show the lumbar chain more clearly. The left and right pelvic sympathetic trunks can be seen uniting on the anterior surface of the coccyx to form the ganglion impar. Gray rami communicantes connecting the sympathetic trunk with spinal nerves are present at all levels. Also notice the major autonomic plexuses found in the abdominal and pelvic cavities. Sympathetic prevertebral ganglia located in the abdominal cavity (such as the celiac, superior mesenteric, and inferior mesenteric) also are shown. In the pelvic cavity the superior hypogastric plexus continues as the left and right hypogastric nerves that, with parasympathetic fibers, form the left and right inferior hypogastric (pelvic) plexuses.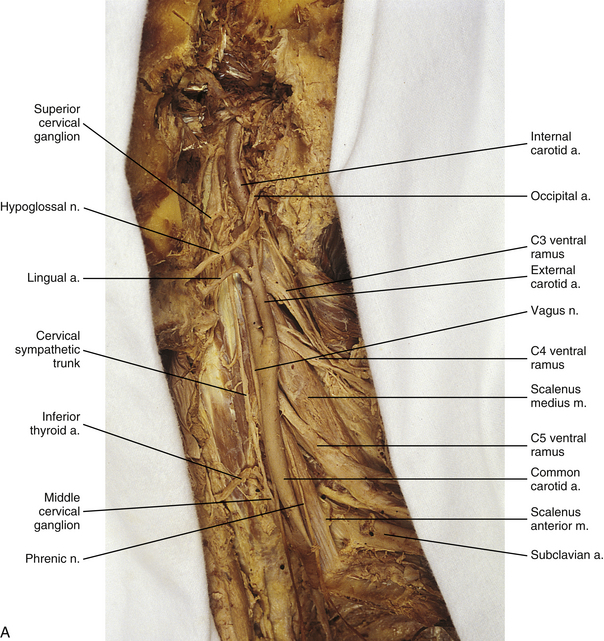
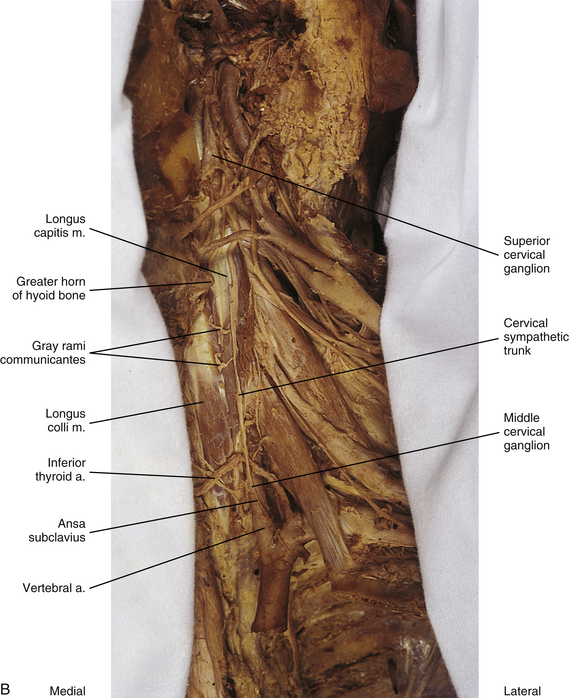
FIG. 10-10 A, Left side of the neck showing the cervical sympathetic trunk. The veins and superior portion of the external carotid artery have been resected. B, The common carotid artery has been reflected laterally to expose the vertebral artery. Notice the relationship of the sympathetic trunk to the longus colli and capitis muscles and note the gray rami communicantes coursing between the two muscles.
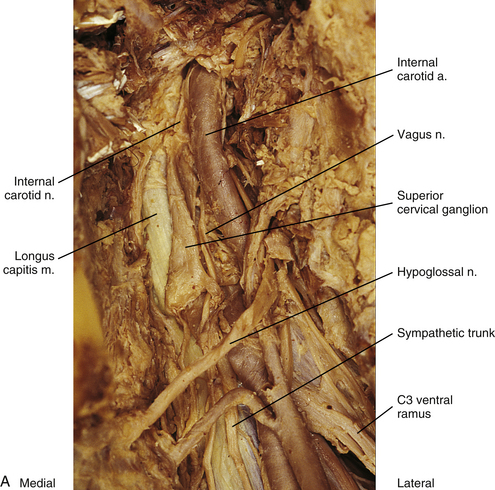
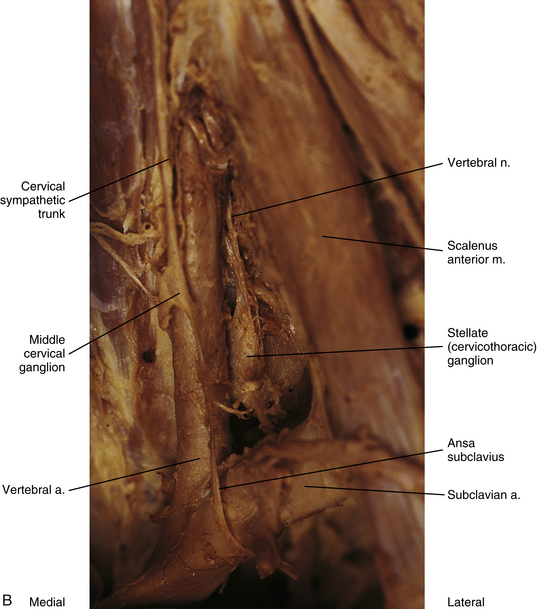
FIG. 10-11 A, Lateral view of the superior aspect of the deep region of the neck near the base of the skull showing the superior cervical ganglion. The internal carotid nerve (postganglionic fibers) is coursing with the internal carotid artery into the carotid canal. B, Stellate ganglion, middle cervical ganglion, and ansa subclavius. The inferior thyroid artery has been resected. The vertebral nerve, which travels with the vertebral artery, courses from the superior aspect of the stellate ganglion.
The middle cervical ganglion (see Figs. 10-9, A, and 10-10) is formed by the fusion of the C5 and C6 ganglia. It is the smallest (≈0.89 cm long and 0.5 cm wide), and often is absent. It lies anterior to the transverse process of the C6 vertebra and near the inferior thyroid artery, which is a branch of the thyrocervical trunk. Measurements show that the ganglion is located 5.95 cm (mean distance) inferior to the superior ganglion and 1.28 cm (mean distance) superior to the cervicothoracic ganglion. In one study, instead of one ganglion, upper and lower ganglia were present 10% of the time (Saylam et al., 2009). Postganglionic fibers include gray rami that enter the C5 and C6 spinal nerves (sometimes the fourth and seventh), thyroid branches (to the thyroid and parathyroid glands), and the largest sympathetic cardiac branch. The cardiac branch often arises from the trunk itself superior or inferior to the ganglion. In addition, slender branches from the ganglion course to the trachea and esophagus (Standring et al., 2008). The ganglion is continuous with the cervicothoracic ganglion by anterior and posterior branches. Although there is variation to this connection, typically the posterior branch splits around the vertebral artery as it descends to the cervicothoracic ganglion; the anterior component descends and loops around the first part of the subclavian artery before connecting with the cervicothoracic ganglion. This loop is called the ansa subclavia (see Fig. 10-11, B).
The cervicothoracic (stellate) ganglion (CTG) (see Figs. 10-9, A, and 10-11, B) is formed by the fusion of the seventh, eighth, and first thoracic ganglia (and sometimes even the second, third, and fourth thoracic ganglia). Most studies indicate the CTG is present 75% to 88% of the time. In a study of 48 cadavers (Pather et al., 2006) in which the cervicothoracic ganglion was present 84% of the time, it was observed that the ganglion was in the shape of either a spindle (28%), a dumbbell (27%), or an inverted “L” (45%). The mean length and width were 16.51 and 6.65 mm, respectively. Another study (Zhang et al., 2009) found the CTG was present 80% of the time and its measurements were about 19.3 mm long and 6.5 mm wide. It is located between the base of the TP of C7 and the neck of the first rib and adjacent to the first intercostal space. It lies on or just lateral to the longus colli muscle and typically behind the vertebral vessels (Saylam et al., 2009) and is in close proximity to the C8 and T1 roots of the brachial plexus (Pather et al., 2006). With the neck fully extended, the ganglion lies two finger’s breadths above the sternoclavicular joint at the level of the transverse process of C7 (Standring et al., 2008). A small ganglion, called the vertebral ganglion, may be present on the sympathetic trunk. It lies anterior or anteromedial to the origin of the vertebral artery and directly above the subclavian artery. The ganglion is thought to be a detached portion of the middle cervical ganglion or stellate/inferior ganglion. Fibers from the ganglion may form or join the ansa subclavia. Other fibers surround the vertebral artery and connect to the stellate ganglion supplying gray rami to the fourth and fifth or the fifth and sixth cervical spinal nerves along the way (Kiray et al., 2005; Standring et al., 2008). The presence of the vertebral ganglion is variable. Saylam and colleagues (2009) found it in only 8% of cadaveric sympathetic trunks whereas Kiray and colleagues (2005) found it in 33% of cadaveric specimens. The ganglion was observed with (12.5%) or without (20.8%) the presence of a middle cervical ganglion (Kiray et al., 2005).
Some postganglionic fibers of the cervicothoracic ganglion travel in gray rami communicantes to enter the C6, C7, C8, and T1 spinal nerves. Song and colleagues (2010) analyzed the morphologic characteristics of these gray rami found in 33 cadavers. The results showed that the gray rami were divided into lateral and medial groups. The lateral group included rami from the CTG to the C8 and T1 nerves. Usually 2 rami coursed to the T1 nerve and only 1 to the C8 nerve although there was some variation, 1-4 and 0-3, respectively. The medial group of rami were associated with the C6, C7, and C8 spinal nerves. The gray ramus joining the C6 nerve root was present on only six sides. The ramus ascended on the posteromedial side of the vertebral artery. Before joining its C6 root, two of the six sides were observed to give off a branch that ascended along the artery to join the C5 nerve root. Medial gray rami connected the CTG with the C8 and C7 nerve roots in 66 and 63 sides, respectively, and in most cases they were shown to give a branch to the adjacent superior nerve root before joining the C8 or C7 root. The gray ramus destined for the C8 nerve root gave off a branch to the C7 nerve root that usually ascended through the C7 transverse foramen. The gray ramus coursing to the C7 root gave off a major branch that then diverged into other small branches. The majority of these ascended through the transverse foramen posteromedially to the vertebral artery to the C6 root and gave off branches to the artery and local joint capsules.
Other postganglionic fibers (83.7%) exit and form a cardiac branch. Most of the time (97.9%) two branches form (an inferior cervical ganglion ramus and a first thoracic ganglion ramus) and contribute to the cardiac plexus located behind the aortic arch (Pather et al., 2006). Some other fibers form plexuses that course on the subclavian artery and its branches, such as the internal thoracic artery, the first part (typically) of the axillary artery, and the vertebral artery. The ascending branch coursing on the vertebral artery is large (see Figs. 10-9, A, and 10-11, B), and frequently called the vertebral nerve (see Chapter 5 and Fig. 5-32). Saylam and colleagues (2009) showed the nerve arising singly from the inferior cervical/cervicothoracic ganglion 85% of the time and arising as two branches, one from that ganglion as well as the vertebral ganglion 15% of the time. In the majority of the specimens (70%) the vertebral nerve was located posterior to the vertebral artery but it also was found traveling anterior, posteromedial, and posterolateral to the artery. Some authors suggest the nerve is essentially a long deep gray ramus from the CTG coursing to the lower cervical spinal nerves within the transverse foramen. Others propose that it looks like a nerve because fibers of cervical gray rami interconnect with each other and with ventral rami of cervical nerves within and between the transverse foramina (Tubbs et al., 2007; Standring et al., 2008; Song et al., 2010). Articular branches, branches to the disc, and branches to the dura come off of the ramus/nerve (Tubbs et al., 2007; Standring et al., 2008). Also, other slender branches intermingle with gray rami communicantes to form the vertebral plexus, which is associated with the vertebral artery (see Cervical Sympathetics in Chapter 5 for further details on the vertebral artery sympathetic nerve plexus). Deep rami communicantes branch from the vertebral plexus and travel with ventral rami of the first five or six cervical spinal nerves. In addition to sympathetic (efferent) fibers, the plexus also contains cervical somatic afferent fibers that innervate the adventitia of the vertebral arterial wall. The plexus travels into the cranial cavity on the vertebral artery and continues on the basilar artery (and its branches) as far as the posterior cerebral artery, where it continues anteriorly to join the internal carotid artery plexus (Standring et al., 2008). The vertebral plexus may be the major continuation of the sympathetic system into the cranium.
Because of its location in the sympathetic chain and the fact that its fibers course to upper extremity and thoracic cavity effectors, lesions of the cervicothoracic ganglion can affect the sympathetic innervation of the head, neck, upper extremity, and thoracic viscera. Having knowledge of the anatomy of this ganglion as well as the anatomy of its gray rami is important (see Kiray et al., 2005; Saylam et al., 2009; Song et al., 2010; for specific morphometric dimensions). Thoracoscopic sympathectomy procedures are commonly used for sympathetically mediated syndromes affecting the upper extremities such as complex regional pain syndrome (CRPS) and hyperhidrosis and also for refractory anginal pain. In some cases the cervicothoracic ganglion needs to be preserved and in other situations the ganglion is selectively blocked. Care must be taken when performing procedures in this area; if the ganglion is injured during thoracic sympathectomy, Horner’s syndrome may result (Pather et al., 2006) (see Clinical Applications section).
Although the cervical sympathetic chain has no white rami communicantes associated with it, numerous gray rami are associated with each spinal nerve. Also, cervical gray rami may pierce the longus capitis and scalenus anterior muscles as they course to the cervical spinal nerves (Standring et al., 2008). Pathological conditions in cervical vertebrae and cervical soft tissue may affect gray rami associated with cervical spinal nerves and the brachial plexus (Song et al., 2010).
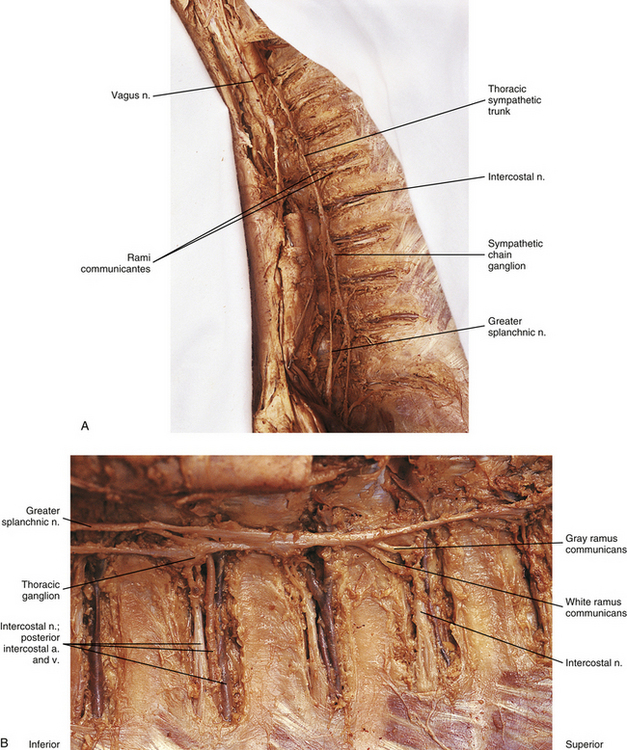
Thoracic sympathetic trunk: Eleven small ganglia usually (70% of the time) are found in the thoracic sympathetic chain (Fig. 10-12; see also Fig. 10-9, B, Figs. 6-11 to 6-13). (Note that approximately 80% of the time the T1 ganglion is fused with the inferior cervical ganglion, in which case the succeeding ganglion is still named the second [T2].) Each ganglion includes 90,000 to 100,000 neurons (Harati, 1993). The thoracic chain lies adjacent to the heads of the ribs and posterior to the costal pleura. In this region of the chain, white rami communicantes, as well as the gray rami communicantes, are clearly evident (see Fig. 10-12, B). The white rami lie more distal (lateral) than the gray rami, and two or more rami may be connected to one spinal nerve. A mixed ramus formed by the fusion of the gray and white rami sometimes may be present. The anatomy of the upper sympathetic thoracic trunk and any variations are clinically important because the trunk is often the site for surgical procedures (sympathectomy, sympathicotomy, clipping of the sympathetic chain, and ramicotomy) as treatments for pathologies such as essential palmar hyperhidrosis, complex regional pain syndrome (CRPS), and Raynaud’s phenomenon. Because of the importance of this region, Cho and colleagues (2005) and Zhang and colleagues (2009) performed cadaveric studies on the anatomy of the upper thoracic trunk. Zhang and colleagues (2009) found that rather than being located anterior to the proximal aspect of the ribs the T2, T3, and T4 ganglia were located in the intercostal spaces 92%, 68%, and 50% of the time, respectively, and that as the trunk descended the ganglia were more likely to be found either at the upper border or on the surface of the lower rib. Additional rami communicantes were also noted and described as follows: intercostal rami that connected adjacent intercostal nerves or ventral rami; ascending rami that connected the sympathetic ganglion with the intercostal nerve or ventral rami at the level above; descending rami that connected the ganglion to the intercostal nerve at the level below. The first intercostal ramus ascending from the second ganglion (nerve of Kuntz) was present 40% of the time but only bilaterally 16% of the time. The only other ramus found was the second intercostal ramus, which was seen in only 6% of the specimens. Ascending rami coursing from the T2, T3, and T4 ganglia were present 40%, 16%, and 6% (Zhang et al., 2009) and 54%, 6%, and 5% (Cho et al., 2005) of the time, respectively, and descending rami coursing from these ganglia were present 30%, 10%, and 8% (Zhang et al., 2009) and 46%, 25%, and 8% (Cho et al., 2005) of the time, respectively. It was infrequent that a ganglion would have both ascending and descending branches. The additional rami communicantes and their relationship with intercostal nerves may act as a conduit for sympathetic fibers to bypass the chain and may be an explanation as to why surgeries on the sympathetic trunk or rami sometimes fail and symptoms reoccur. Another study looked at the relationship of the sympathetic trunk and the intercostal veins at the level of the third and fourth intercostal spaces. The size of the vein and whether the vein crossed anteriorly or posteriorly to the trunk were analyzed. The results showed that large veins (defined as being more than half the width of the intercostal space), which are susceptible to bleeding, and veins crossing anteriorly to the trunk were found more often on the right side than the left. Surgical procedures performed in these intercostal spaces on the right side would present a greater risk for bleeding to occur (Haam et al., 2010). Surgeons should be aware of these variations when upper limb sympathectomies are performed. Postganglionic fibers originating from all thoracic ganglia course in gray rami and enter the thoracic spinal nerves and travel with them to effectors. Some postganglionic fibers from the T1 to T5 ganglia form direct branches to the thoracic aortic, cardiac, and pulmonary plexuses of the thorax. Other large branches of the T5 to T12 ganglia supply the aorta and are associated with the three splanchnic nerves involved with the sympathetic innervation of the abdominal and pelvic viscera. These splanchnic nerves are mixed nerves in that they consist of preganglionic fibers that synapse in prevertebral ganglia located in the abdominal cavity and visceral afferent fibers (see section on Visceral Afferents).
The greater splanchnic nerve (see Figs. 10-9, B and 10-12, A) contains preganglionic fibers exiting medially from the T5 to T9 or T10 ganglia, although there is some variation (see below). As it descends obliquely on the vertebral bodies, it sends branches to the descending thoracic aorta and then pierces the crus of the diaphragm. The main trunk enters the celiac ganglion whereas other fibers course to the medulla of the adrenal gland and sometimes the aorticorenal ganglion. In the ganglia, the preganglionic fibers of the greater splanchnic nerve synapse on postganglionic neurons and interneurons (Standring et al., 2008). The lesser splanchnic nerve consists of preganglionic fibers from the T9 and T10 or T10 and T11 ganglia (see below) and is present 94% of the time. It traverses the diaphragm and enters the abdominal cavity to synapse in the aorticorenal ganglion (the detached lower part of the celiac ganglion) and may supply the lateral side of the celiac ganglion. The third splanchnic nerve is the lowest or least splanchnic nerve and is present 56% of the time. Sometimes this nerve is called the renal nerve. It emerges medially from the T12 (or lowest) ganglion although this is variable (see below) and travels with the sympathetic chain under the medial arcuate ligament of the diaphragm and inferiorly to enter the renal plexus. The majority of the fibers enter the aorticorenal ganglion and may also supply the lateral side of the celiac ganglion. Sometimes it may appear as a part of the lesser splanchnic nerve connecting the aorticorenal ganglion to the renal plexus (Harati, 1993; Standring et al., 2008). From these prevertebral ganglia, postganglionic fibers participate in the formation of the various perivascular plexuses as they travel to abdominal effectors. Because of the clinical relevancy of the splanchnic nerves (see below), Gest and Hildebrandt (2009) conducted a cadaveric study on the splanchnic nerves and their course through the diaphragm. All 3 splanchnic nerves were found 57% (25 of 44 sides) of the time; the least splanchnic was missing in 43% of the sympathetic chains whereas the greater and lesser nerves were always present. Of the 6 different patterns by which the nerves passed through the crura of the diaphragm, the 2 most common were all 3 passing through a hiatus together (16 of 44) and the greater and lesser nerves passing through together (18 of 44). In 3 specimens the greater nerve traveled through alone and the lesser and least nerves ran together, and in 4 of 44 specimens the greater and lesser nerves coursed together and the least nerve passed through alone. In only 3 of 43 specimens did the nerves traverse the diaphragm separately; 65% of the time the pattern was symmetric between the right and left sides. In only two specimens did the least nerve pass through the diaphragm with the sympathetic trunk on both sides. It was also noted in 17 of 35 specimens that the angle of the subdiaphragmatic portion of the greater splanchnic nerve coursing to the celiac ganglion was 90 degrees or greater relative to the intrathoracic portion of the nerve.
Thoracic splanchnic nerves (and the celiac ganglion) are very important from a clinical standpoint because visceral afferent fibers conveying nociception from upper abdominal organs course within these nerves. Splanchnicectomy is a surgical procedure that has been used to treat a number of pathologies such as chronic spastic constipation, chronic intestinal pseudoobstruction, and hypertension. The procedure is also performed to manage intractable abdominal pain related to pancreatic cancer and chronic pancreatitis. Patients with chronic pancreatitis complain of severe intermittent or long-lasting and persistent pain caused likely by damage to intrapancreatic nerves. Patients with pancreatic cancer, although presenting initially with weight loss and jaundice, experience pain thought to be caused by the infiltration of cancer cells into the perineurium of local nerves and extrapancreatic nerves such as those related to the celiac and/or mesenteric plexuses. It appears therefore that pancreatic neuropathy may be the stimulus for the intense pain experienced by patients with both of these conditions. The splanchnicectomy may be performed at the intrathoracic level or at the subdiaphragmatic, retroperitoneal level. Variations of the procedure include dissection of the nerves or transection of the main trunks of the nerve, thermocoagulation, division or transection of the nerve branches, and excision of small sections of the nerve (Loukas et al., 2010). There have been numerous anatomic studies (reviewed by Loukas and colleagues [2010]) indicating (1) variations in the levels and ganglia contributing to the formation of each of the thoracic splanchnic nerves, (2) the absence of a contributing root from a ganglion to the nerve, (3) variations in the frequency of the lesser and least nerves being present, (4) a lack of symmetry between right and left sides in an individual, (5) alternate neural pathways (e.g., thin fibers that interconnect the greater and lesser nerves or the greater and least nerves, and the presence of a fourth accessory splanchnic nerve), and (6) anatomic variations in the distal subdiaphragmatic segments of the nerves (Gest & Hildebrandt, 2009). It is clear that an understanding of the detailed anatomy of these structures and their variations between right and left sides and among patients is essential to have successful outcomes from surgical procedures.
Lumbar sympathetic trunk: The thoracic sympathetic trunk passes posterior to the medial arcuate ligament (or sometimes through the crura of the diaphragm) to become continuous with the lumbar sympathetic trunk found within the abdominal cavity. The trunk has been described as consisting of 4 interconnected lumbar ganglia (each of which contains 60,000 to 85,000 neurons) (Harati, 1993; Standring et al., 2008). However, other data indicate that the number of ganglia varies (Mitchell, 1953; Rocco, Palombi, & Raeke, 1995). Murata and colleagues (2003) studied cadaveric specimens and looked at the anatomic variations of the ganglia and associated rami. They found that the number of ganglia on one side ranged from 2 to 6 (mean, 3.9) and that the majority of ganglia were located on the L2 and L3 vertebrae. Typically no ganglia were found on the L1 and L4 vertebrae. Approximately 40% of the cadavers showed the same number of ganglia on both sides, and those were asymmetrically located. In addition, Murata and colleagues (2003) found 5 to 12 rami communicantes per side (mean of 7.2); in addition, they noted that more than 1 ramus communicans could connect to a lumbar ventral ramus and that a lumbar ventral ramus often received rami communicantes from more than 1 ganglion. In fact, one third of the ganglia associated with the L2 and L3 vertebrae included rami that traveled to three spinal nerves. Rami associated with the L1-5 spinal nerves were measured, and it was found that the ramus of the L4 spinal nerve was significantly similar in length to the L2 and L3 rami. Although this is contrary to what would be expected because the sympathetic chain lies closer to the intervertebral foramina (IVFs) at the L4-5 vertebral levels, it may be due to the fact that there are fewer ganglia at the L4 level and the rami have farther to travel to reach a more superiorly located ganglion. The L1 rami also were significantly longer than the other lumbar rami, most likely for the same reason. The rami of L5 were significantly shorter than those of L1-4. The presence of anatomic variations in the rami and ganglia in this region may be of importance relative to the nociceptive pathway for lower lumbar structures. Based on studies in rats that may apply to humans as well, it has been suggested that nociceptive input from low back structures is carried in two different pathways. Some nociceptive fibers from low back pain generators travel in a segmental fashion, directly within spinal nerves, and terminate in local cord segments. Other nociceptive fibers terminate centrally in a nonsegmental fashion. These course in lower lumbar spinal nerves, enter the sympathetic chain and ascend, and then exit the chain through more superiorly located rami connected to the L2 spinal nerve. These fibers terminate in the lower region of the thoracolumbar cord segments associated with the sympathetic division (Murata et al., 2003). If this pathway is present in humans it may contribute to the wide referral pattern seen in lower lumbar intervertebral discs, lower lumbar zygapophysial (Z) joints, and sacroiliac joint pain conditions (Murata et al., 2000) (see Chapters 7 and 11 for further information).
The lumbar trunk lies adjacent to the anterolateral aspect of the upper lumbar vertebrae and becomes more posterior relative to lower lumbar vertebrae (Murata et al., 2003). It also lies adjacent to the medial margin of the psoas major muscle (Fig. 10-13; see also Fig. 10-9, C). The inferior vena cava, right ureter, and lumbar lymph nodes lie anterior to the right sympathetic trunk. The left sympathetic trunk lies posterior to the aortic lymph nodes and lateral to the aorta. These relationships are important surgically because lumbar ganglia may have to be removed (lumbar sympathectomy; see Innervation of Peripheral Effectors) to treat certain arterial diseases of the lower extremities (Snell, 2008; Standring et al., 2008).
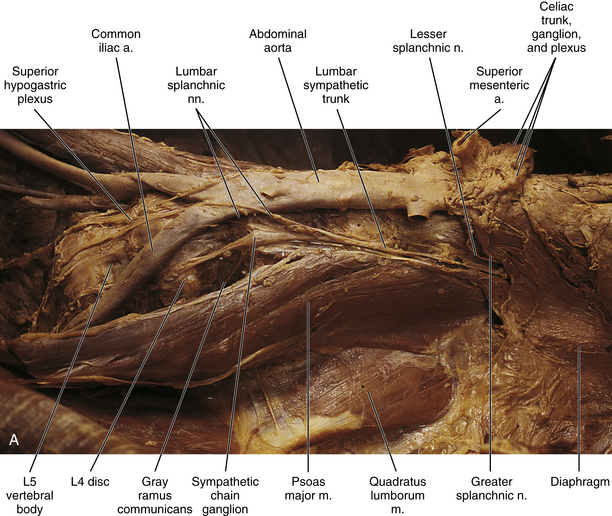
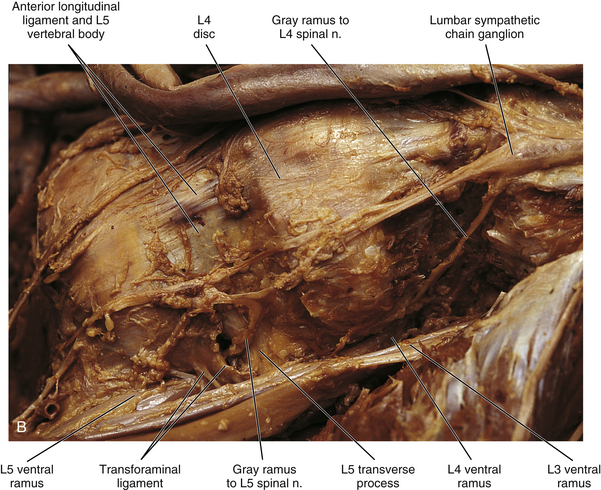
FIG. 10-13 Left lumbar sympathetic trunk. A, Notice the relationship of the sympathetic trunk with the psoas major muscle and vertebral bodies and discs. Lumbar splanchnic nerves are coursing from the sympathetic trunk to the superior hypogastric plexus. The greater and lesser splanchnic nerves pass through the diaphragm to synapse in the celiac ganglion and aorticorenal ganglion. The celiac trunk, superior and inferior mesenteric, and renal arteries have been resected. The inferior vena cava also has been resected. B, Lumbar sympathetic trunk at the level of the L4 and L5 vertebrae. The left common iliac artery has been reflected. The psoas major muscle also has been reflected. Notice the long gray rami communicantes. A transforaminal ligament spanning the intervertebral foramen is present in this specimen. Note the relationship of the L5 ventral ramus and gray ramus communicans to this ligament.
White rami communicantes are associated with the upper two or three ganglia. Some preganglionic fibers descend in the chain to synapse in any lumbar ganglia below the L2 or L3 ganglia and in pelvic ganglia (see below). The gray rami are long (see previous section) and they course with lumbar arteries along the sides of the vertebral bodies to join each lumbar spinal nerve (see Fig. 10-13). The majority of these postganglionic fibers are thought to use the femoral nerve, obturator nerve, and their muscular and cutaneous branches to supply vasoconstrictor fibers to the adjoining blood vessels and fibers to cutaneous effectors. In a manner similar to the lower thoracic ganglia, some preganglionic fibers pass through the lumbar ganglia to form lumbar splanchnic nerves, which will also contain visceral afferent fibers. In general, each lumbar splanchnic nerve corresponds to its ganglion of the same number, although the second lumbar splanchnic nerve receives additional fibers from the third ganglion and the third lumbar splanchnic nerve also receives a contribution from the fourth ganglion. The four splanchnic nerves course into the abdomen and become part of the abdominal plexuses: the first splanchnic nerve courses within the celiac, abdominal aortic (intermesenteric), inferior mesenteric, and renal plexuses; the second splanchnic nerve contributes to the inferior part of the abdominal aortic (intermesenteric) plexus or inferior mesenteric plexus; the third splanchnic nerve passes anterior to the common iliac vessels and travels within the superior hypogastric plexus (see Figs. 10-9, C, and 10-13); the fourth splanchnic nerve also courses anterior to the common iliac vessels and contributes to the lowest portion of the superior hypogastric plexus (or hypogastric nerve). All lumbar ganglia give off vascular branches that enter the abdominal aortic plexus. Lower lumbar splanchnic nerves give off branches that run on the common iliac arteries and continue as a network that extends onto the internal and external iliac arteries (Standring et al., 2008). The lumbar portion of the sympathetic trunk passes inferiorly, posterior to the common iliac vessels, and becomes continuous with the pelvic portion of the trunk.
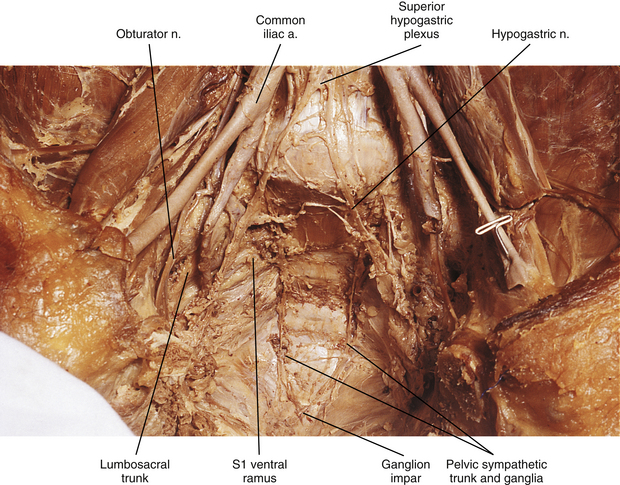
Pelvic sympathetic trunk: The pelvic chain consists of four or five ganglia that lie in extraperitoneal tissue on the anterior aspect of the sacrum beneath presacral fascia. Within the chain are descending preganglionic fibers from the lower thoracic and the upper two or three lumbar cord segments that will synapse in pelvic ganglia. Each side unites to form the ganglion impar on the anterior aspect of the coccyx (Fig. 10-14; see also Fig. 10-9, C). Postganglionic fibers leave the chain in gray rami to enter the sacral spinal nerves and coccygeal nerve. Fibers destined for blood vessels in the leg and foot course primarily with the tibial nerve to connect subsequently with (and supply) the popliteal artery and its branches in the leg and foot. Other fibers travel with the pudendal and gluteal nerves to the internal pudendal artery and gluteal arteries and their branches. In addition, some medial fibers from the first two sacral ganglia referred to as the sacral splanchnic nerves travel within the inferior hypogastric plexus (or hypogastric nerve) to the pelvic viscera as a delicate network of pelvic nerves (the pelvic plexus) (Standring et al., 2008).
Plexuses of the Autonomic Nervous System
The cardiac, pulmonary, celiac, and hypogastric plexuses are the major plexuses (Standring et al., 2008), although secondary plexuses may emanate from each one. The cardiac plexus, which is divided into deep and superficial parts, consists of cardiac branches from cervical and upper thoracic ganglia mixed with cardiac branches of the vagus nerve (see Fig. 10-9, B). A continuation of the cardiac plexus forms secondary coronary and atrial plexuses. The pulmonary plexus is an extension of fibers of the cardiac plexus that course with the pulmonary arteries to the lungs. Therefore the cardiac and pulmonary plexuses consist of the same sympathetic and vagal branches.
The celiac plexus is the largest autonomic plexus (see Fig. 10-9, C). It is located at the level of the T12 and L1 vertebrae and surrounds the celiac artery and the base of the superior mesenteric artery. It is posterior to the stomach and lesser omentum and anterior to the crura of the diaphragm and the beginning of the abdominal aorta. The adrenal glands lie on either side of the plexus. This plexus is a dense fibrous network that interconnects the paired celiac ganglia. Mingling with the celiac plexus and ganglia are the greater and lesser splanchnic nerves and also branches of the vagus and phrenic nerves (Standring et al., 2008). Numerous subsidiary ganglia and fibers extend from the celiac plexus and course along abdominal blood vessels to autonomic effectors. These fibers and ganglia form (in some cases with the help of the lesser and least splanchnic nerves) plexuses that include the phrenic, hepatic, gastric, splenic, testicular, ovarian, superior mesenteric (to small and large intestines), renal, and suprarenal. As one can see, these ganglia and plexuses are responsible for the innervation of the abdominal viscera. The celiac ganglia are irregular masses located on each side of the celiac trunk medial to the adrenal glands and anterior to the crura of the diaphragm. Both are related to other blood vessels in the region (i.e., the left lies posterior to the origin of the splenic artery and the right lies posterior to the inferior vena cava). As observed in other structures related to the sympathetic system, there is much variation in the location of the ganglia (usually at the level of T12 or L1) and in their morphology. Because a celiac plexus block is a surgical procedure used to manage pancreatic pain in conjunction with or as an alternative to splanchnicectomy, understanding the normal anatomy and any variations in this region is essential in order to have positive surgical outcomes (Loukas et al., 2010). The greater splanchnic nerve enters the upper part of each ganglion and the lesser splanchnic nerve ends in the lower part. A portion of the lower part of each ganglion forms a distinct region called the aorticorenal ganglion, which also receives fibers from the lesser splanchnic nerve (Loukas et al., 2010
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree