Laparoscopic Pelvic and Retroperitoneal Lymph Node Dissection
Louis R. Kavoussi
Ornob P. Roy
Brian D. Duty
Regional lymphadenectomy is used to stage and treat a variety of genitourinary (GU) malignancies. Pelvic lymph node dissection (PLND) is performed in prostate, bladder, penile, and urethral carcinoma. Retroperitoneal lymph node dissection (RPLND) is utilized in patients with metastatic germ cell tumors, select gynecologic malignancies, and sarcomas of the cord. Also, retroperitoneal node dissections are being advocated for select patients with transitional cell carcinoma (TCC) of the upper tract and renal cell carcinoma (RCC). Laparoscopic techniques have been applied to these operations to minimize morbidity.
Laparoscopic Pelvic Lymphadenectomy
Prostate cancer is the most common noncutaneous malignancy in men. Approximately 190,000 men were diagnosed with prostate cancer within the United States in 2009. Over 27,000 men died from the disease. Patients with clinically localized disease may undergo active surveillance or primary therapy with either extirpative surgery or radiation treatment.
No randomized studies have shown that local therapy provides a survival advantage in patients with metastatic disease. Unfortunately, no radiologic modality exists to reliably detect lymph node metastases. Computed tomography (CT) and magnetic resonance imaging (MRI) rely on size-based criteria to diagnose node-positive disease,
resulting in sensitivities of roughly 30%. Fine needle aspiration and immunoscintigraphy are also inaccurate. Pelvic lymphadenectomy remains the most accurate means of detecting lymph node involvement.
resulting in sensitivities of roughly 30%. Fine needle aspiration and immunoscintigraphy are also inaccurate. Pelvic lymphadenectomy remains the most accurate means of detecting lymph node involvement.
Laparoscopic PLND for prostate cancer was first reported by Schuessler in 1991. Subsequent studies demonstrated equivalent nodal yield and complication rates compared with the open technique while decreasing blood loss, analgesic use, length of hospital stay, and duration of convalescence.
Following its introduction, laparoscopic PLND became the most common laparoscopic urologic procedure. However, its role has steadily declined following widespread utilization of prostate-specific antigen (PSA) screening, which has resulted in a dramatic stage migration-favoring organ-confined disease. At present, only 5% of individuals present with node-positive disease.
Multiple nomograms have been developed to predict lymph node involvement based upon routinely available parameters (clinical stage, PSA level, and histologic Gleason score), further obviating the need for staging lymphadenectomy. Based upon these clinical parameters patients can be put into low-, moderate-, and high-risk categories. Low-risk patients do not require PLND because their risk of metastatic disease is less than 2%. Pelvic node dissection is recommended for patients with moderate- and high-risk disease undergoing retropubic prostatectomy. Laparoscopic PLND should be performed prior to definitive therapy in moderate- and high-risk patients undergoing perineal prostatectomy or radiotherapy. Additionally, laparoscopic PLND should be contemplated in high-risk patients being considered for open retropubic prostatectomy in order to spare them the morbidity of a lower midline incision should their nodes prove to be positive.
Laparoscopic PLND has been applied to other GU malignancies. For bladder cancer there is almost no role for laparoscopic PLND independent of cystectomy. However, laparoscopic radical cystectomy with extended PLND has been reported. Most series have been robotic assisted. Long-term functional and oncologic outcomes are being assessed with preliminary data suggesting equivalence.
Penile squamous cell carcinoma metastasizes to the inguinal, followed by pelvic, lymph nodes. Patients with locally advanced disease undergo inguinal lymph node dissection for both staging and therapeutic purposes. However, inguinal lymph node dissection provides no oncologic benefit to patients with positive pelvic nodes. Therefore, laparoscopic PLND has the potential to spare patients with suspicious pelvic nodes the morbidity of an inguinal dissection, which includes chronic lymphedema and wound infection in 25% and 10% of cases, respectively.
Carcinoma of the posterior urethra is a rare, but aggressive, malignancy. Treatment involves cystourethrectomy with PLND. In women, en bloc removal of the urethra, bladder, uterus, adnexa, and anterior vaginal wall is required. In many instances, resection of the pubic symphysis and inferior pubic rami are also necessary. Patients with node-positive disease have an exceptionally poor prognosis despite aggressive surgical management. Therefore, staging laparoscopic PLND spares patients with lymph node involvement the considerable morbidity of anterior pelvic exenteration.
A CT or MRI of the abdomen and pelvis should be obtained prior to surgery in patients at high risk of lymphatic metastases. Percutaneous biopsy in patients with gross adenopathy should be considered because it may preclude the need for surgical intervention. Bone scans are reserved for patients with prostate cancer who have a PSA ≥20 ng/mL and for individuals with prostate or bladder cancer who have an elevated alkaline phosphatase and/or bone pain suspicious for metastatic disease.
A single dose of a first-generation cephalosporin or clindamycin should be given prior to starting the case. Patients scheduled to undergo concurrent prostatectomy or cystectomy should receive a mechanical bowel prep. A full bowel prep may be helpful in patients who are expected to have extensive intra-abdominal adhesions. A type and screen for blood products is appropriate in most cases.
Positioning and Setup
General anesthesia is used. Nitrous oxide is avoided given the risk of bowel distention. An orogastric tube and Foley catheter are placed to decompress the stomach and bladder. The genitalia are wrapped with a compressive dressing to prevent tracking of carbon dioxide into the penis and scrotum.
The patient is placed in the supine position with both arms secured at the side (Fig. 1). Patients undergoing robotic-assisted laparoscopic PLND are placed in the low lithotomy position. All pressure points are adequately padded to prevent compression injury. Extended time in the lithotomy position places the peroneal nerve at increased risk of injury due to its superficial location, which results in a foot drop.
The surgical team is positioned as shown in Figure 2. The anesthesiologist is at the head of the table. The operating surgeon is on the contralateral side of the table from the lymph node packet to be excised. The assistant is opposite the surgeon while the scrub nurse and Mayo stand are placed adjacent and superior to the operating surgeon. The video equipment is placed at the foot of the table. In robotic-assisted cases the Da Vinci patient-side cart (Intuitive Surgical, Sunnyvale, CA) is docked between the patient’s legs. The surgeon is located at the console and the assistant at the patient’s right side.
Port Placement
The abdomen is entered using either a Veress needle or the Hasson technique at the base of the umbilicus. If the Veress needle is employed, the surgeon’s hand or towel clamps are used to elevate the abdominal wall away from the viscera. Proper needle placement is confirmed by observing a low intra-abdominal pressure during initiation of pneumoperitoneum and by the saline “drop test.” Insufflation is continued until an intra-abdominal pressure of 20 mm Hg is achieved and then decreased to 15 mm Hg once the trocars have been placed.
A 1-cm incision is made at the umbilicus. A hemostat is used to separate the subcutaneous fat down to the fascia. A laparoscope with 0-degree camera lens is placed within a 10-mm Visiport (U.S. Surgical, Norwalk, CT) or Optiview (Ethicon Endosurgical, Cincinnati, OH) incising visual
obturator. The camera is focused on the tip of the obturator. The camera and trocar are advanced into the peritoneal cavity under direct vision. After removal of the obturator, a 30-degree lens is placed through the umbilical trocar. The abdominal cavity is inspected for viscus or vascular injury. The abdominal wall is transilluminated with the laparoscope to identify blood vessels that may be injured during placement of the remaining ports.
obturator. The camera is focused on the tip of the obturator. The camera and trocar are advanced into the peritoneal cavity under direct vision. After removal of the obturator, a 30-degree lens is placed through the umbilical trocar. The abdominal cavity is inspected for viscus or vascular injury. The abdominal wall is transilluminated with the laparoscope to identify blood vessels that may be injured during placement of the remaining ports.
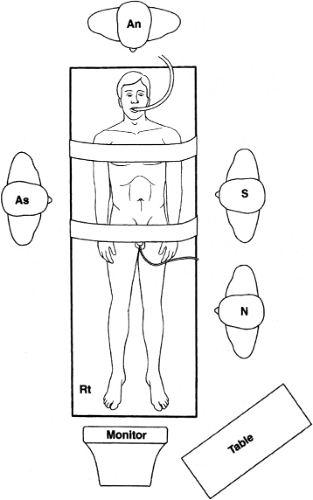 Fig. 2. The surgical team is positioned as depicted for laparoscopic pelvic lymph node dissection. An, anesthesiologist; As, assistant surgeon; N, scrub technician or nurse; S, surgeon. |
The typical port site pattern is illustrated in Figure 3A. A “diamond” configuration is most commonly used. The camera is usually situated at the umbilical port. A trocar is placed approximately four fingerbreadths above the pubic bone along the midline. The lateral trocars are placed one-third of the way from the umbilicus to the anterior superior iliac spine. In obese patients, a “fan” configuration facilitates retraction and dissection (Fig. 3B). For the nonumbilical ports, any combination of 5- and 10-mm ports is acceptable. Following port placement, the patient is placed in the Trendelenburg position to keep the intestines out of the pelvis.
Lymphadenectomy Templates
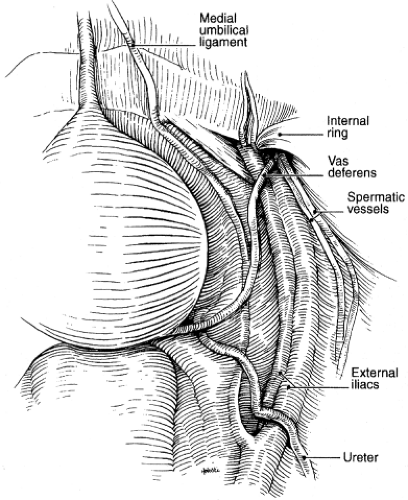
Pertinent anatomic landmarks for PLND are illustrated in Figure 4. The dissection template varies by indication. The standard template boundaries for patients with prostate cancer include the pubic bone distally, the bifurcation of the common iliac vessels proximally, the external iliac vein anteriorly, the obturator nerve posteriorly, the medial umbilical ligament medially, and the pelvic sidewall laterally.
Several recently published observational series support a survival advantage for patients undergoing lymphadenectomy who have micrometastatic nodal involvement. These data, combined with studies showing that up to 50% of nodal metastases occur within the internal iliac chain, which is not included in the standard template dissection, have led some urologists to advocate for an extended PLND in patients with high-risk disease. The limits of the extended lymphadenectomy include the pubic bone distally, the bifurcation of the common iliac vessels proximally, the genitofemoral nerve anterolaterally, the internal iliac artery posteriorly, the medial umbilical ligament medially, and the pelvic sidewall laterally.
The importance of an extended node dissection in patients undergoing radical cystectomy for TCC of the bladder is well established. The limits of the extended dissection for bladder cancer are the same as for prostate cancer except that the dissection is carried up to the aortic bifurcation. The optimal extent of dissection for patients with penile or posterior urethral carcinoma has not been defined. However, most reports have utilized the standard prostate cancer template.
Dissection
After trocar placement, a longitudinal incision is made in the peritoneum lateral to the medial umbilical ligament (Fig. 5). This extends from the pubic symphysis to the iliac bifurcation. On the left side, it is common to encounter adhesions to the sigmoid colon that must first be incised. For patients undergoing concurrent prostatectomy or cystectomy the node dissection may be completed before or after the extirpative portion of the case, depending on surgeon preference.
 Fig. 3. A: “Diamond” configuration for laparoscopic pelvic lymph node dissection port placement. B: “Fan” configuration for port placement in an obese patient. |
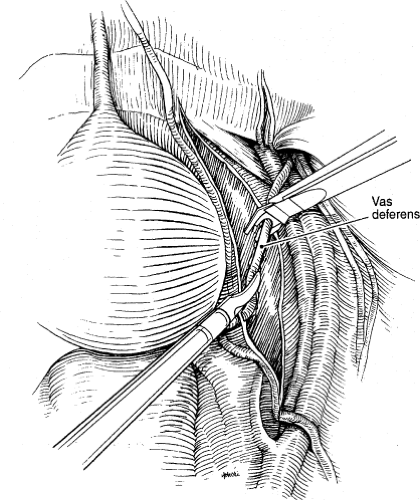
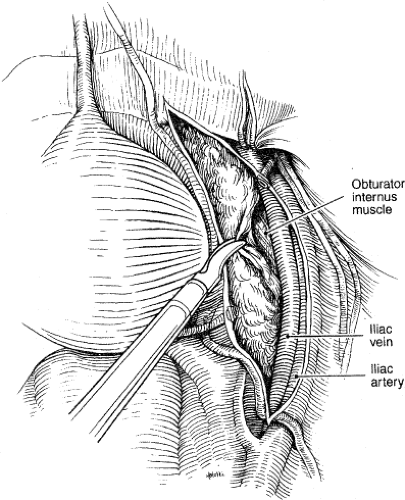
Once the peritoneum is opened, the vas deferens is identified and divided (Fig. 6). Next, the medial edge of the external iliac vein is identified and, by retracting the nodal tissue medially, the nodes are freed from the pubic bone to the bifurcation of the common iliac vessels (Fig. 7). This defines the lateral border of the nodal packet. The nodal tissue is then dissected free from the obturator internus muscle. The dissection proceeds proximally toward the junction of the medial umbilical ligament and the bifurcation of the iliac vessels (Fig. 8). This maneuver exposes the obturator nerve and vessels (Fig. 9). Care must be taken to avoid injury to the ureter, which lies medially at this point.
The medial boundary of dissection is defined by retracting the umbilical ligament medially while pulling the nodal packet laterally. Blunt dissection is usually sufficient to free the nodal tissue along this plane. Distal attachments beneath the pubis are clipped and transected. The entire packet is pulled cranially, and the remaining attachments located at the junction of the medial umbilical ligament and the internal iliac artery are clipped and divided. The specimen can then be extracted using an EndoCatch bag (U.S. Surgical, Norwalk, CT) or a laparoscopic spoon forceps (Fig. 10). The contralateral side is addressed in a similar fashion.
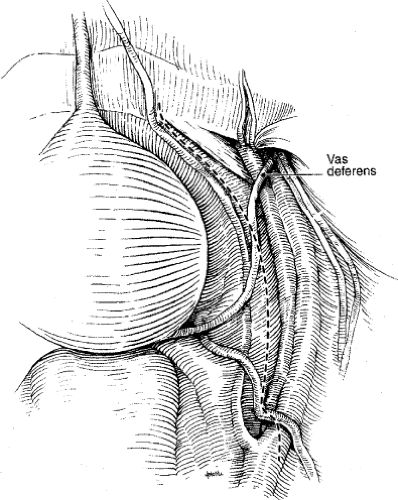 Fig. 5. An incision is made lateral to the medial umbilical ligament. This is extended from the pubic bone to the bifurcation of the common iliac vessels. |
The extended dissection for prostate cancer begins with the external iliac packet. The adventitia overlying the external iliac vein is divided distally near the superior pubic ramus. The nodal tissue is then retracted medially and the dissection is carried proximally to the bifurcation of the common iliac vessels. The fibroalveolar tissue just medial to the genitofemoral nerve is divided and the packet is swept off of the psoas muscle. Clips are usually required along the proximal and distal extents of dissection. The obturator packet is then dissected, as previously described. Dissection of the internal iliac nodal packet is then performed. If not readily apparent after removal of the external iliac and obturator nodal tissue, the origin of the internal iliac artery is identified by tracing the medial umbilical ligament to its origin. The dissection is continued from the origin of the internal iliac vessels down to the floor of the pelvis. Care is taken not to damage the obturator nerve or avulse the obturator vessels. This completes the extended node dissection for patients with prostate cancer (Fig. 11). In patients undergoing a radical cystectomy, the dissection is carried along the common iliac vessels to the aortic bifurcation.
The prostatectomy or cystectomy portion of the case is then performed if indicated.
Upon completion, the pressure within the abdomen is reduced to 5 mm Hg to evaluate for occult sources of bleeding. Hemostasis is achieved using electrocautery, surgical clips (metal or polymer), and hemostatic agents. A final survey of the peritoneal cavity is performed to check for visceral injury. If utilized, the robotic patient-side cart is undocked. The ports are then removed under direct visualization to rule out the onset of abdominal wall bleeding. Closure of the fascia is not required for 5-mm port sites but is for 10-/12-mm ports to prevent hernia formation. The fascia may be repaired extracorporeally following evacuation of the pneumoperitoneum or laparoscopically with the Carter-Thomason (Inlet Medical, Eden Prairie, MN) or Endo Close (U.S. Surgical, Norwalk, CT) devices. Before securing the last trocar site, an effort is made to expel all gas from the peritoneum. The port site skin incisions are closed with absorbable sutures in a subcuticular fashion or with skin glue.
Upon completion, the pressure within the abdomen is reduced to 5 mm Hg to evaluate for occult sources of bleeding. Hemostasis is achieved using electrocautery, surgical clips (metal or polymer), and hemostatic agents. A final survey of the peritoneal cavity is performed to check for visceral injury. If utilized, the robotic patient-side cart is undocked. The ports are then removed under direct visualization to rule out the onset of abdominal wall bleeding. Closure of the fascia is not required for 5-mm port sites but is for 10-/12-mm ports to prevent hernia formation. The fascia may be repaired extracorporeally following evacuation of the pneumoperitoneum or laparoscopically with the Carter-Thomason (Inlet Medical, Eden Prairie, MN) or Endo Close (U.S. Surgical, Norwalk, CT) devices. Before securing the last trocar site, an effort is made to expel all gas from the peritoneum. The port site skin incisions are closed with absorbable sutures in a subcuticular fashion or with skin glue.
For patients undergoing laparoscopic PLND alone, the orogastric tube is removed at the end of the procedure and the Foley catheter is discontinued when the patient is ambulatory. Oral intake is begun the evening of surgery. The length of hospitalization is usually less than 24 hours. Patients undergoing laparoscopic prostatectomy with PLND are typically discharged on postoperative day 1 with the Foley in place for 7 to 10 days. Laparoscopic cystectomy patients have a longer length of stay primarily due to the urinary diversion portion of the case.
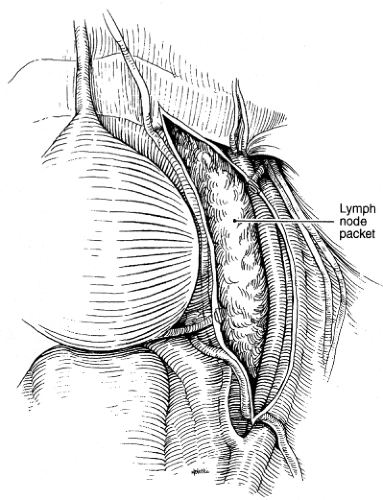 Fig. 7. The lateral edge of the lymph node packet is developed. The medial edge of the iliac vein and the nodal tissue are identified. |
General complications of laparoscopic PLND are similar to those seen in all laparoscopic cases and include ileus, deep vein thrombosis, and urinary retention. Vascular injuries can occur during trocar placement or nodal dissection. Most commonly, the inferior epigastric vessels are injured during lateral trocar placement. The external iliac and obturator vessels can also be injured during nodal dissection. Despite the close proximity of these vessels, vascular injury is rare, occurring in approximately 2% to 3% of cases.
Care must be taken to identify and avoid clipping or transecting the obturator nerve. Injury results in the inability of the patient to adduct the thighs. Injury to the ureter, especially during extended dissections, is also possible. Meticulous hemostasis and attention to anatomical relationships helps in the identification of these structures, thereby minimizing the risk of injury.
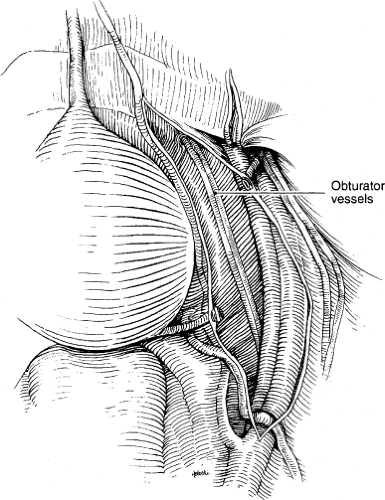 Fig. 9. With the lymphatic tissue dissected free, the obturator nerve vessels should be visible.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|