Left Hemicolectomy for Treatment of Malignancy
Martin R. Weiser
Surgical resection remains the optimal curative treatment for left colon cancer. This includes removal of the primary lesion with clear margins and resection of locoregional lymph nodes. Determining the extent of lymphadenectomy can be the most challenging aspect of an oncologic resection. In colorectal cancer, pathologic evaluation of at least 12 lymph nodes optimizes staging and is associated with better outcome. More extensive resection does not necessarily translate into prolonged survival and can be associated with increased morbidity. For example, “extended” left hemicolectomy, involving ligation of the inferior mesenteric artery at the level of the aorta, increases the risk of autonomic nerve injury but has not been shown to reduce recurrence; therefore, it should be used selectively.
This chapter will focus on left hemicolectomy for cancer in which the extent of colon resection is defined by adequate lymphadenectomy. Successful execution of an oncologic left hemicolectomy first requires a clear understanding of the regional anatomy and lymph node drainage pattern.
The blood vessels and regional lymphatics of the left colon reside in the mesentery. In general, the distal transverse colon is supplied by the middle colic pedicle, which is the first branch of the superior mesenteric artery as it emerges from beneath the pancreas. The left colic artery supplies the left colon distal to the splenic flexure and is the first ascending branch of the inferior mesenteric artery. The left colon has a relatively well-developed marginal artery and vein, which form an adjacent mesenteric vascular arcade. This collateral circulation may be underdeveloped in some patients, however, resulting in a potential watershed area at the splenic flexure, and this should be considered if the inferior mesenteric artery is divided at its origin. The superior hemorrhoidal artery is the distal-most branch of the inferior mesenteric artery, giving rise to multiple sigmoidal arteries before being enveloped in the mesorectum at the sacral promontory. The venous return from the left colon runs with the arteries in the mesentery, coalescing to form the inferior mesenteric vein just lateral to the duodenum, then uniting with the splenic vein and superior mesenteric vein to form the portal vein.
The majority of lymphatic channels run in the submucosa of the colon wall. Invasive colonic neoplasms are defined by extension of dysplastic glands beyond the muscularis mucosa into the submucosa of the intestinal wall. It should be noted that this is distinctly different from the definition of other gastrointestinal malignancies, in which carcinoma is described as dysplastic cell invasion beyond the epithelial basement membrane. The rationale stems from the edict that metastatic colonic disease is not possible unless dysplastic glands penetrate into the submucosa, where the majority of lymphatics reside.
The efferent colonic lymphatic channels pass from the submucosa to the intramuscular and subserosal plexus of the bowel to the epicolic nodes, which are adjacent to the large intestine. Paracolic lymph nodes are adjacent to the marginal vessel along the mesenteric side of the colon; these and the epicolic lymph nodes are usually the first sites of nodal metastases. Intermediate nodes are found along the major arterial branches in the mesocolon, and principal nodes are located at the origin of the vascular pedicles.
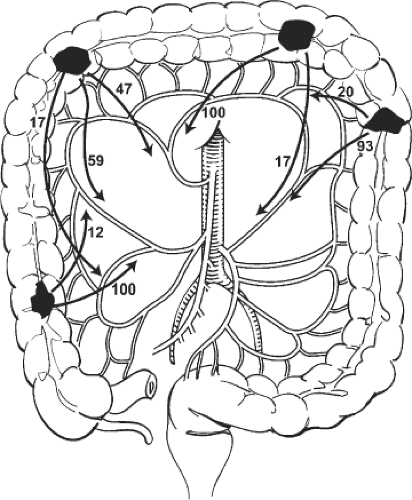
The bidirectional lymphatic flow from colonic tumors has been well documented in classic studies of cleared colon specimens (Fig. 1), forming the basis of oncologic resection. Specifically, lymph node metastases are identified along major vascular pedicles both proximal and distal to the lesion. For example, as noted in Figure 1, lesions at the splenic flexure metastasize along the left colic artery and left branch of the middle colic artery in 93% and 20% of cases, respectively. Therefore, proper lymphadenectomy includes resection of principal vessels above and below the lesion with en bloc removal of epicolic, paracolic, and intermediate lymph nodes.
Left hemicolectomy is indicated for patients with (a) malignant lesions, (b) a history of prior malignant polypectomy with high-risk pathologic features, such that there is a significant possibility of lymph nodes metastases, and (c) high-risk premalignant polyps not amenable to endoscopic removal. High-risk pathologic features include poor differentiation, lymphovascular invasion, involvement of the base of the polyp (Haggitt level 4), and margin less than 2 mm. The rationale for performing colon resection with lymphadenectomy for potentially benign polyps is to obviate the need for reoperation if invasive cancer is ultimately diagnosed. Therefore, patients who have relatively high-risk adenomas that are not amenable to endoscopic polypectomy, large or sessile lesions, or polyps with significant dysplasia are advised to undergo formal hemicolectomy.
Thorough preoperative workup is required to optimize an oncologic resection. This includes a history and physical examination, pulmonary and cardiac evaluations as indicated, and a complete extent-of-disease workup. The colon should be fully evaluated for additional lesions. Synchronous polyps and invasive cancers are noted in 30% and 3% of cases, respectively. If the primary lesion was detected by a “noninvasive” modality such as virtual colonoscopy (computed tomographic (CT) colography), full endoscopic evaluation is indicated for histologic confirmation. Multiple site (three- or four-quadrant) tattooing of small lesions or polypectomy sites can be helpful for localizing disease at the time of resection.
For obstructing lesions, endoscopic stenting is a useful bridge to colectomy. Once stented, the colon can be decompressed, cleared of solid material with a mechanical bowel preparation, and evaluated by colonoscopy for synchronous lesions. In some cases, this may obviate the need for subtotal colectomy.
A thorough workup for metastatic disease, including a CT scan with oral and intravenous contrast, is advocated to optimize treatment planning. Identification of metastatic disease prior to surgery informs patient counseling and consideration of synchronous liver resection. In patients with asymptomatic primary colon lesions and metastatic disease, initiation of chemotherapy is a viable alternative to immediate colectomy. This approach is theoretically advantageous as it allows for rapid treatment of survival-limiting systemic disease, as well as identification of patients with chemo-responsive tumors who may be candidates for aggressive surgical debulking at the time of colectomy.
Recent reports indicate that bowel preparation is not mandatory for successful colon surgery. Indeed, we have known for some time that primary closure of traumatic colotomy is a viable, even preferable treatment option. However, in the setting of elective colectomy for cancer, bowel preparation remains advantageous. Removal of solid stool from the colon enables easy intestinal manipulation, assessment of synchronous disease by palpation, and intraoperative colonoscopy when necessary.
Preoperatively, patients are instructed to follow a clear-liquid diet at home, beginning the morning prior to day of surgery. Although the utility of bowel prep has been called into question, the author favors mechanical bowel preparation in most cases. A second-generation cephalosporin (e.g., 2 g of cefotetan) is administered intravenously within 1 hour of incision. In patients with penicillin allergies we use an aminoglycoside, such as gentamicin (1.5 mg/kg) and clindamycin (900 mg). In patients requiring endocarditis prophylaxis, ampicillin (2 g), clindamycin (900 mg), and an aminoglycoside are administered preoperatively. Lastly, in penicillin-allergic patients who require endocarditis prophylaxis, vancomycin (1 g), metronidazole (500 mg), and an aminoglycoside are used.
Details of Surgical Preparation
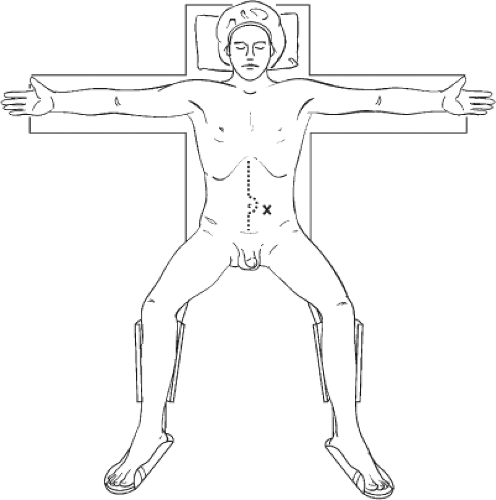
After induction of general anesthesia, the patient is placed in the supine position, with the arms abducted on armboards and the legs in a low (modified Lloyd–Davies) lithotomy position (Fig. 2). The pelvis is elevated on gel pads, permitting easy access to the perineum. The rectum is then irrigated with 1 L of saline to remove any residual fecal material if a colorectal anastomosis is anticipated. Prophylaxis against deep vein thrombosis is accomplished by placement of intermittent pneumatic compression devices at the time of anesthesia induction. In high-risk cases, preoperative unfractionated heparin or low molecular weight heparin is administered before induction as well. Postoperatively, both pneumatic boots and heparin are used. The abdomen and perineum are prepared with an antibacterial wash and then draped. A sterile Foley catheter is inserted.
After squaring the abdomen with towels, a sterile plastic covering is placed over it to secure the draping. The patient is explored through a midline incision; the extent
depends on location of the lesion. Cephalad extension to the xiphoid process is used for splenic flexure lesions, and caudad extension to the pubis is advantageous for lower sigmoid lesions. Moist laparotomy pads and a plastic wound protector are used to contain any intestinal spillage that may occur and to keep the abdominal wall moist. A thorough abdominal exploration is performed with particular attention to the liver, omentum, and peritoneal surfaces. In women, the ovaries are carefully inspected to rule out metastatic disease and ovarian cancer. (Routine prophylactic oophorectomy is generally not recommended unless the patient is postmenopausal or has a significant family history of ovarian cancer.) The intestine is evaluated by palpation to rule out synchronous disease, especially in cases in which a satisfactory colonoscopy could not be performed. Intraoperative colonoscopy, ideally with carbon dioxide insufflation to reduce intestinal distension, is occasionally necessary if the lesion cannot be identified by palpation or visualization of serosal tattoo markings.
depends on location of the lesion. Cephalad extension to the xiphoid process is used for splenic flexure lesions, and caudad extension to the pubis is advantageous for lower sigmoid lesions. Moist laparotomy pads and a plastic wound protector are used to contain any intestinal spillage that may occur and to keep the abdominal wall moist. A thorough abdominal exploration is performed with particular attention to the liver, omentum, and peritoneal surfaces. In women, the ovaries are carefully inspected to rule out metastatic disease and ovarian cancer. (Routine prophylactic oophorectomy is generally not recommended unless the patient is postmenopausal or has a significant family history of ovarian cancer.) The intestine is evaluated by palpation to rule out synchronous disease, especially in cases in which a satisfactory colonoscopy could not be performed. Intraoperative colonoscopy, ideally with carbon dioxide insufflation to reduce intestinal distension, is occasionally necessary if the lesion cannot be identified by palpation or visualization of serosal tattoo markings.
Lymphadenectomy and Intestinal Resection
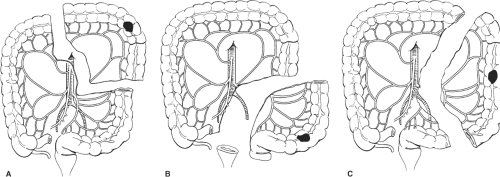
The length of intestine resection is determined by the extent of intestinal devascularization required for proper lymphadenectomy (Fig. 3). Obtaining a 5-cm margin of normal colon proximal and distal to the lesion is rarely an issue. In general, tumors located at the splenic flexure and left colon require resection of lymph nodes along the left branch of the middle colic, left colic, and first sigmoidal pedicles (Fig. 3A). Lesions in the sigmoid colon generally require lymphadenectomy along the inferior mesenteric artery (Fig. 3B). The pedicle is generally ligated distal to the left colic artery at the level of the superior hemorrhoidal artery. Routine high ligation of the inferior mesenteric artery at the level of the aorta does not improve survival, is associated with injury to sympathetic trunks, and should only be performed selectively. The rare lesion located at the junction of the left and sigmoid colon (Fig. 3C), which lies in the narrow area between the left colic and first sigmoidal pedicles, may benefit from ligation of the inferior mesenteric artery at its origin to include lymphadenectomy along all the left colic and superior rectal pedicles.
Tumors of the Splenic Flexure and Left Colon
The operating surgeon stands on the patient’s left side, and the assistant on the right. Optimal exposure is initially obtained by retracting the small bowel cephalad and, to the right, in a moist laparotomy pad, using a self-retaining retractor such as a Bookwalter or Thompson retractor (Fig. 4). The sigmoid colon is first mobilized off the lateral sidewall and retroperitoneum: the assistant retracts the sigmoid colon medially, and the lateral attachment of the colon is meticulously dissected with cautery by the operating surgeon. This maneuver exposes the peritoneal reflection, which often extends onto the sigmoid mesentery. It is important to dissect the parietal peritoneum off the sigmoid mesentery to ensure entry into the correct avascular plane. This maintains the lateral position of the gonadal vessels and ureter. The left ureter is identified medial to the gonadal vessels in the retroperitoneum. The peritoneal reflection is then incised along the white line of Toldt, proximally to the splenic flexure, to mobilize the left colon. During this maneuver, care must be taken to continuously sweep the gonadal vessels and ureter laterally, in order to avoid lateral retroperitoneal bleeding. Bleeding from injury to the gonadal vessels is cautiously controlled with clips or cautery, with attention to the location of the ureter at all times.
After the sigmoid and distal left colon are mobilized, the self-retaining retractor is repositioned to allow visualization and mobilization of the splenic flexure. Lateral colonic dissection is continued along the white line of Toldt to the lower pole of the spleen. For splenic flexure lesions, the distal omentum is resected with the colon by dividing the gastrocolic ligament and gastroepiploic arcades with entry into the lesser sac. For descending colon or sigmoid lesions, the distal omentum does not necessarily need to be resected with the specimen, and the gastrocolic omentum is dissected off the distal transverse colon, which allows entry into the lesser sac. Next, using simultaneous gentle inferior tension on the transverse and descending colon,
the renocolic and splenocolic ligaments are divided with cautery. These maneuvers mobilize the splenic flexure off the retroperitoneum, including the tail of the pancreas. Care must be taken to avoid excessive retraction on the colon, as the splenic flexure is often closely applied to the inferior aspect of the spleen and undue tension can cause a splenic capsular tear, resulting in considerable hemorrhage. If spleno-omental adhesions are noted, they should be initially divided to prevent such a capsular tear when tension is applied to the omentum. Should hemorrhage occur from splenic capsular tears, hemostasis can usually be attained using a variety of methods, including argon beam coagulation and use of hemostatic agents.
the renocolic and splenocolic ligaments are divided with cautery. These maneuvers mobilize the splenic flexure off the retroperitoneum, including the tail of the pancreas. Care must be taken to avoid excessive retraction on the colon, as the splenic flexure is often closely applied to the inferior aspect of the spleen and undue tension can cause a splenic capsular tear, resulting in considerable hemorrhage. If spleno-omental adhesions are noted, they should be initially divided to prevent such a capsular tear when tension is applied to the omentum. Should hemorrhage occur from splenic capsular tears, hemostasis can usually be attained using a variety of methods, including argon beam coagulation and use of hemostatic agents.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree