Injury to Extremities
Todd E. Rasmusen
Adam Stannard
Introduction
More than any other body region, topics related to extremity injury are expansive. This is because of their exposed position and greater survivability from wounding compared to torso and head injury. The incidence and management of extremity trauma in civilian and wartime settings has been well documented over time. The etiology related to extremity trauma is diverse and the types and patterns of resultant limb injury wide-ranging. The complexity of extremity injury portends difficulty in management; however, a simplified approach consists of considering anatomic components first separately and then together in the context of the mangled extremity.
This chapter will address these aspects of extremity injury beginning with a review of epidemiology followed by discussions focused on each of the anatomic components of the injured extremity: vascular, soft tissue, bone, and nerve. Finally, the chapter will combine these components in the context of the mangled extremity, limb salvage, and outcomes. Of the four anatomic components, this chapter will place particular emphasis on vascular and soft-tissue injury as these are more germane to the daily practice of general surgeons and details related to fracture and peripheral nerve injury are more appropriately gleaned from other sources.
Epidemiology
Demographics and Mechanism
Extremity injury is predominantly the preserve of young men (20 to 40 years old) who undertake higher-risk activities. When considering all body regions (extremity, torso, head, and neck), extremity trauma is most common and comprises 70% to 80% of injuries. In civilian practice, blunt injuries predominate in around 85% of cases dependent upon the country being considered, whereas in military conflicts, >90% of limb injuries are a result of a penetrating mechanism. While the rate of amputation due to trauma in civilian practice has been calculated as 1.4/100,000 population, 2,000/100,000 of battle-related injuries result in amputation reflecting the high-energy penetrating mechanisms encountered. As each anatomic component of the extremity can be impacted uniquely by different mechanisms, it is useful to also consider how the injury occurred. Mechanisms of injury are divided into high versus low energy and penetrating versus blunt.
In the setting of penetrating injury, force is a function of the kinetic energy of the projectile (KE = ½mv2) and energy transfer, which is proportional to the density of the tissue through which the projectile passes. In the limb, bone and skeletal muscle are dense and therefore energy transfer through these tissues is high. This means that in the setting of high-energy wounding, anatomic structures remote from the projectile path (e.g., blood vessels and nerves) are at risk for injury. This is in contrast to low-energy penetrating wounds, such as stab wounds, where the injured tissues are typically limited to those directly contacted by the projectile.
Blunt extremity trauma can also be high energy, especially in the combat setting following blast or in the civilian setting following high-speed automotive crashes or falls from heights. The energy transfer from a blunt injury can be discerned based on the presence or absence of associated fractures. For example, blunt extremity injury resulting in a femur fracture indicates a high-energy mechanism, whereas blunt injury resulting in no fractures is likely to have been low energy. One exception is posterior dislocation of the knee, which can occur without a specific or defined fracture, but requires a high-energy mechanism.
Civilian Versus Military Injury
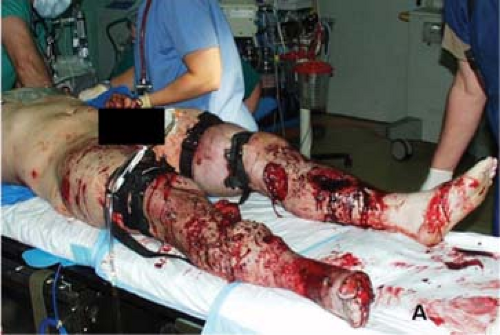
Features of extremity trauma vary depending upon whether the injury occurs in a civilian setting or on the battlefield. Commonly, low-energy extremity injuries are seen and treated in the civilian trauma setting and occur equally from blunt and penetrating mechanisms. In contrast, high-energy penetrating wounds (i.e., explosive or high-energy rounds of various munitions) are more common on the battlefield and thus much more likely to affect multiple components of the four anatomic elements (Fig. 1). In addition to the frequency of concomitant vascular, soft tissue, nerve, and bone damage, extremity trauma in combat is often associated with injury to other body regions, resulting in an associated high Injury Severity Scores (ISS). As an example of the contrast between wounding patterns from civilian and military settings, the rate of vascular injury in civilian trauma centers is 0.2% to 4%, whereas recent data from the wars in Iraq and Afghanistan show the rate to be 12% in modern combat. Also, as an illustration of the high-energy nature of wartime injuries, 35% of extremity vascular injuries in combat have concomitant fractures.
Anatomic Distribution
With regards to the anatomic distribution of extremity injuries, the lower limbs are far more likely to be injured compared to the upper extremities (75% vs. 25%, respectively). The increased frequency of lower limb injuries reflects the greater length and surface area of the legs compared to the arms and the fact that the lower limbs are usually subject to the greatest energy exposure, be that in a motor vehicle collision in civilian practice or the result of an explosive device on the battlefield. The pattern of lower versus upper limb injury holds true when focusing just on extremity vascular trauma as well, with three times the number of lower extremity vascular injuries reported compared to those in the arms. In the lower extremity, there is an equal distribution of vascular injuries between the femoropopliteal and tibial levels.
A full account of the anatomy of the upper and lower limbs is beyond the scope of this chapter. However, some key anatomical points do merit discussion. Neurovascular bundles tend to be superficial in the limbs and may be traumatized by relatively trivial injuries. In young healthy individuals, there tends to be limited collateral circulation so that injury to an axial vessel in the extremity may result in significant ischemia and limb loss. In contrast, age-related occlusive disease typically occurs over time allowing the development of collateral circulation such that eventual occlusion of that axial vessel results in relative, but not limb-threatening, degrees of ischemia. Collateral circulation is best developed and most prominent in the areas of the extremity with high muscle mass (e.g., the thigh and leg). Therefore, when injury occurs to an axial artery in an area of the extremity with limited musculature, such as the popliteal fossa, it often poses a significant threat to the limb. Finally, the presence of fascial compartments in the upper and lower extremities results in fixed potential volumes or spaces in which the major muscle complexes reside. The result of these confined partitions is that any swelling within these spaces may result in increased pressure and the development of compartment syndrome, a topic which is discussed in more detail in the following sections.
Extremity injuries may present in isolation or in the presence of multiregional injuries, depending upon the cause. In low-energy mechanisms, such as stab wounds, it is likely that soft-tissue injury will be minimal and collateral circulation preserved. In high-energy injuries, such as those resulting from explosive devices or high-velocity gunshot wounds, soft-tissue injuries will be more extensive often leading to damage of collateral circulation. High-energy extremity injury often results in what is termed a “mangled extremity” or one in which multiple components, such as bone, nerve, vessel, and soft tissue, are affected (Fig. 1). At the extreme, 2% to 3% of those exposed to a blast injury in modern combat suffer a traumatic amputation. Higher-energy mechanisms also result in more extensive injuries affecting other body regions and result in a more profound systemic injury. As such, priorities for initial management and intervention on the extremity will largely be dictated by the presence of other life-threatening injuries. Recognition of other life-threatening injuries and their early aggressive management will independently impact the outcome of the extremity injury. For example, hemorrhagic shock has recently been shown to reduce neuromuscular recovery of the limb following extremity vascular injury.
Peripheral vascular injuries may present in a number of ways, depending upon the mechanism, type, and location of vessel injury. In addition, the time between the injurious event and the arrival to a care facility will influence the presentation of the injury to the attending surgeon. In the periphery, the two most likely aspects of clinical presentation include hemorrhage and then ischemia. In high-energy wounds, extensive tissue loss bleeding may be apparent, although in low-energy mechanisms with relatively intact tissue plains, tamponade renders hemorrhage an unreliable sign of vessel injury. In addition, an expanding hematoma is considered a hard sign of underlying vascular injury.
As the clinical presentation of vascular injury is variable, it is vital that as much information concerning the timing and mechanism is gathered in order to best estimate the location and severity of the injury. A description of the scene of injury and prehospital care relating to hemorrhage from the limb is important, as bleeding from a vascular injury may have stopped because of hypotension and thrombosis by the time the patient arrives to the care facility.
Nerve injury can usually be grossly identified in a casualty who is conscious, communicating appropriately and thus able to be examined reliably. However, this type of assessment may be difficult in those who are unconscious or have an altered state of awareness because of injury related factors or medications. It is vital that neurological findings are documented as early as possible as interventions, such as nerve blocks to control pain, may render subsequent findings unreliable. Also a change in neurological findings from baseline may suggest an ischemic injury with developing neurological signs.
In the mangled extremity, diagnosis of bone injuries may be obvious on inspection or with subsequent radiographs. Soft-tissue injuries will be apparent although their extent may not initially be appreciated particularly in high-energy cavitating wounds, which underscores the importance of clear history taking and thorough clinical examination. Neurological symptoms may be initially apparent although further examination and exploration may be required to differentiate between the types of peripheral nerve injuries as described earlier, this in turn will dictate further management. The insidious onset of neurological symptoms should raise the suspicion of a vascular injury with progressive ischemia or following reperfusion, the development of compartment syndrome.
Signs and symptoms of arterial injury have been classically defined as either hard or soft in their nature. Hard signs include observed pulsatile bleeding, absent distal pulse, expanding hematoma, and arterial thrill or bruit over or close to the site of suspected arterial injury. Soft signs include a history of significant hemorrhage, diminished distal pulse compared to the contralateral side, distal neurological abnormalities, and the proximity of a vessel to the wound or bone fragments. In addition, there are associations of vascular injury with orthopedic injuries (Table 1), which should lower the threshold of suspicion for injury. In children, a supracondylar fracture of the humerus may lead to injury of the brachial artery and a resultant Volkmann’s contracture; elbow dislocation may injure the brachial artery. In the lower limb, distal femoral fractures may cause superficial femoral artery (SFA) and popliteal artery injuries, while posterior dislocation of the knee may affect the popliteal artery, and tibial fractures damage the popliteal and crural vessels. Significant fractures of the tibial plateau have a particular association with injury to the distal popliteal and tibial-peroneal trunk arterial segments.
A detailed physical examination is mandatory to assess all patients. This includes a full vascular examination including ankle brachial index (ABI). As an objective, readily available, noninvasive method of evaluation, the role of the ABI/Doppler cannot be over emphasized. Certainly, all casualties with soft signs should undergo an ABI as it will guide the next phase of assessment; if it is <1.0, an arterial injury will need to be excluded using more refined techniques. Also, a baseline ABI will give an objective baseline for those whose management is initially planned as conservative, be that due to the presence of other more severe injuries elsewhere, or because there is clinical suspicion of an injury but no objective evidence initially.
Table 1 Vascular Injuries Commonly Associated with Orthopedic Injuries | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
As already discussed, it is now known that the complications from ischemic limb injuries are proportional to the ischemic time and all efforts should be made to reperfuse the limb within 3 hours. It is during this initial phase of assessment that the decision needs to be made whether to proceed directly to the operating room (OR) with an on-table angiogram providing additional detail of the constellation of vascular injuries while soft tissue, nerve, and bone injuries
can simultaneously be explored to diagnose the extent of injury, or to perform additional investigations prior to operating.
can simultaneously be explored to diagnose the extent of injury, or to perform additional investigations prior to operating.
In the presence of hard signs, it is appropriate to proceed directly to the OR without further workup (i.e., in the presence of hemorrhage, expanding hematoma, and/or severe ischemia). However, hard signs are only present in a minority of vascular injuries; therefore, a strategy to stratify risk of vascular injury is needed. This allows resources to be used appropriately to investigate aggressively using invasive techniques, investigate further using noninvasive techniques, or treat conservatively with a period of observation. This strategy is described as selective angiography. Multiple studies have addressed the question of risk stratification and gradually consensus has developed, at its core is measurement of ABI and pulse examination. If the ABI is 1.0 or above and there is no pulse deficit, then the risk of vascular injury is defined as low. A number of studies have demonstrated no complications or need for surgical intervention for this group when the intention to treat was conservative. A combination of an ABI of <1.0 and soft signs of vascular injury are ascribed as an intermediate risk by a number of authors to give better resolution when assessing the risk of vascular injury; however, in practical terms in nearly all instances in which the ABI is measured at <1.0, the extremity should be considered a high-risk group and additional imaging obtained (i.e., angiography).
The three main modalities used in imaging vascular injuries are angiography, computed tomographic angiography (CTA), and color-flow duplex (CFD) ultrasonography. The gold standard for many years has been angiography, now enhanced by the use of high-resolution CTA. This modality provides rapid and detailed information on extremity blood vessels; however, its availability and quality may be institution dependent. It should be emphasized that either form of angiography is an invasive test and the use of contrast media means neither is without risk to the patient (e.g., risk of contrast media to renal function or anaphylaxis). As such, the practice of selective angiography based on noninvasive imaging as described above is advisable. CTA has a high sensitivity (93%) and specificity (95%) in this setting. If anything, CTA is more likely to overestimate the significance of a lesion.
Unfortunately, in the context of trauma, the noninvasive, contrast-free advantages of magnetic resonance imaging (MRI) cannot be applied due to the time required to complete the examination, the presence, or potential presence of metallic foreign bodies, and availability of the scanner. CFD does have the advantage over angiography of being noninvasive and able to be applied at the bedside without the need for contrast media. Several studies describe sensitivity and specificity of duplex as equivalent to CTA; however, as with all forms of ultrasonography, it is operator dependent and unless there is a well-established service locally, these excellent results may be diluted. Certainly, there is a case to promote the development of duplex ultrasound skills amongst surgeons so that they may perform their own exams as necessary. Duplex is generally the investigation of choice in pediatric patients with extremity injuries.
The assessment of compartment syndrome starts with a high index of suspicion for any casualty who is at risk, for example, following ischemia-reperfusion injury or blunt/crush injuries to the limb. The most evident symptom is pain described as being out of proportion to the injury. The most important signs are distal motor and sensory nerve deficits, typically loss of sensation in the first web space of the hand and reduced power of pronation (median nerve), or reduced sensation in the first web space of the foot with reduced power of ankle dorsiflexion (peroneal nerve). On examination, the compartments feel tense with painful passive flexion/extension of the wrist and ankle. The loss of a distal pulse may never occur since the compartment pressure required to create a compartment syndrome is well below arterial pressure. In a number of circumstances, the subjective feedback used in the assessment of compartment syndrome may not be available (e.g., unconscious casualty or following major neurological injury). Under these circumstances, an objective measure of compartment syndrome is required. The first objective test is to measure the pressure within the compartment using pressure transduction. A number of specialist manometers have been developed to do this; however, the basic principles are the same. The pressure is recorded within the compartment and compared to diastolic blood pressure. If the discrepancy in pressure is less than 20 to 30 mm Hg, then fasciotomy is recommended; however, measurement is at times variable. Therefore, if compartment syndrome is considered despite a measure of compartment pressure below this threshold, fasciotomy should be performed. In addition, some authors have looked at venous outflow obstruction as a surrogate measure of compartment syndrome. While CFD ultrasound examination of the venous outflow vessels cannot confirm compartment syndrome, it has been reported that normal flow rules out compartment syndrome. However, it should be remembered that if compartment syndrome is suspected clinically, fasciotomies should not be delayed in order to confirm with objective testing.
Once the diagnoses of the individual components of the mangled extremity injury have been established, it is possible to stratify the degree of injury using the mangled extremity severity score (MESS).
Skeletal/Soft-Tissue Injury
Low energy (stab, simple fracture, pistol gunshot wound): 1
Medium energy (open or multiple fractures, dislocation): 2
High energy (high-speed motor vehicle accident or rifle gunshot wound): 3
Very high energy (high-speed trauma + gross contamination): 4
Limb Ischemia
Pulse reduced or absent but perfusion normal: 1*
Pulseless; paresthesias, diminished capillary refill: 2
Cool, paralyzed, insensate, numb: 3*
Shock
Systolic BP always >90 mm Hg: 0
Hypotensive transiently: 1
Persistent hypotension: 2
Age (Years)
<30: 0
30 to 50: 1
>50: 2
*Score doubled for ischemia >6 hours
The utility of the MESS is in part controversial. However, most authors now conclude that a low MESS <4 is predictive of the ability to salvage the limb and that a high MESS >7 is predictive of a requirement for amputation. Limbs which score between 4 and 7 will have variable outcome depending on individual circumstance, with soft-tissue and bone injuries providing the major influence on limb salvage. Therefore, appropriate MESS scoring may influence the treatment of the mangled extremity particularly in the presence of additional life-threatening injuries where resources and time are limited.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree