CHAPTER 76 Male reproductive system
The male reproductive system consists of the spermatic cord and scrotum, testes and epididymes, vas deferens, prostate, seminal vesicles and penis.
SPERMATIC CORD
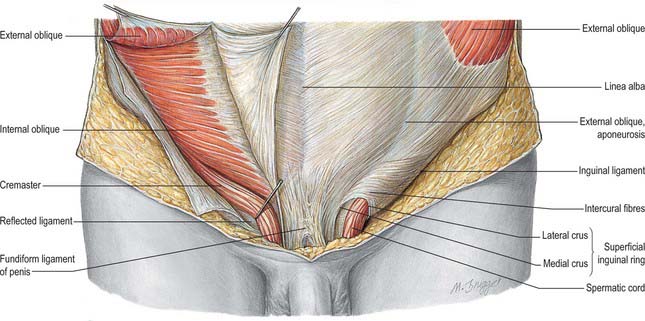
As the testis traverses the abdominal wall into the scrotum during early life, it carries its vessels, nerves and vas deferens with it. These meet at the deep inguinal ring to form the spermatic cord, which suspends the testis in the scrotum and extends from the deep inguinal ring to the posterior aspect of the testis (Fig. 76.1). The left cord is a little longer than the right. Between the superficial ring and testis the cord is anterior to the tendon of adductor longus. It is crossed anteriorly by the superficial external pudendal artery and posteriorly by the deep external pudendal artery. The ilioinguinal nerve lies inferior to the cord as it traverses the inguinal canal.
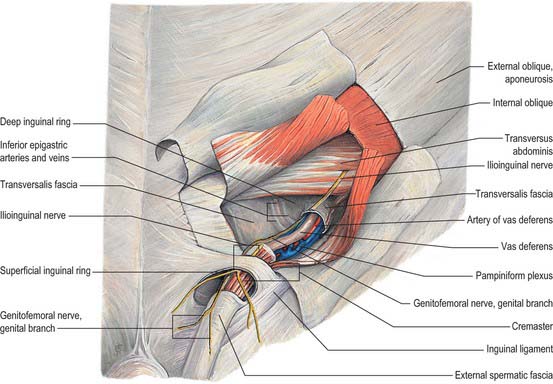
In the canal the cord acquires coverings from the layers of the abdominal wall that extend into the scrotal wall as the internal spermatic, cremasteric and external spermatic fasciae (Fig. 76.2). The internal spermatic fascia is derived from the transversalis fascia and forms a thin, loose layer around the spermatic cord. The cremasteric fascia contains fasciculi of skeletal muscle united by loose connective tissue to form the cremaster muscle which is continuous with internal oblique. The external spermatic fascia descends from the crura of the superficial ring and is a thin fibrous stratum continuous above with the aponeurosis of external oblique. The spermatic cord contains the vas deferens; the testicular artery and veins, cremasteric artery (a branch of the inferior epigastric artery) and artery to the vas deferens (from the superior vesical artery); the genital branch of the genitofemoral nerve and cremasteric nerve and the sympathetic components of the testicular plexus (which are joined by filaments from the pelvic plexus accompanying the artery to the vas deferens); 4–8 lymph vessels draining the testis. All of these structures are conjoined by loose connective tissue (Figs 76.2, 76.3A).
SCROTUM
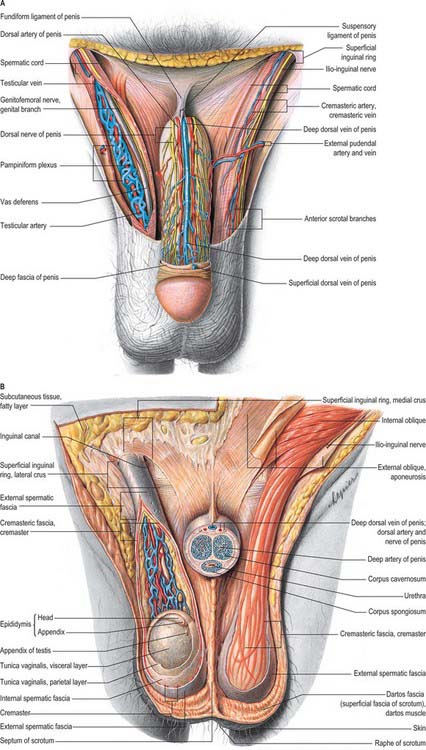
The scrotum is a cutaneous fibromuscular sac containing the testes and lower parts of the spermatic cords and hangs below the pubic symphysis between the anteromedial aspects of the thighs. It consists of skin, dartos muscle and external spermatic, cremasteric and internal spermatic fasciae. The internal spermatic fascia is loosely attached to the parietal layer of the tunica vaginalis (Fig. 76.3B). The scrotum is divided into right and left halves by a cutaneous raphe, which continues ventrally to the inferior penile surface and dorsally along the midline of the perineum to the anus. The raphe indicates the bilateral origin of the scrotum from the genital swellings. The left side of the scrotum is usually lower because the left spermatic cord is longer.
Vascular supply and lymphatic drainage
The arteries supplying the scrotum include the external pudendal branches of the femoral artery, the scrotal branches of the internal pudendal artery, and a cremasteric branch from the inferior epigastric artery. Dense subcutaneous plexuses of scrotal vessels carry a substantial blood flow, facilitating heat loss. Arteriovenous anastomoses of a simple but large-calibre type are also prominent. The veins follow the corresponding arteries. The skin of the scrotum is drained by vessels that accompany the external pudendal blood vessels to the superficial inguinal nodes (Fig. 76.3A).
TESTES AND EPIDIDYMIS
TESTES
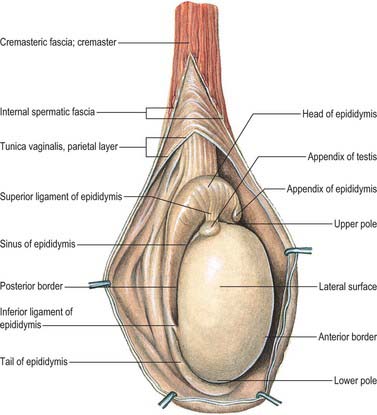
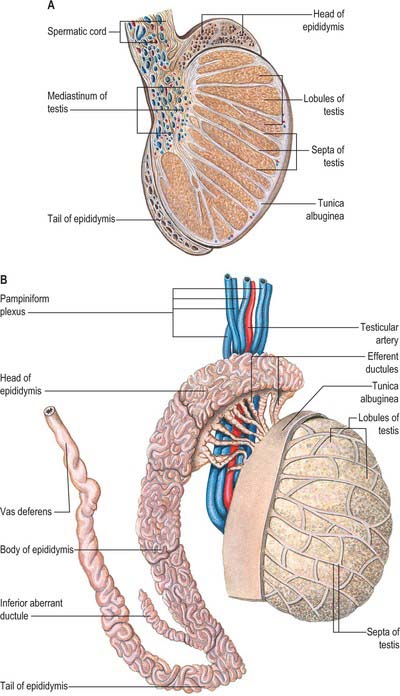
The testes are the primary reproductive organs or gonads in the male. They are ovoid, reproductive and endocrine organs responsible for sperm production and testosterone production. They are suspended in the scrotum by scrotal tissues including the dartos muscle and spermatic cords. Average testicular dimensions are 4–5 cm in length, 2.5 cm in breadth and 3 cm in anteroposterior diameter; their weight varies from 10.5–14 g. The left testis usually lies lower than the right testis. Each testis lies obliquely within the scrotum, the upper pole tilted anterolaterally and the lower posteromedially. The anterior aspect is convex, whereas the posterior aspect is nearly straight and has the spermatic cord attached to it. Anterior, medial and lateral surfaces and both poles are convex, smooth and covered by the visceral layer of the serosal tunica vaginalis, the parietal layer and the scrotal tissues, in that order from within outwards (Fig. 76.4). Each testis is separated from its fellow by a fibrous median raphe which is deficient superiorly. The posterior aspect of each testis is only partly covered by serosa; the epididymis adjoins its lateral part (see below). The testis is invested by three coats, which are, from outside inwards, the tunica vaginalis, tunica albuginea and tunica vasculosa (Fig. 76.4).
Tunica vaginalis
The tunica vaginalis is the lower end of the peritoneal processus vaginalis, whose formation precedes the descent of the fetal testis from the abdomen to the scrotum (see p. 1320). After this migration, the proximal part of the tunica, from the internal inguinal ring almost to the testis, contracts and is obliterated, leaving a closed distal sac into which the testis is invaginated. The tunica is reflected from the testis onto the internal surface of the scrotum, so forming the visceral and parietal layers of the tunica. The visceral layer covers all aspects of the testis except most of the posterior aspect. Posteromedially it is reflected forwards to the parietal layer. Posterolaterally it passes to the medial aspect of the epididymis and lines the epididymal sinus, and then passes laterally to its posterior border where it is reflected forwards to become continuous with the parietal layer. The visceral and parietal layers are continuous at both poles, but at the upper pole the visceral layer surmounts the head of the epididymis before reflexion. There is always a very fine film of fluid between the two layers of the tunica vaginalis. This fluid layer can increase in inflammatory and neoplastic conditions of the testis, leading to a hydrocele (see below).
Tunica albuginea
The tunica albuginea is a dense, bluish-white covering for the testis, composed mainly of interlacing bundles of collagen fibres. It is covered externally by the visceral layer of the tunica vaginalis, except at the epididymal head and tail and the posterior aspect of the testis, where vessels and nerves enter (Fig. 76.5A,B). The tunica vaginalis covers the tunica vasculosa and, at the posterior border of the testis, projects into the testicular interior as a thick, incomplete fibrous septum, the mediastinum testis, which extends from the upper to the lower end of the testis. Testicular vessels run within the mediastinum testis.
Vascular supply and lymphatic drainage
Testicular arteries
The testicular arteries are two long, slender vessels which arise anteriorly from the aorta a little inferior to the renal arteries. Each passes inferolaterally under the parietal peritoneum on psoas major. The right testicular artery lies anterior to the inferior vena cava and posterior to the horizontal part of the duodenum, right colic and ileocolic arteries, root of the mesentery and terminal ileum. The left testicular artery lies posterior to the inferior mesenteric vein, left colic artery and lower part of the descending colon. Each artery crosses anterior to the genitofemoral nerve, ureter and the lower part of the external iliac artery and passes to the deep inguinal ring to enter the spermatic cord and travel via the inguinal canal to enter the scrotum (Figs 76.3A, 76.6). At the posterosuperior aspect of the testis the testicular artery divides into two branches on its medial and lateral surfaces: these pass through the tunica albuginea and ramify in the tunica vasculosa. Terminal branches enter the testis over its surface. Some pass into the mediastinum testis and loop back before reaching their distribution. Capillaries lying next to seminiferous tubules penetrate the layers of interstitial tissue and may form part of the ‘blood–testis’ barrier. They run either parallel to the tubules or across them but do not enter their walls. They are separated from germinal and supporting cells by a basement membrane and variable amounts of fibrous tissue containing interstitial cells: selective exchange phenomena involving androgens and immune substances occur here.
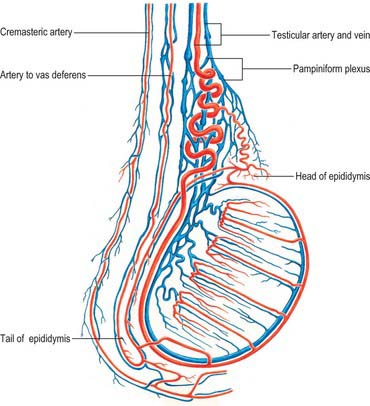
The testis also receives blood from the cremasteric branch of the inferior epigastric artery, and from the artery to the vas deferens (Fig. 76.6). Interference with the testicular artery high in the abdomen therefore usually leaves the testis unharmed, whereas interruption in the region of the spermatic cord may interfere with all of these vessels and lead to infarction. Ligating both the testicular artery and vein high up interrupts the venae comitantes of the artery which anastomose with the internal spermatic veins and can be responsible for recurrence of a varicocele (see below).
Testicular veins
The testicular veins emerge posteriorly from the testis, drain the epididymis and unite to form the pampiniform plexus, a major component of the spermatic cord that ascends anterior to the vas deferens (Fig. 76.7). In the inguinal canal the pampiniform plexus is drained by three or four veins which run into the abdomen through the deep inguinal ring. Within the abdomen these veins coalesce into two veins, which ascend on each side of the testicular artery, anterior to psoas major and the ureter, and behind the peritoneum. The left veins pass behind the lower descending colon and inferior margin of the pancreas and are crossed by the left colic vessels, and the right veins pass behind the terminal ileum and horizontal part of the duodenum and are crossed by the root of the mesentery, ileocolic and right colic vessels. The veins join to form single right or left testicular veins: the right testicular vein opens into the inferior vena cava at an acute angle just inferior to the level of the renal veins, and the left testicular vein opens into the left renal vein at a right angle (Fig. 76.7). The testicular veins contain valves.
Microstructure
The surface of the testis is covered closely by the visceral tunica vaginalis, a layer of flat mesothelial cells similar to, and continuous with, the peritoneal lining. The visceral layer is separated from the parietal tunica vaginalis (the outer layer of the double fold of peritoneum that accompanies the descending testis (see p. 1320)), by a potential space containing serous fluid, which acts as a lubricant and allows movement of the testis within the scrotum. The testicular capsule proper, the tunica albuginea, is tough and collagenous and thickened posteriorly as the mediastinum testis. Beneath the tunica albuginea is a thin layer of connective tissue containing the superficial blood vessels. Blood vessels, lymphatics and the genital ducts enter or leave the body of the testis at the mediastinum (Fig. 76.8).
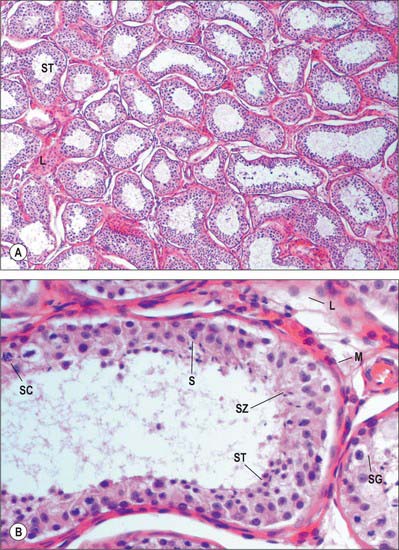
Septa from the mediastinum extend internally to partition the testis into approximately 250 lobules (Fig. 76.5A). These differ in size, and are largest and longest in the centre. Each lobule contains one to four convoluted seminiferous tubules, much-coiled loops whose free ends both open into channels (the rete testis) within the mediastinum. The loose connective tissue between seminiferous tubules contains several layers of contractile peritubular myoid cells and clusters of androgen-producing interstitial (Leydig) cells.
There are 400–600 seminiferous tubules in each testis, each 70–80 cm long and 0.12–0.3 mm. in diameter. They are pale in early life, but in old age they contain much fat and are deep yellow. Each tubule is surrounded by a basal lamina, on which rests a complex, stratified seminiferous epithelium containing spermatogenic cells and supportive Sertoli cells (Fig. 76.9A). When active, the spermatogenic cells include basally situated spermatogonia and their progeny in the adluminal compartment, spermatocytes, spermatids and mature spermatozoa. Among the spermatids may be residual bodies, spherical structures derived from surplus spermatid cytoplasm shed during maturation and phagocytosed by Sertoli cells.
Spermatogonia
Spermatogonia, the stem cells for all spermatozoa, are descended from primordial germ cells which migrate into the genital cords of the developing testis (Fig. 76.9B). In the fully differentiated testis they are located along the basal laminae of the seminiferous tubules. Several types of spermatogonia are recognized on the basis of cell and nuclear dimensions, distribution of nuclear chromatin (dark, condensed or pale, euchromatic) and histochemical and ultrastructural data. The three basic groups of spermatogonia are dark type A (Ad), pale type A (Ap), and type B. Ad cells divide mitotically to maintain the population of spermatogonia which, before puberty, is small but increases under androgenic stimulation. Some divisions give rise to Ap cells which also divide mitotically but remain linked in clusters by fine cytoplasmic bridges. These are the precursors of type B cells, which are committed to the spermatogenic sequence. At about the time that type B cells enter a final round of DNA synthesis, without undergoing cytokinesis, they leave the basal compartment and cross the blood–testis barrier to enter meiotic prophase as primary spermatocytes. These coordinated processes are under the control of Sertoli cells.
Primary and secondary spermatocytes
Primary spermatocytes have a diploid chromosome number but duplicated sister chromatids (DNA content is thus 4N, where N is the DNA content of haploid spermatozoa), and are all at some stage of a long meiotic prophase (p. 22) of approximately 3 weeks. Primary spermatocytes are large cells with large round nuclei in which the nuclear chromatin is condensed into dark, threadlike, coiled chromatids at different stages in the process of crossing over and genetic exchange between chromatids of maternal and paternal homologues. These cells give rise to secondary spermatocytes with a haploid chromosome complement (but 2N DNA content), the reduction division is designated as meiosis I. Few secondary spermatocytes are seen in tissue sections because they rapidly undergo the second meiotic (equatorial) division, where sister chromatids separate (DNA content now being N), to form haploid spermatids. Theoretically each primary spermatocyte produces four spermatids, but some degenerate during maturation so that the yield is lower.
Spermatids
Spermatids do not divide again but gradually mature into spermatozoa by a series of nuclear and cytoplasmic changes known as spermiogenesis. All of these maturational changes take place while the spermatids remain closely associated with Sertoli cells and linked by cytoplasmic bridges with each other. The first phase of spermiogenesis is the Golgi phase, when hydrolytic enzymes accumulate in Golgi vesicles that subsequently coalesce into a single large acrosomal vesicle close to the nucleus. The pair of centrioles migrates to the opposite posterior pole. The distal centriole begins to generate the axoneme, a circular arrangement of nine microtubule doublets surrounding a central pair. In the cap phase, the acrosomal vesicle flattens and envelops the anterior half of the nucleus to form an acrosomal cap which comes to occupy the presumptive anterior pole of the spermatozoon, furthest from the lumen of the tubule.