Gastrointestinal Infections and Enterotoxigenic Poisonings
KEY CONCEPTS
![]() The etiology of infectious diarrhea includes bacteria, viruses, and protozoans. Viral infections are the leading cause of diarrhea in the world.
The etiology of infectious diarrhea includes bacteria, viruses, and protozoans. Viral infections are the leading cause of diarrhea in the world.
![]() The common pathogens responsible for watery diarrhea are norovirus and enterotoxigenic Escherichia coli. The pathogens that produce dysentery or bloody diarrhea are Shigella spp., Campylobacter jejuni, nontyphoid Salmonella, and enterohemorrhagic E. coli.
The common pathogens responsible for watery diarrhea are norovirus and enterotoxigenic Escherichia coli. The pathogens that produce dysentery or bloody diarrhea are Shigella spp., Campylobacter jejuni, nontyphoid Salmonella, and enterohemorrhagic E. coli.
![]() Fluid and electrolyte replacement is the cornerstone of therapy for diarrheal illnesses. Oral rehydration therapy is preferred in most cases of mild and moderate diarrhea.
Fluid and electrolyte replacement is the cornerstone of therapy for diarrheal illnesses. Oral rehydration therapy is preferred in most cases of mild and moderate diarrhea.
![]() Antibacterial therapy often is not indicated for gastroenteritis because many cases are mild and self-limited or are viral in nature. Antibiotic therapy is recommended in severe cases of diarrhea, moderate-to-severe cases of traveler’s diarrhea, most cases of febrile dysenteric diarrhea, and culture-proven bacterial diarrhea.
Antibacterial therapy often is not indicated for gastroenteritis because many cases are mild and self-limited or are viral in nature. Antibiotic therapy is recommended in severe cases of diarrhea, moderate-to-severe cases of traveler’s diarrhea, most cases of febrile dysenteric diarrhea, and culture-proven bacterial diarrhea.
![]() Loperamide offers symptomatic relief in patients with moderate watery diarrhea. However, the use of antimotility agents should be avoided in patients with dysentery diarrhea.
Loperamide offers symptomatic relief in patients with moderate watery diarrhea. However, the use of antimotility agents should be avoided in patients with dysentery diarrhea.
![]() Diarrheal illness can be largely prevented by following simple rules of personal hygiene and safe food preparation.
Diarrheal illness can be largely prevented by following simple rules of personal hygiene and safe food preparation.
![]() Metronidazole is the drug of choice for mild to moderate Clostridium difficile infection (CDI). In patients with severe disease, contraindication or intolerance to metronidazole, and inadequate response to metronidazole, oral vancomycin or fidaxomicin, is recommended.
Metronidazole is the drug of choice for mild to moderate Clostridium difficile infection (CDI). In patients with severe disease, contraindication or intolerance to metronidazole, and inadequate response to metronidazole, oral vancomycin or fidaxomicin, is recommended.
![]() The most common pathogens for traveler’s diarrhea include enterotoxigenic E. coli, Shigella, Campylobacter, Salmonella, and viruses.
The most common pathogens for traveler’s diarrhea include enterotoxigenic E. coli, Shigella, Campylobacter, Salmonella, and viruses.
![]() Patient education in prevention strategies and self-treatment of traveler’s diarrhea is recommended. Prophylaxis with antibiotics is not recommended in most situations.
Patient education in prevention strategies and self-treatment of traveler’s diarrhea is recommended. Prophylaxis with antibiotics is not recommended in most situations.
![]() Common pathogens responsible for food poisoning include Staphylococcus, Salmonella, Shigella, and Clostridium.
Common pathogens responsible for food poisoning include Staphylococcus, Salmonella, Shigella, and Clostridium.
GI infections and enterotoxigenic poisonings encompass a wide variety of medical conditions characterized by inflammation of the GI tract. Vomiting and diarrhea are commonly associated with GI inflammation. The resulting dehydration is responsible for much of the morbidity and mortality. Diarrhea is defined as a decrease in consistency of bowel movements (i.e., unformed stool) and an increase in frequency of stools to ≥3 per day.1 Acute disease is commonly associated with diarrhea lasting ≤14 days in duration while persistent diarrhea lasts >14 days.
This chapter focuses on infectious etiologies of acute GI infections and enterotoxigenic poisonings. A wide variety of viral, bacterial, and parasitic pathogens are responsible for this disease. Chapter 93 discusses the common protozoans that cause gasteroenteritis. Therefore, this chapter focuses on common viral and bacterial etiologies. Because the clinical consequences of bloody or dysenteric diarrhea can be more severe compared with cases of watery or nonbloody diarrhea, the chapter is organized accordingly. Epidemiology, clinical presentation, diagnosis, treatment, and prevention strategies are discussed for all GI infections, and further elaborated in subsequent sections in regards to specific diseases such as Clostridium difficile–associated diarrhea, traveler’s diarrhea, and foodborne illnesses.
EPIDEMIOLOGY
Dehydration resulting from acute diarrhea is the second leading cause of morbidity and mortality worldwide, and infants and children younger than 5 years of age are at the highest risk. Among the 139 low- to middle-income countries, there were approximately 1.7 billion episodes of childhood diarrhea in 2010. The incidence of diarrhea for all children younger than age 5 years was estimated to be 2.9 episodes per child per year. The incidence of diarrhea was higher in younger children, with 4.5 episodes per child per year among children aged 6 to 11 months, compared with 2.3 episodes per child per year for children 24 to 59 months of age.2 Younger children also had a higher risk of death from acute dehydrating diarrhea. For children younger than age 1 year and those aged 1 to 4 years, the median mortality rates were 8.5 and 3.8 per 1,000 children per year, respectively.3 Although incidence of childhood diarrhea has been declining, diarrhea remains a major health problem in children, especially in those younger than 1 year.
In the United States, 179 million episodes of acute gastroenteritis occur each year, resulting in more than 600,000 hospitalizations and more than 5,000 deaths.4,5 In contrast to the developing world where the risk of death is highest among young children, most of those who die of diarrheal illness in the United States are elderly. During 1979 to 1987, 51% of deaths caused by diarrheal illness were among patients older than age 74 years, and 27% were among the ages of 55 to 74 years; only 11% of deaths were in those younger than age 5 years.6 Similarly, a study of the McDonnell-Douglas Health Information System database revealed that 25% of all hospitalizations and 85% of all mortality associated with diarrhea involved the elderly (≥age 60 years).4 In addition to children and elderly, other groups at risk for GI infections include travelers and campers, patients in chronic care facilities, military personnel assigned overseas, and immunocompromised patients such as those with acquired immunodeficiency syndrome (AIDS).
Etiology
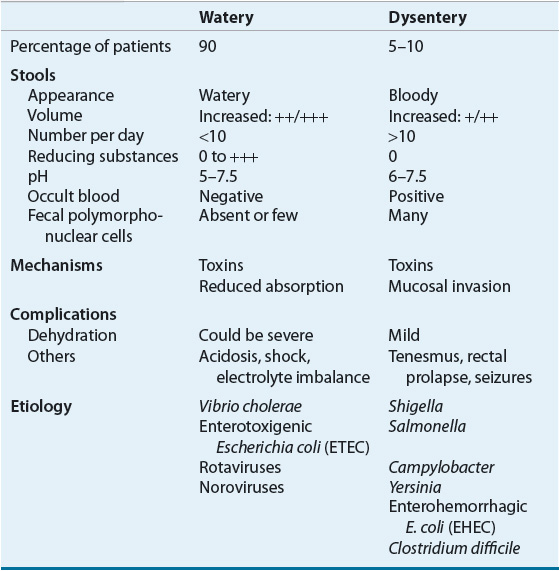
![]() The etiology of GI infections and enterotoxigenic poisonings includes a wide variety of virus, bacteria, and parasites, but contribution of each is unknown. Etiologic agents are rarely identified due to the infrequency of stool samples collected, or inability of many laboratories to detect the full range of pathogens, especially viruses. In this chapter, discussions of pathogens responsible for watery diarrhea focus on virus (rotavirus and norovirus), enterotoxigenic Escherichia coli (ETEC), and cholera. Pathogens commonly associated with dysenteric or bloody diarrhea are Shigella spp., Salmonella spp., Campylobacter spp., enterohemorrhagic E. coli (EHEC), Yersinia enterocolitica, and C. difficile. Characteristics of watery and dysenteric diarrhea and common pathogens responsible for them are outlined in Table 91–1.
The etiology of GI infections and enterotoxigenic poisonings includes a wide variety of virus, bacteria, and parasites, but contribution of each is unknown. Etiologic agents are rarely identified due to the infrequency of stool samples collected, or inability of many laboratories to detect the full range of pathogens, especially viruses. In this chapter, discussions of pathogens responsible for watery diarrhea focus on virus (rotavirus and norovirus), enterotoxigenic Escherichia coli (ETEC), and cholera. Pathogens commonly associated with dysenteric or bloody diarrhea are Shigella spp., Salmonella spp., Campylobacter spp., enterohemorrhagic E. coli (EHEC), Yersinia enterocolitica, and C. difficile. Characteristics of watery and dysenteric diarrhea and common pathogens responsible for them are outlined in Table 91–1.
TABLE 91-1 Acute Infectious Diarrhea Clinical Syndromes: Watery versus Dysentery
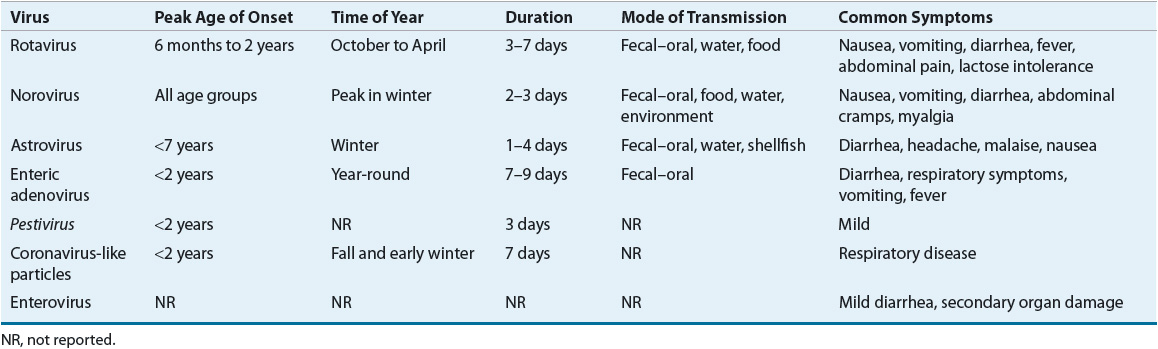
Viruses are now recognized as the leading cause of diarrhea in the world. In Asia, Africa, and Latin America, viral gastroenteritis accounts for 3 to 5 billion cases and is associated with 5 to 10 million deaths.7 Noroviruses, previously known as Norwalk-like viruses, account for greater than 90% of viral gastroenteritis among all age groups and 50% of outbreaks worldwide. In the United States, noroviruses have been estimated to cause 21 million cases of acute gastroenteritis annually including >70,000 hospitalizations and nearly 800 deaths.5 Outbreaks occur throughout the year and have been documented in families, healthcare systems, cruise ships, and college dormitories.
In infants and children, rotavirus, a double-stranded, wheel-shaped, RNA virus, is the most common cause of diarrhea worldwide, and 1 million people die annually from the infection. In the United States, approximately 3.5 million cases of diarrhea, 500,000 physician visits, 50,000 hospitalizations, and 20 deaths occur each year in children younger than age 5 years.7 Nearly all children are infected by age 5 years. After the initial infection with rotavirus, 40% of children are protected against subsequent infection, 75% are protected against subsequent gastroenteritis, and up to 88% are protected against severe gastroenteritis. Other viruses, enteric adenovirus, and astrovirus, coronaviruses, enteroviruses, and pestiviruses, are being identified increasingly as causative agents of diarrhea. Characteristics of viral pathogens causing gastroenteritis are outlined in Table 91–2.
TABLE 91-2 Characteristics of Agents Responsible for Acute Viral Gastroenteritis
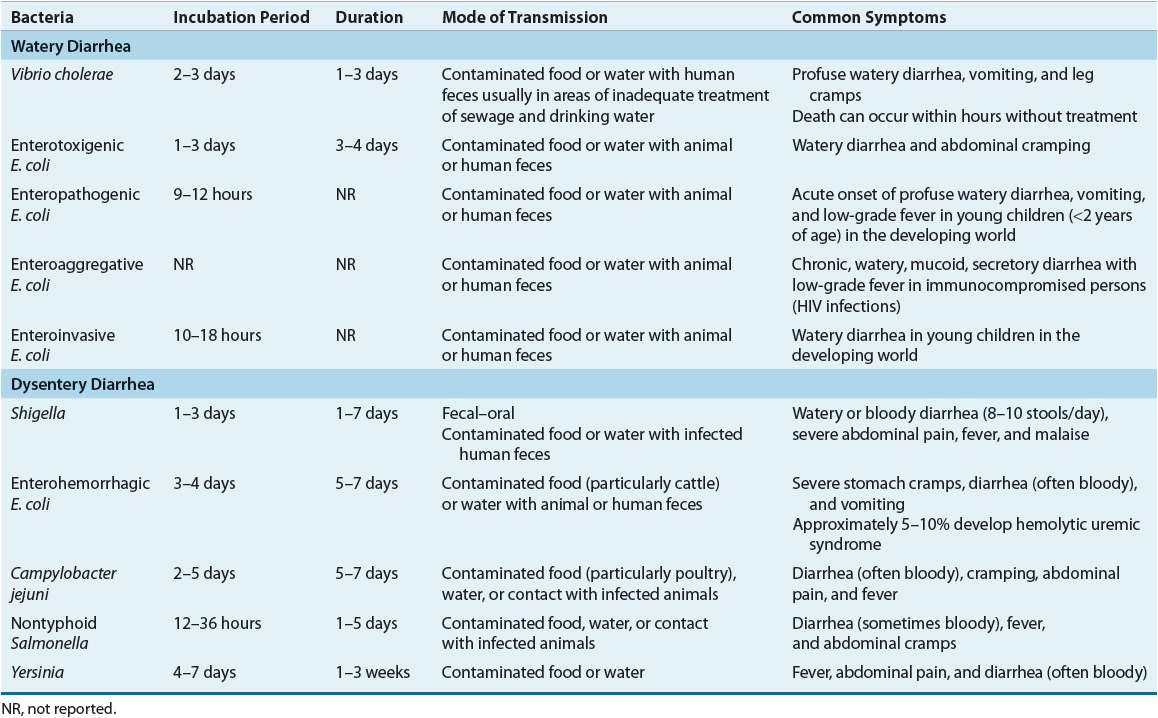
![]() In the United States, bacterial causes of acute gastroenteritis account for more than 5.2 million cases of diarrhea annually including 46,000 hospitalizations and 1,500 deaths.4 However, there appears to be substantial underreporting and the cause is identified in less than 3% of cases. In the United States, common pathogens responsible for watery diarrhea are norovirus and ETEC while those associated with dysentery or bloody diarrhea are Shigella spp. (15.3%), Campylobacter spp. (6.2%), Salmonella spp. (5.8%), EHEC (2.6%), and others (0.6%).8 Other organisms that are responsible for dysentery include Aeromonas species, noncholera Vibrio, and Y. enterocolitica. Characteristics of acute bacterial pathogens causing gastroenteritis are summarized in Table 91–3.
In the United States, bacterial causes of acute gastroenteritis account for more than 5.2 million cases of diarrhea annually including 46,000 hospitalizations and 1,500 deaths.4 However, there appears to be substantial underreporting and the cause is identified in less than 3% of cases. In the United States, common pathogens responsible for watery diarrhea are norovirus and ETEC while those associated with dysentery or bloody diarrhea are Shigella spp. (15.3%), Campylobacter spp. (6.2%), Salmonella spp. (5.8%), EHEC (2.6%), and others (0.6%).8 Other organisms that are responsible for dysentery include Aeromonas species, noncholera Vibrio, and Y. enterocolitica. Characteristics of acute bacterial pathogens causing gastroenteritis are summarized in Table 91–3.
TABLE 91-3 Characteristics of Acute Bacterial Gastroenteritis
Cholera has been rare in the United States because of advanced water and sanitation systems. However, the disease that causes profuse watery diarrhea is endemic in the Indian subcontinent and sub-Saharan Africa. Vibrio cholerae is a gram-negative bacillus sharing similar characteristics with the family Enterobacteriaceae, and the disease is caused by toxigenic V. cholerae serogroups O1 or O139. Approximately half of the people infected with V. cholerae O1 are symptomatic, whereas only 1% to 5% of those infected with V. cholerae O139 manifest symptoms.9
E. coli is a gram-negative bacillus commonly found in the human GI tract, and diarrheagenic E. coli is differentiated into several distinct categories based on pathogenic features of diarrheal disease: ETEC, enteroinvasive E. coli (EIEC), enteropathogenic E. coli (EPEC), enteroaggregative E. coli (EAEC), and EHEC. ETEC occurs most commonly, and accounts for about half of all cases of E. coli diarrhea. There are an estimated 79,000 cases of ETEC in the United States each year.4 ETEC is also the most common cause of traveler’s diarrhea and a common cause of food- and water-associated outbreaks. Infections with EIEC and EPEC are primarily a disease of children in developing countries.10,11 EAEC strains are implicated in persistent diarrhea (≥14 days) in human immunodeficiency virus (HIV)-infected patients.12 EHEC, also known as Shiga toxin–producing E. coli (STEC), causes watery diarrhea that becomes bloody in 1 to 5 days in 80% of patients.10
Approximately 165 million cases of shigellosis occur worldwide with 450,000 cases from the United States annually.13 Shigella species are gram-negative bacilli belonging to the family Enterobacteriaceae. Four species most often associated with disease are Shigella dysenteriae type 1, Shigella flexneri, Shigella boydii, and Shigella sonnei.13 In the United States, S. sonnei and S. flexneri are the most common causes of gastroenteritis. The other two Shigella species are commonly acquired during travel to developing countries. Poor sanitation or personal hygiene, inadequate water supply, malnutrition, and increased population density are associated with an increased risk of Shigella gastroenteritis epidemics, even in developed countries.
The Campylobacter species are flagellated, curved, gram-negative rods. Although there are 14 different species, Campylobacter jejuni is the species responsible for more than 99% of Campylobacter-associated gastroenteritis. Approximately 2.4 million persons are affected each year in the United States, involving almost 1% of the entire population.4
Salmonella enterica are gram-negative bacilli belonging to the family Enterobacteriaceae. The most prevalent S. enterica serotypes are Typhi and Paratyphi, which cause enteric fever. Gastroenteritis is caused by S. enterica serotypes Typhimurium or Enteritidis. In the United States, the largest burden of Salmonella infection is due to nontyphoidal serotypes, causing approximately 1.4 million cases of salmonellosis, 16,000 hospitalizations, and 600 deaths, occurring annually.14
Recognized as a common and potentially deadly cause of infectious diarrhea, EHEC is believed to be the major etiologic factor responsible for the development of hemorrhagic colitis and hemolytic uremic syndrome (HUS). The annual disease burden of E. coli O157:H7 in the United States is more than 20,000 infections and as many as 250 deaths; however, the failure of many clinical laboratories to screen for this organism greatly complicates any estimates.15 In the United States, serotype O157:H7 causes 50% to 60% of all EHEC infections, but in the southern hemisphere, including Argentina, Australia, Chile, and South Africa, non-O157:H7 serotypes are often more prevalent. Non-O157 STEC strains in general produce a lower frequency of dysentery than O157 positive strains (62% vs. 85%).
Yersinia species are non–lactose-fermenting gram-negative coccobacilli that are widely distributed in nature. The genus Yersinia includes six species known to cause disease in humans. Y. enterocolitica and, to a lesser extent, Yersinia pseudotuberculosis are most likely associated with intestinal infection, but overall both are a relatively infrequent cause of diarrhea and abdominal pain. More than 50 serotypes of Y. enterocolitica exist; of these, serotypes 0:3, 0:8, and 0:9 are associated most frequently with enterocolitis.16 Children are the most likely to experience illness with Y. enterocolitica infection.
PATHOPHYSIOLOGY
Acute gastroenteritis and its resulting diarrhea are caused by altered movement of ions and water resulting in increased colonic secretion. Under normal conditions, the GI tract has the tremendous capacity to absorb fluid and electrolytes allowing only 100 to 200 mL of fluid to be excreted in the stool daily.17 The classic enteric pathogen that causes secretary diarrhea is V. cholerae, but ETEC and rotavirus are also responsible for watery diarrhea.
V. cholerae is an enteric pathogen that causes classical secretory diarrhea due to changes in ion secretion and absorption. Among the toxins produced by V. cholerae, the most important is cholera toxin.9 Cholera toxin consists of two subunits, A and B. The B subunits are responsible for delivery of the A subunit into the cell. The A subunit stimulates adenylate cyclase, which increases intracellular cyclic adenosine monophosphate (cAMP) and results in protein kinase A (PKA)-mediated activation of cystic fibrosis transmembrane conductance regulator (CFTR). This leads to increased chloride secretion and decreased sodium absorption producing the severe watery diarrhea characteristic of the disease.18 The toxin likely acts along the entire intestinal tract, but most fluid loss occurs in the duodenum. The net effect of the cholera toxin is isotonic fluid secretion (primarily in the small intestine) that exceeds the absorptive capacity of the intestinal tract (primarily the colon).
ETEC also causes watery diarrhea characterized by severe intestinal water secretion by producing plasmid-mediated enterotoxins: heat-labile toxin and heat-stable toxin. The heat-labile toxin has two subunits (A and B) that have similar antigenic properties and action on the gut mucosa as cholera toxin. Heat-labile toxins increase chloride secretion via activation of cAMP. The net effect is luminal accumulation of electrolytes that draws water into the intestine, and production of a cholera-like secretory diarrhea.19 Heat-stable toxin is thought to be nonantigenic and produces watery diarrhea by acting on the small intestine.
Rotavirus induces changes in transepithelial fluid balance, and causes malabsorption as a consequence of destruction of epithelial lining of intestine, and vascular damage and ischemia of villi. Once rotavirus infects small intestinal villus cells, viroplasms are formed and its toxin, nonstructural protein 4 (NSP4), is released. The viral enterotoxin increases intracellular calcium, and the increase in calcium disrupts microvillus cytoskeleton, as well as barrier function. Changes to the villi include shortening of villus height, crypt hyperplasia, and mononuclear cell infiltration of the lamina propria.20
Inflammatory diarrhea is caused by two groups of organisms—enterotoxin-producing, noninvasive bacteria (e.g., EAEC, EHEC) or invasive organisms (e.g., Salmonella spp., Shigella spp., Campylobacter spp.). The enterotoxin-producing organisms adhere to the mucosa, activate cytokines, and stimulate the intestinal mucosa to release inflammatory mediators. Invasive organisms, which can also produce enterotoxin, invade the intestinal mucosa to induce an acute inflammatory reaction, involving the activation of cytokines and inflammatory mediators.
Ingestion of as few as 10 to 200 viable organisms of the Shigella species causes disease in healthy adults.13 Shigella multiply and spread within the submucosa of the small bowel, but they rarely extend beyond the mucosa. Inflammatory diarrhea is caused by the pathogens invading the epithelial barrier through M cells where they encounter and eliminate macrophages. The destruction of macrophages after emergence from M cells causes an initial release of interleukin (IL)-1β. This initial inflammatory process is exacerbated by free bacteria binding to toll-like receptor (TLR4) that causes the production of IL-6 and IL-8. Both IL-1β and IL-8 attract polymorphonucleocytes.21 Release of polymorphonucleocytes activates chloride secretion and subsequent diarrhea. Degranulation and release of toxic substances by neutrophils cause ulceration of the epithelium, distortion of the crypts, death to intestinal epithelium, sloughing of mucosal cells, bloody mucoid exudate into the gut lumen, and submucosal accumulation of inflammatory cells with microabscess formation.22 Microabscesses eventually may coalesce, forming larger abscesses. Infection frequently involves the entire colon. In addition to the virulence characteristics of invasiveness, S. dysenteriae type 1 and, to a lesser degree, S. flexneri and S. sonnei produce a cytotoxin or Shiga toxin, which can lead to HUS.10
The pathogenicity of EHEC is related to the production of Shiga-like toxins, so named because of their resemblance to the Shiga toxin of S. dysenteriae.17 The cytotoxic effect of Shiga-like toxins disrupts the mucosal integrity of the large intestine, causing diarrhea. In addition, the toxin is able to pass through the intestinal epithelium to reach the endothelial cells lining small blood vessels that supply the gut, kidney, and other viscera, causing the myriad metabolic events that eventually lead to HUS.
CLINICAL PRESENTATION AND DIAGNOSIS
Gastroenteritis is an illness characterized by diarrhea, which may be accompanied by nausea, vomiting, fever, and abdominal pain. For the best diagnosis and management, it is important to distinguish secretory diarrhea that produces watery diarrhea from inflammatory diarrhea or dysentery. Most enteric pathogens produce acute diarrhea. Dysentery is defined as passing grossly bloody stools. Not all stools in dysenteric illness may contain visible blood, while most stools contain mucus. Systemic toxicity such as fever is often associated with dysentery of infectious origin. Symptoms of enteric pathogens that cause watery and dysentery diarrhea are listed in Table 91–1.
A physical examination and careful history that includes information about symptoms, the length of time the patient has been sick, the number of individuals affected, and recent history of travel, diet, and medications are important factors in making a diagnosis. Infections with norovirus or ETEC result in mild, self-limiting disease. Cholera produces severe dehydrating diarrhea. Infections with enteric pathogens such as Shigella spp., Salmonella spp., Campylobacter spp., EHEC, and Y. enterocolitica can result in dysentery diarrhea and severe complications. The clinical presentation of acute viral and bacterial gastroenteritis is summarized in Tables 91–2 and 91-3, respectively.
![]() Stool culture is an important tool in making an organism-specific diagnosis and determining susceptibility to antimicrobial agents. Due to the low yield, stool cultures are not recommended in most mild to moderate watery diarrhea. Instead, indications for stool cultures include dysenteric diarrhea, persistent diarrhea especially in immunocompromised patients (i.e., persons aged 65 years and older with comorbid diseases, neutropenia, or HIV infection), and diarrhea where an outbreak is suggested.1 A routine stool culture identifies the presence of Campylobacter, Salmonella, and Shigella species. The yield of stool cultures for other pathogens is increased if the test is ordered specifically based on history and physical examination. For dysenteric diarrhea, the laboratory should be instructed to look for EHEC including E. coli O157:H7. In hospitalized patients who develop diarrhea 3 days after hospitalization or in those with recent exposure to antimicrobials or chemotherapy, stool specimen should be sent for C. difficile toxins A and B. In addition to stool cultures, microscopic examination for fecal polymorphonuclear cells, or a simple immunoassay for the neutrophil marker lactoferrin, can further provide evidence of an inflammatory process and increase the yield of cultures in patients presenting with dysenteric diarrhea.
Stool culture is an important tool in making an organism-specific diagnosis and determining susceptibility to antimicrobial agents. Due to the low yield, stool cultures are not recommended in most mild to moderate watery diarrhea. Instead, indications for stool cultures include dysenteric diarrhea, persistent diarrhea especially in immunocompromised patients (i.e., persons aged 65 years and older with comorbid diseases, neutropenia, or HIV infection), and diarrhea where an outbreak is suggested.1 A routine stool culture identifies the presence of Campylobacter, Salmonella, and Shigella species. The yield of stool cultures for other pathogens is increased if the test is ordered specifically based on history and physical examination. For dysenteric diarrhea, the laboratory should be instructed to look for EHEC including E. coli O157:H7. In hospitalized patients who develop diarrhea 3 days after hospitalization or in those with recent exposure to antimicrobials or chemotherapy, stool specimen should be sent for C. difficile toxins A and B. In addition to stool cultures, microscopic examination for fecal polymorphonuclear cells, or a simple immunoassay for the neutrophil marker lactoferrin, can further provide evidence of an inflammatory process and increase the yield of cultures in patients presenting with dysenteric diarrhea.
Complications
Complications associated with acute watery diarrhea most likely result from dehydration and, therefore, Treatment below focuses on rehydration therapy. Dysenteric diarrhea is more likely to have severe complications, especially in children less than 5 years of age and in elderly. Enteric pathogens responsible for complications include Shigella spp., Salmonella spp., Campylobacter spp., EHEC, and Y. enterocolitica.
Bacteremia is the most common complication of gastroenteritis and can be seen after infections with nontyphoid Salmonella, C. jejuni or C. fetus, and Y. enterocolitica.15 Nontyphoid Salmonella is most common in children less than 5 years of age, elderly, and patients with hemoglobinopathy, malaria, or immunosuppression. Bacteremia due to Campylobacter spp. has been reported in patients with HIV infection, malignancy, transplantation, and hypogammaglobulinemia. Although rare, Y. enterocolitica bacteremia has been reported in patients with diabetes mellitus, severe anemia, hemochromatosis, cirrhosis, and malignancy, the elderly, and those who have received frequent red blood cell transfusions (iron overload).23 The clinical syndrome is characterized by persistent bacteremia and prolonged intermittent fever with chills. Stool cultures frequently are negative. Leukocyte counts are often within the normal range. Vascular complications such as seeding of atherosclerotic plaques or aneurysms in arterial vessels occur in 10% to 25% of adults with bacteremia. Localized infections involving bone, cysts, heart, kidney, liver, lungs, pericardium, and spleen develop in 5% to 10% of patients with bacteremia.
Severe complication in patients infected with EHEC is HUS. HUS is defined by the triad of acute renal failure, thrombocytopenia, and microangiopathic hemolytic anemia.24 This syndrome is commonly observed in children less than 5 years of age and the elderly. Approximately 2% to 7% of cases infected with O157:H7 strains are complicated by development of HUS. Death may occur rarely, usually as a result of HUS. S. dysenteriae type 1 can also cause HUS, although more rarely than observed with EHEC.13
Shigella infection may also lead to complications such as generalized seizures, sepsis, toxic megacolon, perforated colon, arthritis, and protein-losing enteropathy. Mortality is rare, but it may be more likely with S. dysenteriae type I. Less than 3% of persons who are infected with S. flexneri will later develop Reiter’s syndrome, characterized by pains in the joints, irritation of the eyes, and painful urination. This can lead to chronic arthritis.25
Infection with C. jejuni has been associated with Guillain-Barré syndrome (GBS), but the relationship is not well understood.26 The risk of developing GBS after C. jejuni infection appears to be low (approximately 1 case of GBS per 1,000 C. jejuni infections). The weakness usually starts in the legs with difficulty in walking and may progress to a complete paralysis of all extremities that lasts several weeks and usually requires intensive care.
Approximately 10% to 30% of adult patients develop a reactive arthritis 1 to 2 weeks after recovery from gastroenteritis secondary to S. flexneri, Salmonella spp., C. jejuni, and Y. enterocolitica. This arthritis, involving the knees, ankles, toes, fingers, and wrists, usually resolves in 1 to 4 months but may persist in approximately 10% of patients.26 This complication is more common in persons with the HLA-B27 antigen.
TREATMENT
Fortunately, diarrheal mortality has declined substantially in the past 2 decades, especially among children younger than 1 year of age. Interventions for diarrheal disease such as improved sanitation, increased use of oral rehydration therapy (ORT), breast-feeding, and better weaning practices are responsible for the decrease in case-fatality rates.
General Approach to Treatment
The cornerstone of management for all GI infections and enterotoxigenic poisonings is to prevent dehydration by correcting fluid and electrolyte imbalances. In mild, self-limiting acute gastroenteritis, a diet of oral fluids and easily digestible foods such as chicken soup and crackers is recommended. In patients with severe dehydrating watery diarrhea and dysenteric diarrhea, IV rehydration therapy, antibiotics, and/or antimotility treatments are needed.
Rehydration Therapy
Initial assessment of fluid loss is essential for successful rehydration therapy and should include acute weight loss, as it is the most reliable means of determining the extent of water loss. However, if accurate baseline weight is not available, clinical signs are helpful in determining approximate deficits (Table 91–4). Physical assessment generally is more reliable in young children and infants than in adults.
TABLE 91-4 Clinical Assessment of Degree of Dehydration in Children Based on Percentage of Body Weight Lossa
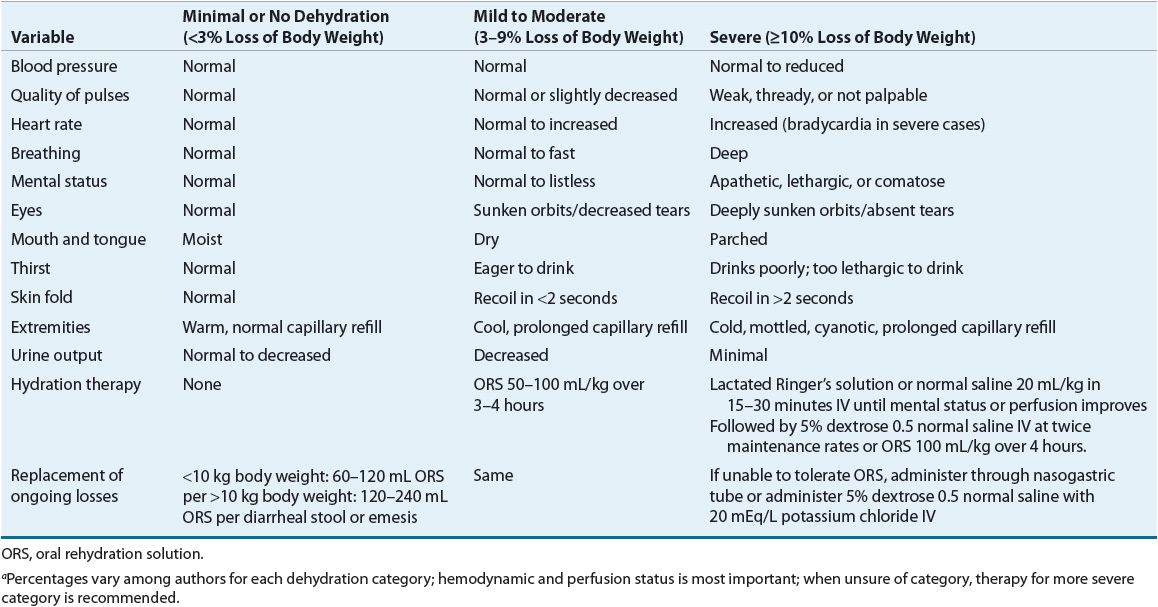
![]() Fluid replacement is the cornerstone of therapy for dehydration due to diarrhea regardless of etiology. For the treatment of mild to moderate dehydration, ORT is superior to administration of IV fluids. ORT reverses dehydration in nearly all patients with mild to moderate diarrhea with 94% to 97% efficacy.27 It offers the advantages of being inexpensive and noninvasive, and does not require hospitalization for administration. Moreover, thirst drives use of ORT and provides a safeguard against overhydration. Therefore, treatment of dehydration consists of ORT for rehydration and replacement of ongoing losses as well as continuation of normal feeding.
Fluid replacement is the cornerstone of therapy for dehydration due to diarrhea regardless of etiology. For the treatment of mild to moderate dehydration, ORT is superior to administration of IV fluids. ORT reverses dehydration in nearly all patients with mild to moderate diarrhea with 94% to 97% efficacy.27 It offers the advantages of being inexpensive and noninvasive, and does not require hospitalization for administration. Moreover, thirst drives use of ORT and provides a safeguard against overhydration. Therefore, treatment of dehydration consists of ORT for rehydration and replacement of ongoing losses as well as continuation of normal feeding.
The necessary components of oral rehydration solutions (ORS) include carbohydrates (typically glucose), sodium, potassium, chloride, and water. In 1979, the World Health Organization (WHO) introduced a glucose-based ORS formulation that is 310 mOsm/L. This original formation known as ORS ≥310 takes advantage of glucose-coupled sodium transport in the small bowel and enhances sodium and subsequently water transport across intestinal walls. Therefore, this glucose-based ORS is effective in replacing the fluid from acute diarrhea but does not reduce stool output or shorten duration of diarrhea. In 2002, the World Health Organization/United Nations Children’s Fund (WHO/UNICEF) changed its recommendation to a reduced osmolarity solution (osmolarity = 245 mOsm/L). The use of ORS ≤270 reduced stool volume, shortened duration of diarrhea, and decreased need for unscheduled IV therapy when compared with ORS ≥310.28 The newer formulation of ORS ≤270 mOsm/L was, however, more likely to cause hyponatremia (blood sodium levels <130 mmol/L).29
In restoring fluid and electrolyte balance in cholera infections, polymer-based ORS may be more efficacious than glucose-based ORS. Polymer-based ORS contains rice, wheat, sorghum, or maize. This polymer-based ORS releases glucose more slowly after digestion, and when absorbed in the small bowel, enhances the reabsorption of water and electrolyte secreted into the bowel lumen during diarrhea. In a meta-analysis of 34 trials, polymer-based ORS has been shown to reduce the duration of diarrhea in adults with cholera when compared with glucose-based ORS ≥310 and ≤270.30
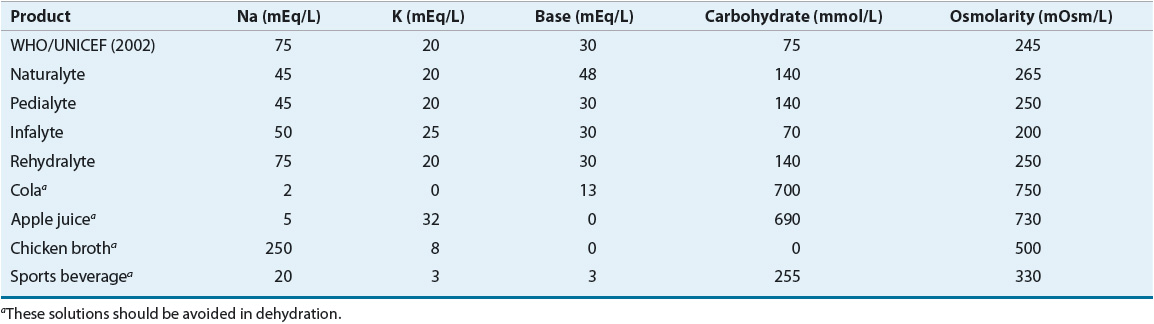
Guidelines on rehydration therapy based on the degree of dehydration and replacement of ongoing losses are outlined in Table 91–4. ORS should be given in small and frequent volumes (5 mL every 2 to 3 minutes in a teaspoon or oral syringe). Nasogastric administration of ORT is an alternative method of administration in a child with persistent vomiting. For breastfed infants, nursing should be continued. The composition of commercial ORS and commonly consumed beverages is listed in Table 91–5. Clear fluids, such as soft drinks, sweetened fruit drinks, broth, and sports drinks, should be avoided in the treatment of dehydration. Those solutions may cause an osmotic diarrhea and hypernatremia.
TABLE 91-5 Comparison of Common Solutions Used in Oral Rehydration and Maintenance