Nausea and Vomiting
KEY CONCEPTS
![]() Nausea and/or vomiting is often a part of the symptom complex for a variety of GI, cardiovascular, infectious, neurologic, metabolic, or psychogenic processes.
Nausea and/or vomiting is often a part of the symptom complex for a variety of GI, cardiovascular, infectious, neurologic, metabolic, or psychogenic processes.
![]() Nausea or vomiting is caused by a variety of medications or other noxious agents.
Nausea or vomiting is caused by a variety of medications or other noxious agents.
![]() The overall goal of treatment should be to prevent or eliminate nausea and vomiting regardless of etiology.
The overall goal of treatment should be to prevent or eliminate nausea and vomiting regardless of etiology.
![]() Treatment options for nausea and vomiting include drug and nondrug modalities such as relaxation, biofeedback, and self-hypnosis.
Treatment options for nausea and vomiting include drug and nondrug modalities such as relaxation, biofeedback, and self-hypnosis.
![]() The primary goal with chemotherapy-induced nausea and vomiting (CINV) is to prevent nausea and/or vomiting. Optimal control of acute nausea and vomiting positively impacts the incidence and control of delayed and anticipatory nausea and vomiting.
The primary goal with chemotherapy-induced nausea and vomiting (CINV) is to prevent nausea and/or vomiting. Optimal control of acute nausea and vomiting positively impacts the incidence and control of delayed and anticipatory nausea and vomiting.
![]() The emetic risk of the chemotherapeutic regimen is the primary factor to consider when selecting prophylactic antiemetics for CINV.
The emetic risk of the chemotherapeutic regimen is the primary factor to consider when selecting prophylactic antiemetics for CINV.
![]() Patients at high risk of vomiting should receive prophylactic antiemetics for postoperative nausea and vomiting.
Patients at high risk of vomiting should receive prophylactic antiemetics for postoperative nausea and vomiting.
![]() Patients undergoing radiation therapy to the upper abdomen or receiving total or hemibody irradiation should receive prophylactic antiemetics for radiation-induced nausea and vomiting.
Patients undergoing radiation therapy to the upper abdomen or receiving total or hemibody irradiation should receive prophylactic antiemetics for radiation-induced nausea and vomiting.
Nausea and vomiting are common complaints from individuals of all ages. Management can be quite simple or detailed and complex, essentially innocuous or associated with therapy-induced adverse reactions. This chapter provides an overview of nausea and vomiting, two multifaceted problems.
Nausea is defined as the inclination to vomit or as a feeling in the throat or epigastric region alerting an individual that vomiting is imminent. Vomiting is defined as the ejection or expulsion of gastric contents through the mouth and is often a forceful event. Either condition may occur transiently with no other associated signs or symptoms; however, these conditions also may be only part of a more complex clinical presentation.
ETIOLOGY
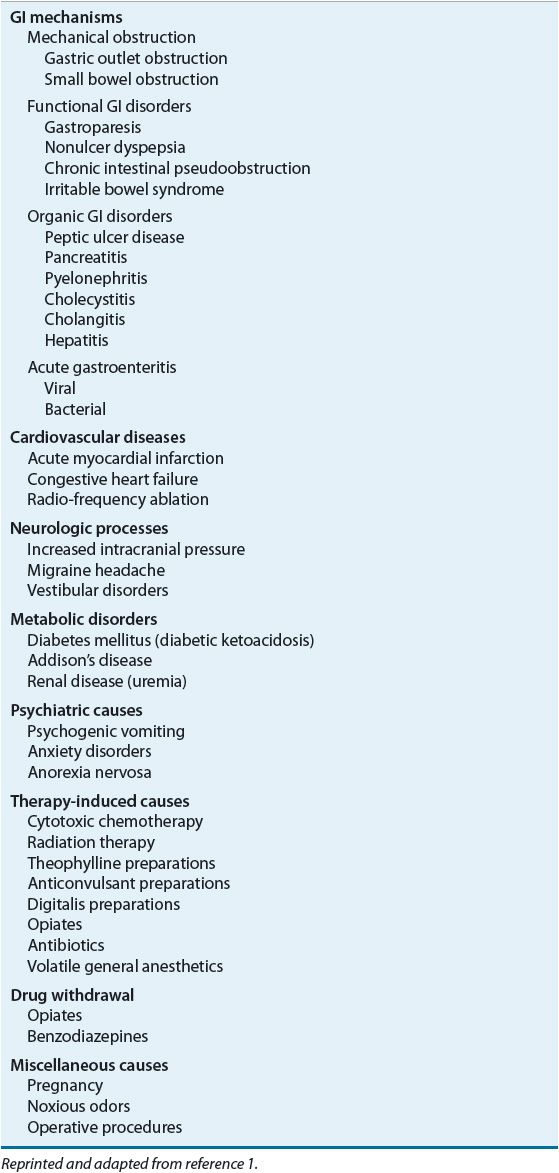
![]() Nausea and vomiting may be associated with a variety of conditions, including GI, cardiovascular, infectious, neurologic, or metabolic disease processes. Nausea and vomiting may be a feature of such conditions as pregnancy, or may follow operative procedures or administration of certain medications, such as those used in cancer chemotherapy. Psychogenic etiologies of these symptoms may be present. Anticipatory etiologies may be involved, such as in patients who have previously received cytotoxic chemotherapy. Table 22-1 lists specific etiologies associated with nausea and vomiting.1
Nausea and vomiting may be associated with a variety of conditions, including GI, cardiovascular, infectious, neurologic, or metabolic disease processes. Nausea and vomiting may be a feature of such conditions as pregnancy, or may follow operative procedures or administration of certain medications, such as those used in cancer chemotherapy. Psychogenic etiologies of these symptoms may be present. Anticipatory etiologies may be involved, such as in patients who have previously received cytotoxic chemotherapy. Table 22-1 lists specific etiologies associated with nausea and vomiting.1
TABLE 22-1 Specific Etiologies of Nausea and Vomiting
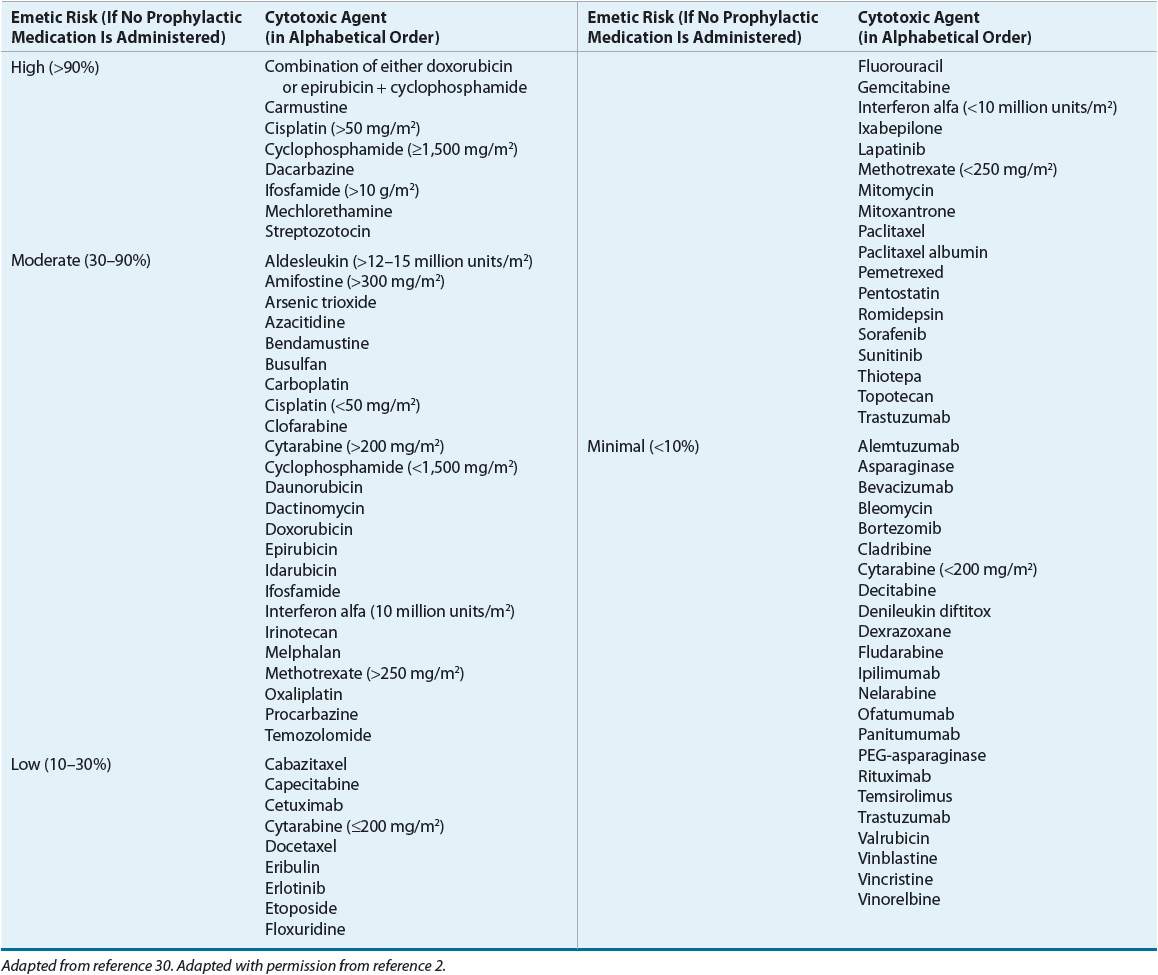
The etiology of nausea and vomiting may vary with the age of the patient. For example, vomiting in the newborn during the first day of life suggests upper digestive tract obstruction or an increase in intracranial pressure. ![]() Drug-induced nausea and vomiting are of particular concern, especially with the increasing number of patients receiving cytotoxic treatment. A four-level classification system defines the risk for emesis with agents used in oncology (Table 22-2).2 Although some agents may have greater emetic risk than others, combinations of agents, high doses, clinical settings, psychological conditions, prior treatment experiences, and unusual stimulus of sight, smell, or taste may alter a patient’s response to drug treatment. In this setting, nausea and vomiting may be unavoidable and some patients experience these problems so intensely that chemotherapy is postponed or discontinued.
Drug-induced nausea and vomiting are of particular concern, especially with the increasing number of patients receiving cytotoxic treatment. A four-level classification system defines the risk for emesis with agents used in oncology (Table 22-2).2 Although some agents may have greater emetic risk than others, combinations of agents, high doses, clinical settings, psychological conditions, prior treatment experiences, and unusual stimulus of sight, smell, or taste may alter a patient’s response to drug treatment. In this setting, nausea and vomiting may be unavoidable and some patients experience these problems so intensely that chemotherapy is postponed or discontinued.
TABLE 22-2 Emetic Risk of Agents Used in Oncology
PATHOPHYSIOLOGY
The three consecutive phases of emesis include nausea, retching, and vomiting. Nausea, the imminent need to vomit, is associated with gastric stasis and may be considered a separate and singular symptom. Retching is the labored movement of abdominal and thoracic muscles before vomiting. The final phase of emesis is vomiting, the forceful expulsion of gastric contents caused by GI retroperistalsis. The act of vomiting requires the coordinated contractions of the abdominal muscles, pylorus, and antrum, a raised gastric cardia, diminished lower esophageal sphincter pressure, and esophageal dilation.1 Vomiting should not be confused with regurgitation, an act in which the gastric or esophageal contents rise to the pharynx but is not usually associated with forceful ejection seen with vomiting. Accompanying autonomic symptoms of pallor, tachycardia, and diaphoresis account for many of the distressing feelings associated with emesis.
Vomiting is triggered by afferent impulses to the vomiting center, a nucleus of cells in the medulla. Impulses are received from sensory centers, which include the chemoreceptor trigger zone (CTZ), cerebral cortex, and visceral afferents from the pharynx and GI tract. The vomiting center integrates the afferent impulses, resulting in efferent impulses to the salivation center, respiratory center, and the pharyngeal, GI, and abdominal muscles, leading to vomiting.
The CTZ, located in the area postrema of the fourth ventricle of the brain, is a major chemosensory organ for emesis and is usually associated with chemically induced vomiting. Because of its location, bloodborne and cerebrospinal fluid toxins have easy access to the CTZ. Cytotoxic agents primarily stimulate this area rather than the cerebral cortex and visceral afferents. Similarly, pregnancy-associated vomiting probably occurs through stimulation of the CTZ.
Numerous neurotransmitter receptors are located in the vomiting center, CTZ, and GI tract, including cholinergic, histaminic, dopaminergic, opiate, serotonergic, neurokinin (NK), and benzodiazepine receptors. Chemotherapeutic agents, their metabolites, or other emetic compounds theoretically trigger the process of emesis through stimulation of one or more of these receptors. Antiemetics have been developed to antagonize or block these emetogenic receptors.
CLINICAL PRESENTATION
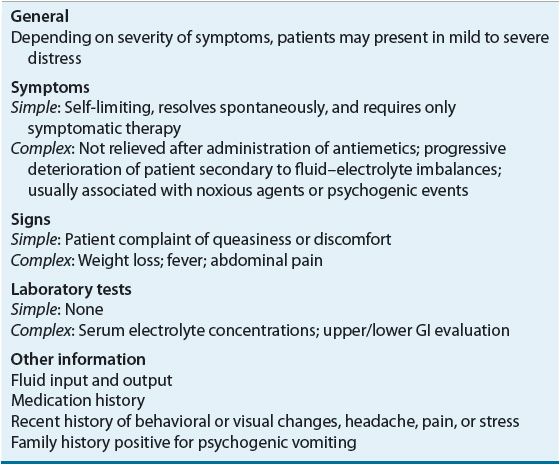
Nausea and vomiting are commonly seen in many clinical situations. Patients may present in varying degrees of distress summarized in Table 22-3 as simple or complex in presentation.
TABLE 22-3 Clinical Presentation of Nausea and Vomiting
TREATMENT
Desired Outcome
![]() The overall goal of antiemetic therapy is to prevent or eliminate nausea and vomiting. This should be accomplished without adverse effects or with clinically acceptable adverse effects. Although this goal may be accomplished easily in patients with simple nausea and vomiting, patients with more complex problems require greater assistance. In addition to these clinical goals, appropriate cost issues should be considered, particularly in the management of chemotherapy-induced nausea and vomiting (CINV) and postoperative nausea and vomiting (PONV).
The overall goal of antiemetic therapy is to prevent or eliminate nausea and vomiting. This should be accomplished without adverse effects or with clinically acceptable adverse effects. Although this goal may be accomplished easily in patients with simple nausea and vomiting, patients with more complex problems require greater assistance. In addition to these clinical goals, appropriate cost issues should be considered, particularly in the management of chemotherapy-induced nausea and vomiting (CINV) and postoperative nausea and vomiting (PONV).
General Approach to Treatment
![]() Treatment options include drug and nondrug modalities. Initially patients may choose to do nothing or to self-medicate with nonprescription drugs. As symptoms become worse or are associated with more serious medical problems, patients are more likely to benefit from prescription antiemetic drugs. When prescribed according to reliable clinical information, these agents often provide acceptable relief; however, some patients will never be totally free of symptoms. This lack of relief is most disabling when it is associated with an unresolved medical problem or when the necessary therapy for this condition is the cause of the nausea or vomiting, as in the case of patients who are receiving chemotherapy of moderate or high emetic risk.
Treatment options include drug and nondrug modalities. Initially patients may choose to do nothing or to self-medicate with nonprescription drugs. As symptoms become worse or are associated with more serious medical problems, patients are more likely to benefit from prescription antiemetic drugs. When prescribed according to reliable clinical information, these agents often provide acceptable relief; however, some patients will never be totally free of symptoms. This lack of relief is most disabling when it is associated with an unresolved medical problem or when the necessary therapy for this condition is the cause of the nausea or vomiting, as in the case of patients who are receiving chemotherapy of moderate or high emetic risk.
Nonpharmacologic Management
Nonpharmacologic management of nausea and vomiting involves dietary, physical, or psychological strategies that are consistent with the etiology of nausea and vomiting. For patients with simple complaints, perhaps resulting from excessive or disagreeable food or beverage consumption, avoidance or moderation in dietary intake may be preferable. Patients suffering symptoms of systemic illness may improve dramatically as their underlying condition resolves. Finally, patients in whom these symptoms result from labyrinthine changes produced by motion may benefit quickly by assuming a stable physical position.
Nonpharmacologic interventions are classified as behavioral interventions and include relaxation, biofeedback, self-hypnosis, cognitive distraction, guided imagery, acupuncture, and systematic desensitization.3,4 The reader is referred to references 5 to 7 for a more complete discussion on nonpharmacologic strategies.
Pharmacologic Therapy
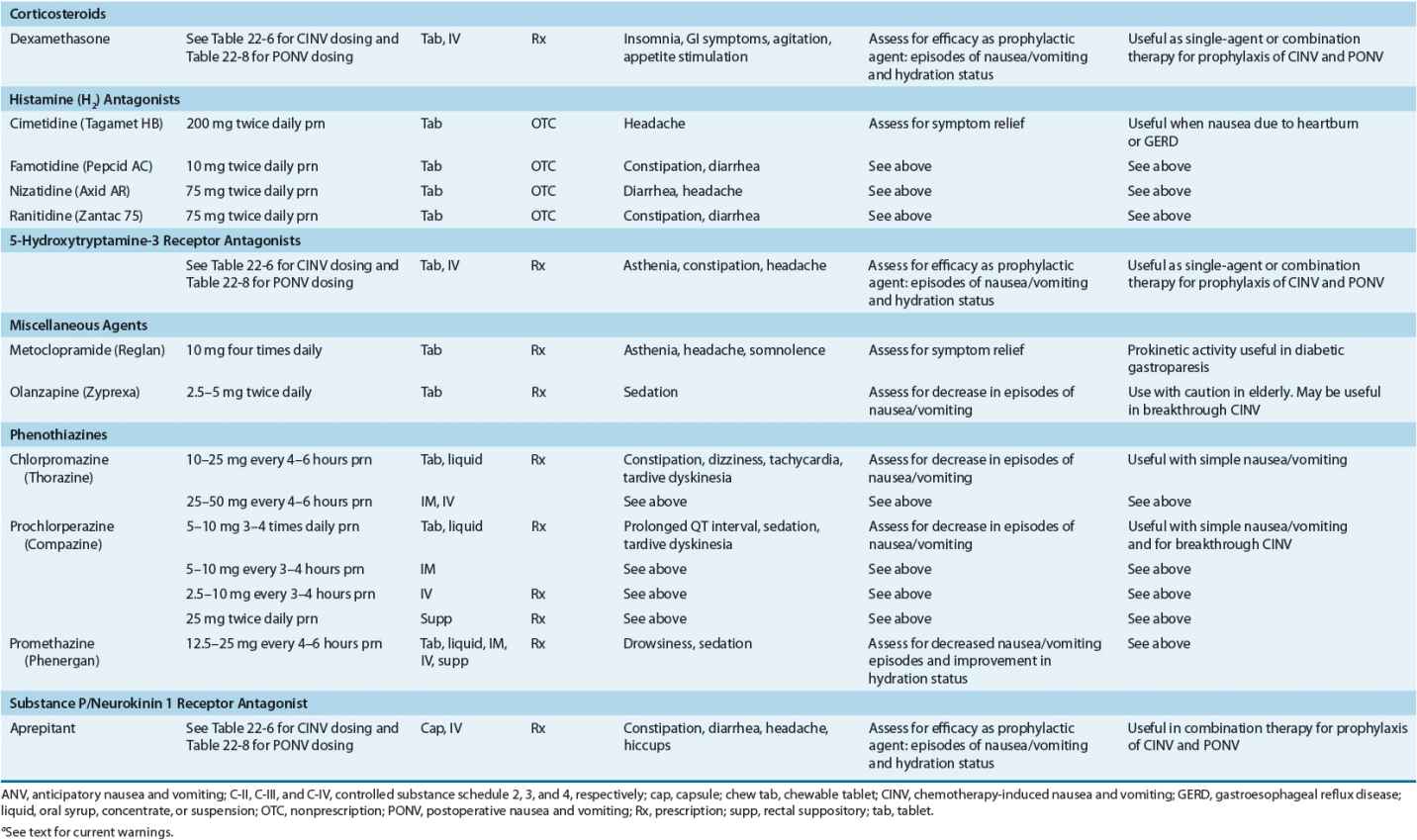
![]() Although many approaches to the treatment of nausea and vomiting have been suggested, antiemetic drugs (nonprescription and prescription) are most often recommended. These agents represent a variety of pharmacologic and chemical classes, as well as dosage regimens and routes of administration. With so many treatment possibilities available, factors that enable the clinician to discriminate among various choices include (a) the suspected etiology of the symptoms; (b) the frequency, duration, and severity of the episodes; (c) the ability of the patient to use oral, rectal, injectable, or transdermal medications; and (d) the success of previous antiemetic medications. Please see Table 22-4 for dosing information of commonly available antiemetic preparations.
Although many approaches to the treatment of nausea and vomiting have been suggested, antiemetic drugs (nonprescription and prescription) are most often recommended. These agents represent a variety of pharmacologic and chemical classes, as well as dosage regimens and routes of administration. With so many treatment possibilities available, factors that enable the clinician to discriminate among various choices include (a) the suspected etiology of the symptoms; (b) the frequency, duration, and severity of the episodes; (c) the ability of the patient to use oral, rectal, injectable, or transdermal medications; and (d) the success of previous antiemetic medications. Please see Table 22-4 for dosing information of commonly available antiemetic preparations.
TABLE 22-4 Common Antiemetic Preparations and Adult Dosage Regimens
The treatment of simple nausea and vomiting often involves self-care from a lengthy list of nonprescription products. Both nonprescription and prescription drugs useful in the treatment of simple nausea and vomiting are usually effective in small, infrequently administered doses associated with minimal side effects. Although suitable for occasional simple nausea and vomiting, nonprescription agents are often abandoned by the patient as symptoms continue or become progressively worse. As the patient’s condition warrants, prescription medications may be chosen, either as single-agent therapy or in combination.
The management of complex nausea and vomiting, for example, in patients who are receiving cytotoxic chemotherapy, may require combination therapy. In combination regimens, the goal is to achieve symptomatic control through administration of agents with different pharmacologic mechanisms of action.
Antacids
Patients who are experiencing simple nausea and vomiting may use various antacids. In this setting, single or combination nonprescription antacid products, especially those containing magnesium hydroxide, aluminum hydroxide, and/or calcium carbonate, may provide sufficient relief, primarily through gastric acid neutralization.
Antihistamine–Anticholinergic Drugs
Antiemetic drugs from the antihistaminic–anticholinergic category appear to interrupt various visceral afferent pathways that stimulate nausea and vomiting. These drugs are often initiated as self-care by the patient for simple nausea or vomiting, especially associated with motion sickness.
Benzodiazepines
Since nausea and vomiting are often associated with chemotherapy, radiotherapy, and surgery, anticipatory anxiety may occur prior to these therapies and may exacerbate symptoms. Benzodiazepines may be useful in this setting.
Benzodiazepines are relatively weak antiemetics and are primarily used to prevent anxiety or anticipatory nausea and vomiting (ANV) in patients receiving highly emetogenic chemotherapy. They are also useful against akathisia associated with metoclopramide therapy. Both alprazolam and lorazepam are used as adjuncts to other antiemetics in patients treated with cisplatin-containing regimens. Alprazolam is usually given orally and has an onset of about 60 minutes, while lorazepam is given orally or sublingually, which has a more rapid onset.
Butyrophenones
Two butyrophenone compounds that have antiemetic activity are haloperidol and its congener droperidol; both block dopaminergic stimulation of the CTZ. Although each agent is effective in relieving nausea and vomiting, haloperidol is not considered first-line therapy for uncomplicated nausea and vomiting but has been used in palliative care situations.8 The current labeling of droperidol recommends that all patients should undergo a 12-lead electrocardiogram prior to administration, followed by cardiac monitoring for 2 to 3 hours after administration because of the possibility of the development of potentially fatal QT prolongation and/or torsade de pointes.9 The clinical use of droperidol has effectively ceased outside of clinical trials in anesthesia.
Cannabinoids
Cannabinoids have complex effects on the CNS and their effects at receptors in neural tissues may explain efficacy in CINV. Oral dronabinol and nabilone are therapeutic options when CINV is refractory to other antiemetics; they are not indicated as first-line agents.10
Corticosteroids
Corticosteroids have demonstrated antiemetic efficacy since the initial recognition that patients who received prednisone as part of their Hodgkin’s disease protocol appeared to develop less nausea and vomiting than did those patients who were treated with protocols that excluded this agent. Methylprednisolone has also been used as a component of an antiemetic regimen, but the majority of trials have included dexamethasone. The site and mechanism of action of corticosteroids for CINV is unknown.
Dexamethasone is the most commonly used corticosteroid in the management of CINV and PONV, either as a single agent or in combination with 5-hydroxytryptamine-3 receptor antagonists (5-HT3-RA). Dexamethasone is effective in the prevention of both cisplatin-induced acute emesis and delayed nausea and vomiting associated with CINV when used alone or in combination.11–14 Corticosteroids affect almost every organ system. For patients with simple nausea and vomiting, steroids are not indicated and may be associated with unacceptable risks.
H2-Receptor Antagonists
Histamine2-receptor antagonists work by decreasing gastric acid production and are used to manage simple nausea and vomiting associated with heartburn or gastroesophageal reflux. Except for potential drug interactions with cimetidine, these agents cause few side effects when used for episodic relief.
5-Hydroxytryptamine-3 Receptor Antagonists
5-HT3-RAs block presynaptic serotonin receptors on sensory vagal fibers in the gut wall, effectively blocking the acute phase of CINV. These agents do not completely block the acute phase of CINV and are less efficacious in preventing the delayed phase, but they are the standard of care in the management of CINV, PONV, and radiation-induced nausea and vomiting (RINV). Issues involved in the use of dolasetron, granisetron, ondansetron, and palonosetron are reviewed in detail in the sections that follow.
Metoclopramide
Metoclopramide, a procainamide congener, blocks dopaminergic receptors centrally in the CTZ. It increases lower esophageal sphincter tone, aids gastric emptying, and accelerates transit through the small bowel, possibly through the release of acetylcholine. The prokinetic activity of metoclopramide makes it useful in patients with nausea and vomiting associated with diabetic gastroparesis. The introduction of the 5-HT3-RAs in the early 1990s supplanted the use of metoclopramide in CINV.
Olanzapine
Olanzapine is an antipsychotic that blocks several neurotransmitters including dopamine at D2 and 5-HT3-RA. Use of olanzapine, in combination with palonosetron and dexamethasone, effectively controlled acute and delayed CINV in patients receiving highly emetogenic chemotherapy as compared with aprepitant, palonosetron, and dexamethasone in a randomized, phase 3 clinical trial.15 The National Comprehensive Cancer Network (NCCN) antiemesis practice guideline includes olanzapine as one of many options in patients who experience breakthrough nausea and/or vomiting following prophylaxis for CINV.16 Sedation is the most common side effect with olanzapine; it should be used with caution in the elderly.
Phenothiazines
Phenothiazines have been the most widely prescribed antiemetic agents and appear to block dopamine receptors, most likely in the CTZ. They are marketed in an array of dosage forms, none of which appears to be more efficacious than another. These agents may be most practical for long-term treatment and are inexpensive in comparison with newer drugs. Rectal administration is a reasonable alternative in patients in whom oral or parenteral administration is not feasible.
Phenothiazines are most useful in adult patients with simple nausea and vomiting. IV prochlorperazine provided quicker and more complete relief with less drowsiness than IV promethazine in adult patients treated in an emergency department for nausea and vomiting associated with uncomplicated gastritis or gastroenteritis.17
Substance P/Neurokinin 1 Receptor Antagonists
Substance P is a peptide neurotransmitter in the NK family whose preferred receptor is the NK1 receptor. The acute phase of CINV is believed to be mediated by both serotonin and substance P, whereas substance P is believed to be the primary mediator of the delayed phase. Aprepitant and fosaprepitant are the first substance P/NK1 receptor antagonists in clinical use; at the time of this writing, casopitant is still in development.
The efficacy of aprepitant was demonstrated in patients receiving high-dose cisplatin-based chemotherapy13,14 and in patients receiving doxorubicin and cyclophosphamide,18 a regimen of moderate to high emetic risk. The three-drug regimen of aprepitant, dexamethasone, and ondansetron provided improved protection from vomiting for the 5 days after chemotherapy administration as compared with the combination of dexamethasone and ondansetron.
Aprepitant has the potential for numerous drug interactions because it is a substrate, moderate inhibitor, and an inducer of cytochrome isoenzyme CYP3A4 and an inducer of CYP2C9. It can increase serum concentrations of many drugs metabolized by CYP3A4, including docetaxel, paclitaxel, etoposide, irinotecan, ifosfamide, imatinib, vinorelbine, vincristine, and vinblastine. In clinical studies, aprepitant was concomitantly administered with etoposide, vinorelbine, or paclitaxel, with no adjustment in the doses of these agents to account for potential drug interactions. The efficacy of oral contraceptives may be reduced when given with aprepitant. Concomitant administration with warfarin may result in a clinically significant decrease in the international normalized ratio.19 The dose of oral dexamethasone should be reduced 50% when coadministered with aprepitant, because of the 2.2-fold increase in observed area under the plasma-concentration-versus-time curve.20 Aprepitant is not approved for use in children.
Fosaprepitant, an injectable form of aprepitant, has been approved by the FDA as an IV substitute for oral aprepitant on day 1 of the standard 3-day CINV prevention regimen, with oral aprepitant administered on days 2 and 3.21
CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING
There are five categories of CINV: acute, delayed, anticipatory, breakthrough, and refractory. Nausea and vomiting that occurs within 24 hours of chemotherapy administration is defined as acute CINV, whereas when it starts more than 24 hours after chemotherapy administration, it is defined as delayed CINV.
Nausea or vomiting that occurs prior to receiving chemotherapy is termed ANV. ANV is believed to be a learned, conditioned, or psychological response that occurs in about 25% of patients by the fourth cycle of chemotherapy.22 ANV triggers include tastes, odors, sights, or thoughts associated with chemotherapy. Risk factors associated with ANV include age under 50, nausea and/or vomiting after the previous chemotherapy session, anxiety, sweating and a feeling of warmth after the last chemotherapy cycle, and susceptibility to motion sickness.23
In the setting of optimal antiemetic prophylaxis and no prior emesis, reported chemotherapy-induced ANV is rare. Use of newer antiemetic regimens appears to have resulted in a decreased rate of ANV.24
Breakthrough nausea and vomiting is defined as emesis occurring despite prophylactic administration of antiemetics and requiring the use of rescue antiemetics. Historically, breakthrough emesis occurs in 10% to 40% treated with modern-day antiemetics.25
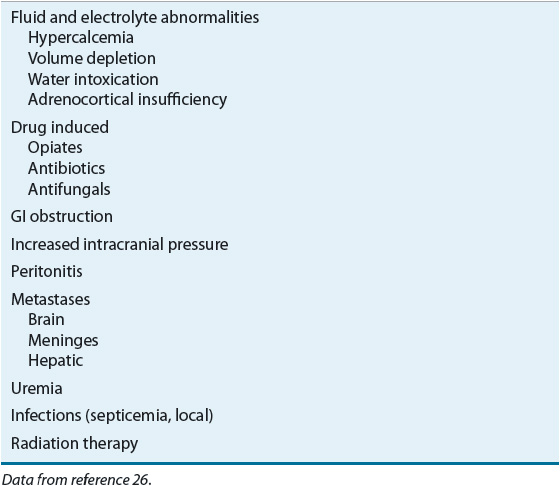
Refractory nausea and vomiting is evident when there is a poor response to multiple antiemetic regimens. In addition to the emetic risk of various cytotoxic regimens, other common etiologies have been proposed for the development of nausea and vomiting in cancer patients (Table 22-5).26
TABLE 22-5 Nonchemotherapy Etiologies of Nausea and Vomiting in Cancer Patients
![]() The primary goal for CINV is to prevent nausea and/or vomiting. Optimal control of acute nausea and vomiting is known to impact positively on the incidence and control of delayed nausea and vomiting and ANV.25
The primary goal for CINV is to prevent nausea and/or vomiting. Optimal control of acute nausea and vomiting is known to impact positively on the incidence and control of delayed nausea and vomiting and ANV.25
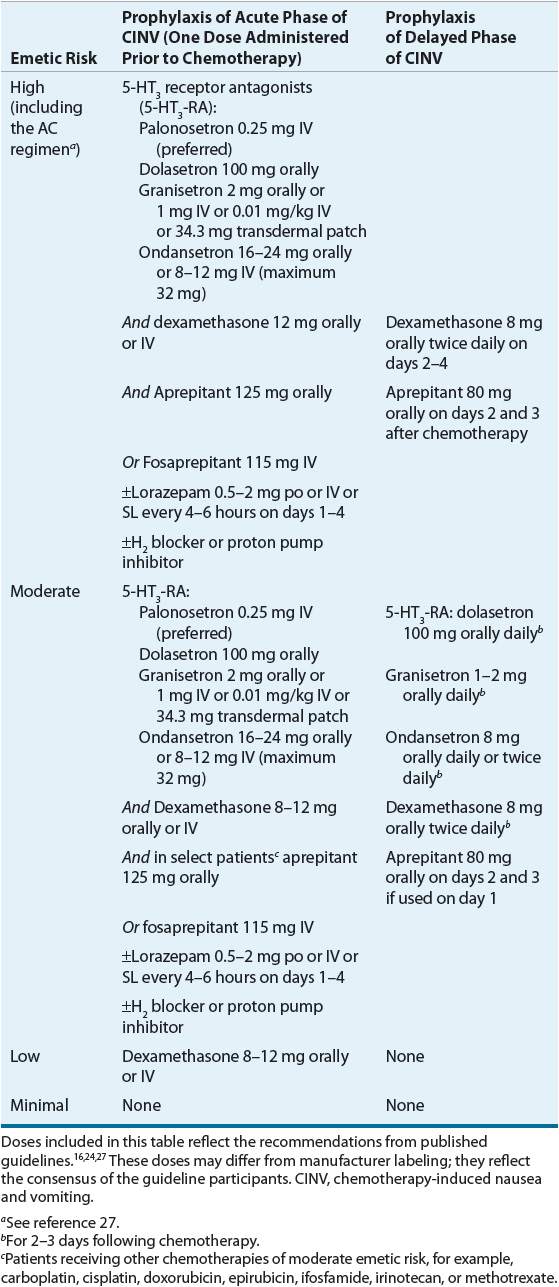
Clinical practice guidelines for the use of antiemetics in CINV have been published by the NCCN,16 the Multinational Association of Supportive Care in Cancer/European Society of Oncology (MASCC/ESMO),24 and the American Society of Clinical Oncology (ASCO).27 The NCCN guidelines are updated annually, while the ASCO and ESMO guidelines appear to be updated less frequently. Despite the demonstrated improvement in outcomes with the use of these practice guidelines, they are underutilized by a high percentage of practitioners.28 Furthermore, product availability and recommended doses are often institution-specific and may vary considerably from the doses listed in Table 22-6.
TABLE 22-6 Dosage Recommendations for CINV for Adult Patients
Principles of Antiemetic Use for CINV
ASCO, MASCC, and the NCCN consensus groups share several of the principles listed below that appear to be important for the effective prevention of CINV in adults29,30:
1. The primary goal of emesis prevention is no nausea and/or vomiting throughout the period of emetic risk.
2. The duration of emetic risk is 2 days for patients receiving moderately emetogenic chemotherapy and 3 days for highly emetogenic chemotherapy. Emetic prophylaxis should be provided through the entire period of risk.
3. ![]() The selection of the antiemetic regimen should be based on the chemotherapy drug with highest emetogenicity (see Table 22-2). Prior emetic experience and patient-specific factors should also be considered.
The selection of the antiemetic regimen should be based on the chemotherapy drug with highest emetogenicity (see Table 22-2). Prior emetic experience and patient-specific factors should also be considered.
4. When given in equipotent doses, oral and IV 5-HT3-RAs are equivalent in efficacy.
5. The toxicities of antiemetics should be considered and managed appropriately.
Prophylaxis of Acute CINV
Each of the practice guidelines states that the most effective classes of drugs for the prevention of acute emesis are the 5-HT3-RAs, NK1 receptor antagonists, and glucocorticoids (especially dexamethasone). Treatment recommendations for the different categories of emesis are outlined in Table 22-6.
High Emetogenic Chemotherapy (HEC)
Patients receiving HEC should receive a three-drug antiemetic regimen given before the administration of chemotherapy (day 1) that includes a 5-HT3-RA (e.g., dolasetron, granisetron, ondansetron, or palonosetron), dexamethasone, and an NK1 receptor antagonist (e.g., aprepitant or fosaprepitant). Dexamethasone should be given on days 2 to 4 and aprepitant on days 2 and 3 to prevent delayed emesis.
Any of the 5-HT3-RAs may be used on day 1. The ASCO and NCCN guidelines prefer IV palonosetron. The use of IV dolasetron is contraindicated because it has a higher association of prolonged QTc interval. Granisetron transdermal patch should be applied 24 to 48 hours prior to chemotherapy (duration of effect is 7 days).