Fallopian Tube
Mitra Mehrad
John D. Pfeifer
Phyllis C. Huettner
I. NORMAL ANATOMY. The fallopian tubes are formed from the Müllerian (paramesonephric duct) system and lie within the broad ligament between the ovary and the uterus. They conduct eggs from the surface of the ovary to the uterine cavity and are the usual site of fertilization. Each fallopian tube is shaped like an elongated funnel and is divided into four parts from lateral to medial: infundibulum, ampulla, isthmus, interstitium; the infundibulum contains the finger-like fimbriae distally.
The fallopian tube mucosa is branched and folded into plicae. The mucosa is lined by a nonstratified epithelium composed of three cell types: ciliated, secretory, and intercalated cells. The most common are the ciliated cells, followed by secretory cells, which together comprise over 90% of the cell population. The intercalated cells are seen as elongated nuclei sporadically present between the ciliated cells. The wall of the fallopian tube contains smooth muscle to aid in moving the fertilized egg into the uterus. The serosa contains abundant blood vessels and is continuous with the broad ligament.
II. GROSS EXAMINATION, TISSUE SAMPLING, AND HISTOLOGIC SLIDE PREPARATION. Fallopian tubes are usually received as a portion of a total abdominal hysterectomy-bilateral salpingo-oophorectomy specimen. Short cross sections of fallopian tube are received after tubal ligation procedures. Ectopic pregnancy specimens also usually contain a portion of fallopian tube. Rarely, specimens are received for primary fallopian tube tumors.
At the grossing station, the length and diameter of the fallopian tube should be documented, as well as the presence or absence of a fimbriated end, or evidence of prior ligation. The serosal surface should also be assessed for the presence of adhesions, cysts, exudates, rupture, or metastatic tumor. The tube is then serially sectioned.
A. Benign specimens. Three sections are submitted from a normal fallopian tube, specifically from the fimbriated end, ampulla, and isthmus end. Additional sections of any gross lesions are also submitted. Complete cross sections must be identified from a tubal ligation specimen.
When examining an ectopic pregnancy specimen, an embryo and/or placental villi will often be grossly evident. Hemorrhagic areas, including blood clot, along with obvious villous or embryonic tissue, should be submitted for histologic examination.
B. Neoplastic specimens. Primary tubal carcinoma specimens will show a dilated lumen filled with a papillary or solid tumor. An ovarian tumor secondarily involving the fallopian tube is more common than a primary fallopian tube tumor, and careful sectioning can help distinguish the two. Grossly, papillary and solid areas should be sampled thoroughly (at least one section per centimeter of tumor), along with uninvolved areas. If possible, a section showing the tumor’s relationship to the ovary should be submitted.
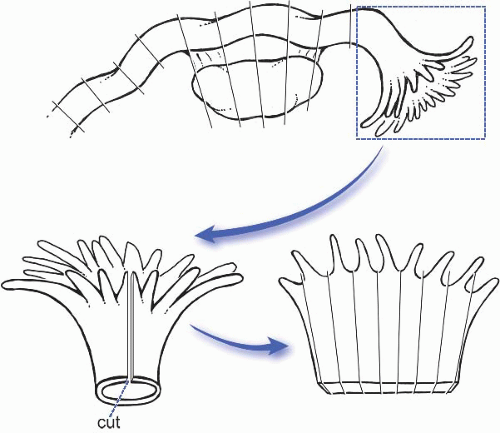
C. Prophylactic excision specimens. Fallopian tube specimens received as part of a prophylactic hysterectomy-oophorectomy from patients with hereditary cancer syndromes (e.g., BRCA1 syndrome) should be, together with the ovaries, entirely submitted for histologic examination. For the fallopian tube, the goal is to ensure sectioning and extensive examination of the fimbria (so-called SEEFIM protocol), since the majority of early serous tumors occur in this area.
The protocol (Am J Surg Pathol. 2006;30:230) specifies that the entire tube is fixed for at least 4 hours to minimize loss of epithelium during manipulation, the distal 2 cm of the fimbriated end is transected, the fimbrial mucosa is sectioned longitudinally into four pieces, the remainder of the tube is sectioned transversely every 2 to 3 mm, and that all the sections are submitted in toto (Fig. 32.1).
III. DIAGNOSTIC FEATURES OF COMMON DISEASES
A. Inflammatory and nonneoplastic lesions of the fallopian tube
1. Cystic lesions. Embryologic remnants may be found in the fallopian tube wall, usually as an incidental finding (e-Fig. 32.1).* Paratubal cysts of Müllerian origin (ciliated lining) or Wolffian origin (stratified transitional lining) may be encountered; if the lining is atrophic secondary to compression, the two can be difficult to differentiate microscopically. Pedunculated paratubal cysts with a Müllerian epithelium present along the fimbriae are termed hydatid of Morgagni.
2. Inflammatory lesions
a. Acute salpingitis usually presents in young- to middle-aged women, is generally an ascending infection initiated by Chlamydia or Neisseria
gonorrhoeae, and is often followed by polymicrobial infection (pelvic inflammatory disease). The damage to the tube that results from acute salpingitis may lead to infertility and/or ectopic pregnancy. Grossly, the tubal lumen may be distended by pus, blood, or secretions. Histologic sections show marked acute inflammation, with congestion and edema in the plicae and the tubal wall which often severely distorts the normal tubal architecture (e-Fig. 32.2).
b. Chronic salpingitis is usually due to resolving acute salpingitis. Grossly, the tube is often enlarged, fibrotic, distorted, and adherent to the ovary or adjacent structures. Microscopically, a lymphoplasmacytic infiltrate is seen in the plicae. Fusion of the tubal plicae after resolution of acute salpingitis may lead to formation of follicle-like spaces, a histologic pattern known as salpingitis follicularis. Hydrosalpinx may be seen in endstage chronic salpingitis, microscopically characterized by a dramatically thinned wall with few plicae and a lumen filled with clear fluid. Numerous adhesions are typically present on the serosal surface (e-Fig. 32.3).
c. Granulomatous salpingitis may be caused by tuberculosis, fungal infection, Crohn disease, or sarcoidosis.
d. Salpingitis isthmica nodosum typically presents in young women. It is associated with ectopic pregnancy and infertility, and has an unclear pathogenesis. Grossly, it presents as 1 to 2 cm diameter nodules in the wall of the fallopian tube isthmus. It is bilateral in 85% of cases. The lesions consist of outpouchings of tubal epithelium surrounded by a thickened wall of smooth muscle (e-Fig. 32.4).
3. Other nonneoplastic lesions of the fallopian tube
a. The fallopian tube is a frequent site of involvement of endometriosis, primarily in women of reproductive age, which may be associated with infertility. Grossly, tubal endometriosis consists of dark brown serosal nodules. The microscopic findings include endometrial glands surrounded by a cuff of endometrial stroma with associated hemosiderin-laden macrophages and chronic inflammatory cells (e-Fig. 32.5).
b. Ectopic pregnancy affects 1% to 2% of all conceptions, and the fallopian tube is the most common site; the most commonly involved region of the tube is the ampulla. Risk factors include prior ectopic pregnancy, salpingitis, congenital tubal anomalies, salpingitis isthmica nodosum, and endometriosis. Patients may present with tubal rupture and shock. Grossly, the fallopian tube is dilated and hemorrhagic with identifiable chorionic villi, with or without an identifiable embryo. Histologic sections should show chorionic villi or trophoblast in the tubal mucosa or wall (e-Fig. 32.6).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree