Intraabdominal Infections
KEY CONCEPTS
![]() Most intraabdominal infections are “secondary” infections that are polymicrobial and are caused by a defect in the GI tract that must be treated by surgical drainage, resection, and/or repair.
Most intraabdominal infections are “secondary” infections that are polymicrobial and are caused by a defect in the GI tract that must be treated by surgical drainage, resection, and/or repair.
![]() Primary peritonitis is generally caused by a single organism (Staphylococcus aureus in patients undergoing chronic ambulatory peritoneal dialysis [CAPD] or Escherichia coli in patients with cirrhosis).
Primary peritonitis is generally caused by a single organism (Staphylococcus aureus in patients undergoing chronic ambulatory peritoneal dialysis [CAPD] or Escherichia coli in patients with cirrhosis).
![]() Secondary intraabdominal infections are usually caused by a mixture of bacteria, including enteric Gram-negative bacilli and anaerobes, which enhance the pathogenic potential of the bacteria.
Secondary intraabdominal infections are usually caused by a mixture of bacteria, including enteric Gram-negative bacilli and anaerobes, which enhance the pathogenic potential of the bacteria.
![]() For peritonitis, early and aggressive IV fluid resuscitation and electrolyte replacement therapy are essential. A common cause of early death is hypovolemic shock caused by inadequate intravascular volume and tissue perfusion.
For peritonitis, early and aggressive IV fluid resuscitation and electrolyte replacement therapy are essential. A common cause of early death is hypovolemic shock caused by inadequate intravascular volume and tissue perfusion.
![]() Treatment is generally initiated on a “presumptive” or empirical basis and should be based on the likely pathogen(s) and local resistance patterns.
Treatment is generally initiated on a “presumptive” or empirical basis and should be based on the likely pathogen(s) and local resistance patterns.
![]() Antimicrobial regimens for secondary intraabdominal infections should include coverage for enteric Gram-negative bacilli and anaerobes. Antimicrobials that may be used for the treatment of secondary intraabdominal infections depending on severity of illness and microbiology data include (a) third-generation cephalosporin (ceftriaxone or cefuroxime) with metronidazole, (b) ticarcillin–clavulanate or piperacillin–tazobactam, (c) a carbapenem (imipenem, meropenem, doripenem, and ertapenem), and (d) quinolone (levofloxacin or ciprofloxacin) plus metronidazole or moxifloxacin alone.
Antimicrobial regimens for secondary intraabdominal infections should include coverage for enteric Gram-negative bacilli and anaerobes. Antimicrobials that may be used for the treatment of secondary intraabdominal infections depending on severity of illness and microbiology data include (a) third-generation cephalosporin (ceftriaxone or cefuroxime) with metronidazole, (b) ticarcillin–clavulanate or piperacillin–tazobactam, (c) a carbapenem (imipenem, meropenem, doripenem, and ertapenem), and (d) quinolone (levofloxacin or ciprofloxacin) plus metronidazole or moxifloxacin alone.
![]() Treatment of patients with peritoneal dialysis-associated peritonitis should include an antistaphylococcal antimicrobial such as a first-generation cephalosporin (cefazolin) or vancomycin (intraperitoneal administration is preferred).
Treatment of patients with peritoneal dialysis-associated peritonitis should include an antistaphylococcal antimicrobial such as a first-generation cephalosporin (cefazolin) or vancomycin (intraperitoneal administration is preferred).
![]() The duration of antimicrobial treatment should be for 4 to 7 days for most secondary intraabdominal infections.
The duration of antimicrobial treatment should be for 4 to 7 days for most secondary intraabdominal infections.
![]() Patients treated for intraabdominal infections should be assessed for the occurrence of drug-related adverse effects, particularly hypersensitivity reactions (β-lactam antimicrobials), diarrhea (most agents), fungal infections (most agents), and nephrotoxicity (aminoglycosides).
Patients treated for intraabdominal infections should be assessed for the occurrence of drug-related adverse effects, particularly hypersensitivity reactions (β-lactam antimicrobials), diarrhea (most agents), fungal infections (most agents), and nephrotoxicity (aminoglycosides).
Intraabdominal infections are those contained within the peritoneal cavity or retroperitoneal space. The peritoneal cavity extends from the undersurface of the diaphragm to the floor of the pelvis and contains the stomach, small bowel, large bowel, liver, gallbladder, and spleen. The duodenum, pancreas, kidneys, adrenal glands, great vessels (aorta and vena cava), and most mesenteric vascular structures reside in the retroperitoneum. Intraabdominal infections may be generalized or localized, complicated or uncomplicated, and community or healthcare-associated. Uncomplicated intraabdominal infections are confined within visceral structures, such as the liver, gallbladder, spleen, pancreas, kidney, or female reproductive organs while complicated intraabdominal infections involve anatomical disruption, extend beyond a single organ, and yield peritonitis and/or abscess. Peritonitis is defined as the acute inflammatory response of the peritoneal lining to microorganisms, chemicals, irradiation, or foreign-body injury. This chapter deals only with peritonitis of infectious origin.
An abscess is a purulent collection of fluid separated from surrounding tissue by a wall consisting of inflammatory cells and adjacent organs. It usually contains necrotic debris, bacteria, and inflammatory cells. These processes differ considerably in presentation and approach to treatment.
EPIDEMIOLOGY
Peritonitis may be classified as primary, secondary, or tertiary.1–5 Primary peritonitis, also called spontaneous bacterial peritonitis, is an infection of the peritoneal cavity without an evident source in the abdomen.6 Bacteria may be transported from the bloodstream to the peritoneal cavity, where the inflammatory process begins. In secondary peritonitis, a focal disease process is evident within the abdomen. Secondary peritonitis may involve perforation of the GI tract (possibly because of ulceration, ischemia, or obstruction), postoperative peritonitis, or posttraumatic peritonitis (blunt or penetrating trauma). Tertiary peritonitis occurs in critically ill patients and is infection that persists or recurs at least 48 hours after apparently adequate management of primary or secondary peritonitis.
![]() Primary peritonitis occurs in both children and adults, although the rates in children have been declining.4 Primary peritonitis develops in up to 10% to 30% of patients with alcoholic cirrhosis.4–7 Patients undergoing chronic ambulatory peritoneal dialysis (CAPD) average one episode of peritonitis every 33 months.8 Epidemiologic data for secondary and tertiary intraabdominal infections are less understood. Secondary peritonitis may be caused by perforation of a peptic ulcer; traumatic perforation of the stomach, small or large bowel, uterus, or urinary bladder; appendicitis; pancreatitis; diverticulitis; bowel infarction; inflammatory bowel disease; cholecystitis; operative contamination of the peritoneum; or diseases of the female genital tract, such as septic abortion, postoperative uterine infection, endometritis, and salpingitis. Appendicitis is one of the most common causes of intraabdominal infection. In 2006, 353,000 appendectomies were performed in the United States for suspected appendicitis.9 Most healthcare-associated intraabdominal infections occur as complications following intraabdominal surgeries.
Primary peritonitis occurs in both children and adults, although the rates in children have been declining.4 Primary peritonitis develops in up to 10% to 30% of patients with alcoholic cirrhosis.4–7 Patients undergoing chronic ambulatory peritoneal dialysis (CAPD) average one episode of peritonitis every 33 months.8 Epidemiologic data for secondary and tertiary intraabdominal infections are less understood. Secondary peritonitis may be caused by perforation of a peptic ulcer; traumatic perforation of the stomach, small or large bowel, uterus, or urinary bladder; appendicitis; pancreatitis; diverticulitis; bowel infarction; inflammatory bowel disease; cholecystitis; operative contamination of the peritoneum; or diseases of the female genital tract, such as septic abortion, postoperative uterine infection, endometritis, and salpingitis. Appendicitis is one of the most common causes of intraabdominal infection. In 2006, 353,000 appendectomies were performed in the United States for suspected appendicitis.9 Most healthcare-associated intraabdominal infections occur as complications following intraabdominal surgeries.
ETIOLOGY
Primary peritonitis in adults occurs most commonly in association with alcoholic cirrhosis, especially in its end stage, or with ascites caused by postnecrotic cirrhosis, chronic active hepatitis, acute viral hepatitis, congestive heart failure, malignancy, systemic lupus erythematosus, or nephritic syndrome. It may also result from the use of a peritoneal catheter for dialysis or CNS ventriculoperitoneal shunting for hydrocephalus. Rarely, primary peritonitis occurs without apparent underlying disease.
Table 92–1 summarizes many of the potential causes of bacterial peritonitis. Causes include inflammatory processes of the GI tract or abdominal organs, bowel obstruction, vascular occlusions that may lead to gangrene of the intestines, and neoplasia that may cause intestinal perforation or obstruction. Other possible causes include those resulting from traumatic injuries, postoperative infections, or solid organ transplant in the abdomen.
TABLE 92-1 Causes of Bacterial Peritonitis

Abscesses are the result of chronic inflammation and may occur without preceding generalized peritonitis. They may be located within one of the spaces of the peritoneal cavity or within one of the visceral organs, and may range from a few milliliters to a liter or more in volume. These collections often have a fibrinous capsule and may take from a few weeks to years to form.
The causes of intraabdominal abscess overlap those of peritonitis and, in fact, may occur sequentially or simultaneously. Appendicitis is the most frequent cause of abscess. Other potential causes of intraabdominal abscess include pancreatitis, diverticulitis, lesions of the biliary tract, genitourinary tract infections, perforation in the abdomen, trauma, and leaking intestinal anastomoses. In addition, pelvic inflammatory disease in women may lead to tuboovarian abscess. For some diseases, such as appendicitis and diverticulitis, abscesses occur more frequently than generalized peritonitis.
Microflora of the Gastrointestinal Tract and Female Genital Tract
A full appreciation of intraabdominal infection requires an understanding of the normal microflora within the GI tract. There are striking differences in bacterial species and concentrations of flora within the various segments of the GI tract (Table 92–2), and this bacterial environment usually determines the severity of infectious processes in the abdomen. Generally, the low gastric pH eradicates bacteria that enter the stomach. With achlorhydria, bacterial counts may rise to 105 to 107 organisms/mL (108 to 1010/L). The normally low bacterial count may also increase by 1,000- or 10,000-fold with gastric outlet obstruction, hemorrhage, gastric cancer, and in patients receiving histamine 2 (H2)-receptor antagonists, proton pump inhibitors, or antacids.
TABLE 92-2 Usual Microflora of the GI Tract

The biliary tract (gallbladder and bile ducts) is sterile in most healthy individuals, but in people older than 70 years of age, those with acute cholecystitis, jaundice, or common bile duct stones, it is likely to be colonized by aerobic Gram-negative bacilli (particularly Escherichia coli and Klebsiella spp.) and enterococci.10 Patients with biliary tract bacterial colonization are at greater risk of intraabdominal infection.
In the distal ileum, bacterial counts of aerobes and anaerobes are quite high. In the colon, there may be 500 to 600 different types of bacteria in stool, with concentrations often reaching 1011 organisms/mL (1014/L) and anaerobic bacteria outnumbering aerobic bacteria by more than 1,000 to 1.2,11 In fact, up to 50% of the dry mass of stool is Bacteroides spp. Fortunately, most colonic bacteria are not pathogens because they cannot survive in environments outside the colon. Perforation of the colon results in the release of large numbers of anaerobic and aerobic bacteria into the peritoneum.
The colonic flora are generally consistent unless broad-spectrum antimicrobials have been used. Depending on the type of antibiotic and spectrum, the duration of use, route of administration, and the pharmacokinetic and pharmacodynamic properties, antibiotics can cause shifts in the normal GI microflora including causing increased drug resistance.12
The lower female genital tract is generally colonized by a large number of aerobic and anaerobic bacteria. Anaerobes may number 109 organisms per milliliter (1012/L) and often include lactobacilli, eubacteria, clostridia, anaerobic streptococci, and, less frequently, Bacteroides fragilis. Aerobic bacteria most often are streptococci and Staphylococcus epidermidis, and these may number 108 organisms per milliliter (1011/L).
PATHOPHYSIOLOGY
Intraabdominal infection results from bacterial entry into the peritoneal or retroperitoneal spaces or from bacterial collections within intraabdominal organs. In primary peritonitis, bacteria may enter the abdomen via the bloodstream or the lymphatic system by transmigration through the bowel wall, through an indwelling peritoneal dialysis catheter, or via the fallopian tubes in females. Hematogenous bacterial spread (through the bloodstream) occurs more frequently with tuberculosis peritonitis or peritonitis associated with cirrhotic ascites. When peritonitis results from peritoneal dialysis, skin surface flora are introduced via the peritoneal catheter. In secondary peritonitis, bacteria most often enter the peritoneum or retroperitoneum as a result of perforation of the GI or female genital tracts caused by diseases or traumatic injuries. In addition, peritonitis or abscess may result from contamination of the peritoneum during a surgical procedure or following anastomotic leak.
The physiologic characteristics of the peritoneal cavity determine the nature of the response to infection or inflammation within it.1,4 The peritoneum is lined by a highly permeable serous membrane with a surface area approximately that of skin. The peritoneal cavity is lubricated with less than 100 mL of sterile, clear yellow fluid, normally with fewer than 250 cells/mm3 (250 × 106/L), a specific gravity below 1.016, and protein content below 3 g/dL (30 g/L). These conditions change drastically with peritoneal infection or inflammation, as described below.
After bacteria are introduced into the peritoneal cavity, there is an immediate response to contain the insult. Humoral and cellular defenses respond first; then the omentum adheres to the affected area. A limited bacterial inoculum is handled rapidly by defense mechanisms, including complement activation and a leukocyte response. Under certain conditions, the bacterial insult is not contained, and bacteria disseminate throughout the peritoneal cavity, resulting in peritonitis. This is more likely to occur in the presence of a foreign body, hematoma, dead tissue, a large bacterial inoculum, continuing bacterial contamination, and contamination involving a mixture of synergistic organisms. Protein–calorie malnutrition, antecedent steroid therapy, and diabetes mellitus may also contribute to the formation of an intraabdominal abscess.
When bacteria become dispersed throughout the peritoneum, the inflammatory process involves most of the peritoneal lining. There is an outpouring into the peritoneum of fluid containing leukocytes, fibrin, and other proteins that form exudates on the inflamed peritoneal surfaces and begin to form adhesions between peritoneal structures. This process, combined with a paralysis of the intestines (ileus), may result in confinement of the contamination to one or more locations within the peritoneum. Fluid also begins to collect in the bowel lumen and wall, and distension may result.
The fluid and protein shift into the abdomen (called third-spacing) may be so dramatic that circulating blood volume is decreased, which may cause decreased cardiac output and hypovolemic shock. Accompanying fever, vomiting, or diarrhea may worsen the fluid imbalance. A reflex sympathetic response, manifested by sweating, tachycardia, and vasoconstriction, may be evident. With an inflamed peritoneum, bacteria and endotoxins are absorbed easily into the bloodstream (translocation), and this may result in septic shock.1,4,5 Other foreign substances present in the peritoneal cavity potentiate peritonitis. These adjuvants, notably feces, dead tissues, barium, mucus, bile, and blood, have detrimental effects on host defense mechanisms, particularly on bacterial phagocytosis.
Many of the manifestations of intraabdominal infections, particularly peritonitis, result from cytokine activity. Inflammatory cytokines, such as tumor necrosis factor-α (TNF-α), interleukin (IL) 1, IL-6, IL-8, and interferon γ (INF-γ), are produced by macrophages and neutrophils in response to bacteria and bacterial products or in response to tissue injury resulting from the surgical incision.1,4 These cytokines produce wide-ranging effects on the vascular endothelium of organs, particularly the liver, lungs, kidneys, and heart. With uncontrolled activation of these mediators, sepsis may result (see Chap. 97, Sepsis and Septic Shock).13,14
Peritonitis may result in death because of the effects on major organ systems. Fluid shifts, cytokines and endotoxin may result in hypovolemia, hypoperfusion, and shock. Hypoalbuminemia may result from protein loss into the peritoneum exacerbating intravascular volume loss. Pulmonary function may be compromised by the inflamed peritoneum, producing splinting (muscle rigidity caused by pain) that inhibits adequate diaphragmatic movement leading to atelectasis and pneumonia. Increased lung vascular permeability and resulting shunting of blood may induce onset of the respiratory distress syndrome and associated hypoxemia and hypercarbia. With fluid loss and hypotension, renal and hepatic perfusion may be compromised, and acute renal and hepatic failure are potential threats.
If peritoneal contamination is localized but bacterial elimination is incomplete, an abscess results. This collection of necrotic tissue, bacteria, and white blood cells may be at single or multiple sites and may be within one of the spaces of the peritoneal cavity or in one of the visceral organs. The location of the abscess is often related to the site of primary disease. For example, abscesses resulting from appendicitis tend to appear in the right lower quadrant or the pelvis; those resulting from diverticulitis tend to appear in the left lower quadrant or pelvis.
An abscess begins by the combined action of inflammatory cells (such as neutrophils), bacteria, fibrin, and other inflammatory mediators. Bacteria may release heparinases that cause local thrombosis and tissue necrosis or fibrinolysins, collagenases, or other enzymes that allow extension of the process into surrounding tissues. Neutrophils gathered in the abscess cavity die in 3 to 5 days, releasing lysosomal enzymes that liquefy the core of the abscess. A mature abscess may have a fibrinous capsule that isolates bacteria and the liquid core from antimicrobials and immunologic defenses.
Within the abscess, the oxygen tension is low and anaerobic bacteria thrive; thus, the size of the abscess may increase because it is hypertonic, resulting in an additional influx of fluid. Hypertonicity promotes the formation of bacterial L forms, which are resistant to antimicrobial agents that disrupt cell walls. Abscess formation may continue and mature for long periods of time and may not be readily evident to either patient or physician. In some instances, the abscess may resolve spontaneously, and, infrequently, it may erode into adjacent organs or rupture and cause diffuse peritonitis. If the abscess erodes through the skin, it may result in an enterocutaneous fistula, connecting bowel to skin, or in a draining sinus tract.
The overall outcome from an intraabdominal infection depends on key factors: inoculum size, virulence of the contaminating organisms, the presence of adjuvants within the peritoneal cavity that facilitate infection, the adequacy of host defenses, source control, and the adequacy of initial treatment.15,16
Microbiology of Intraabdominal Infection
![]() Primary bacterial peritonitis is often caused by a single organism. In children, the pathogen is usually group A Streptococcus, E. coli, Streptococcus pneumoniae, or Bacteroides species.4,17–20 When peritonitis occurs in association with cirrhotic ascites, E. coli is isolated most frequently. Other potential pathogens are: Haemophilus influenzae, Klebsiella spp., Pseudomonas spp., anaerobes, and S. pneumoniae.21 Occasionally, primary peritonitis may be caused by Mycobacterium tuberculosis. Peritonitis in patients undergoing peritoneal dialysis is caused most often by common skin organisms, such as coagulase-negative staphylococci, Staphylococcus aureus, streptococci, and enterococci. Gram-negative bacteria associated with peritoneal dialysis infections include E. coli, Klebsiella spp., and Pseudomonas spp.6 Mortality from primary peritonitis caused by Gram-negative bacteria is much greater than that from Gram-positive bacteria.4,5
Primary bacterial peritonitis is often caused by a single organism. In children, the pathogen is usually group A Streptococcus, E. coli, Streptococcus pneumoniae, or Bacteroides species.4,17–20 When peritonitis occurs in association with cirrhotic ascites, E. coli is isolated most frequently. Other potential pathogens are: Haemophilus influenzae, Klebsiella spp., Pseudomonas spp., anaerobes, and S. pneumoniae.21 Occasionally, primary peritonitis may be caused by Mycobacterium tuberculosis. Peritonitis in patients undergoing peritoneal dialysis is caused most often by common skin organisms, such as coagulase-negative staphylococci, Staphylococcus aureus, streptococci, and enterococci. Gram-negative bacteria associated with peritoneal dialysis infections include E. coli, Klebsiella spp., and Pseudomonas spp.6 Mortality from primary peritonitis caused by Gram-negative bacteria is much greater than that from Gram-positive bacteria.4,5
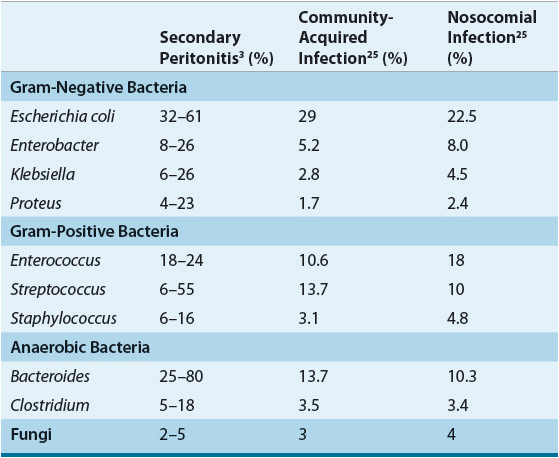
![]() Because of the diverse bacteria present in the GI tract, secondary intraabdominal infections are often polymicrobial.2 The mean number of different bacterial species isolated from infected intraabdominal sites ranged from 2.9 to 3.7, including an average of 1.3 to 1.6 aerobes and 1.7 to 2.1 anaerobes.21,22 With proper anaerobic specimen collection, anaerobic organisms are isolated in most patients. In one report of patients with gangrenous and perforated appendicitis, an average of 10.2 different organisms was isolated from each patient, including 2.7 aerobes and 7.5 anaerobes.23 Purely aerobic or anaerobic infections are uncommon, as are infections caused by fungi. Table 92–3 gives the frequencies with which specific bacteria were isolated from patients with peritonitis and other intraabdominal infections.3,24 Nosocomial infections tend to have a more diverse array of pathogens and higher likelihood of multidrug resistance compared with isolates from community-acquired infections.25 E. coli, Streptococcus spp., and Bacteroides spp. were isolated most often from the infection site, as well as from blood cultures. In patients diagnosed with severe infections, the pattern of bacterial isolates may change and commonly includes Candida spp., enterococci, Enterobacteriaceae, and S. epidermidis.
Because of the diverse bacteria present in the GI tract, secondary intraabdominal infections are often polymicrobial.2 The mean number of different bacterial species isolated from infected intraabdominal sites ranged from 2.9 to 3.7, including an average of 1.3 to 1.6 aerobes and 1.7 to 2.1 anaerobes.21,22 With proper anaerobic specimen collection, anaerobic organisms are isolated in most patients. In one report of patients with gangrenous and perforated appendicitis, an average of 10.2 different organisms was isolated from each patient, including 2.7 aerobes and 7.5 anaerobes.23 Purely aerobic or anaerobic infections are uncommon, as are infections caused by fungi. Table 92–3 gives the frequencies with which specific bacteria were isolated from patients with peritonitis and other intraabdominal infections.3,24 Nosocomial infections tend to have a more diverse array of pathogens and higher likelihood of multidrug resistance compared with isolates from community-acquired infections.25 E. coli, Streptococcus spp., and Bacteroides spp. were isolated most often from the infection site, as well as from blood cultures. In patients diagnosed with severe infections, the pattern of bacterial isolates may change and commonly includes Candida spp., enterococci, Enterobacteriaceae, and S. epidermidis.
TABLE 92-3 Pathogens Isolated from Patients with Intraabdominal Infection
Visceral organ abscesses differ in character from the typical intraabdominal abscess. Hepatic abscesses may be polymicrobial (involving E. coli, Klebsiella spp., and anaerobes) or occasionally may be caused by amoeba.11 Pancreatic abscesses are often polymicrobial, involving enteric bacteria that ascend through the biliary system. Splenic abscesses usually result from hematogenous dissemination of bacteria, such as E. coli, S. aureus, Proteus mirabilis, Enterococcus spp., and K. pneumoniae, as well as anaerobes.11 Pelvic inflammatory disease is associated initially with Neisseria gonorrhoeae or Chlamydia trachomatis. However, tuboovarian abscesses are usually polymicrobial, having a mix of Gram-positive and Gram-negative aerobes and anaerobes.
Bacterial Synergism
The size of the bacterial inoculum and the number and types of bacterial species present in intraabdominal infections influence patient outcome. The combination of aerobic and anaerobic organisms appears to greatly increase the severity of infection. In animal studies, combinations of aerobic and anaerobic bacteria were much more lethal than infections caused by aerobes or anaerobes alone.
Facultative bacteria may provide an environment conducive to the growth of anaerobic bacteria.2 Although many bacteria isolated in mixed infections are nonpathogenic by themselves, their presence may be essential for the pathogenicity of the bacterial mixture.7 The role of facultative bacteria in mixed infections can include (a) promotion of an appropriate environment for anaerobic bacterial growth through oxygen consumption, (b) production of nutrients necessary for anaerobes, and (c) production of extracellular enzymes that promote tissue invasion by anaerobes.
Rat models of intraabdominal infection demonstrate that uncontrolled infection with an implanted mix of aerobes and anaerobes leads to a two-stage (biphasic) infectious process. There is an early peritonitis phase with a high mortality rate and isolation of E. coli from blood and a late abscess formation phase in all survivors with isolation of anaerobes such as B. fragilis and Fusobacterium varium. These experiments and others support the concept that aerobic enteric organisms and anaerobes are pathogens in intraabdominal infection. Aerobic bacteria, particularly E. coli, appear responsible for the early mortality from peritonitis, whereas anaerobic bacteria are major pathogens in abscesses, with B. fragilis predominating.26
Enterococcus can be isolated from many intraabdominal infections in humans, but its role as a pathogen is not clear. Enterococcal infection occurs more commonly in postoperative peritonitis, in the presence of specific risk factors indicating failure of the host’s defenses (immunocompromised patients), or with the use of broad-spectrum antibiotics.27,28
CLINICAL PRESENTATION
Intraabdominal infections have a wide spectrum of clinical features often depending on the specific disease process, the location and magnitude of bacterial contamination, and concurrent host factors. Peritonitis is usually recognized easily, but intraabdominal abscess may often continue for considerable periods of time, either going unrecognized or being attributed to an unrelated disease process. Patients with primary and secondary peritonitis present quite differently (Table 92–4).1,4,5
TABLE 92-4 Clinical Presentation of Peritonitis




