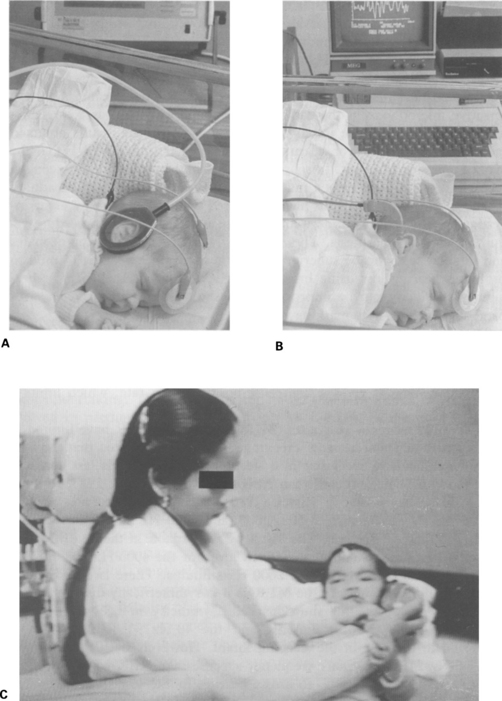
12 Accurate hearing levels are difficult to measure in early infancy, and the epidemiological picture is inadequate. A World Health Organization (WHO) report (1967) suggests that the incidence of severe hearing loss in neonates is about 1:1000, and that of some degree of hearing impairment may be as high as 5:1000. Certain children have an increased risk of hearing loss and are in a special ‘at risk’ group. It has been shown that in some groups, for example, very low birth-weight children (less than 1500 g) in intensive care, the incidence of bilateral severe hearing loss is 4% and the incidence of any degree of hearing loss, including mild and unilateral, reaches 9% for those aged 3–6 years (Abramovich et al 1979). It should be noted that low birth-weight alone is not a ‘risk’ factor for hearing loss. However, factors such as perinatal illness, particularly those conditions, including apnoea, known or likely to have caused hypoxia in the neonatal period, are significantly associated with sensorineural hearing loss, and jaundice appears to have an additive effect (Abramovich et al 1979). A register of the main risk factors is a useful way of identifying children who are most likely to suffer impaired hearing and who, therefore, need reliable testing. A practical version of a risk register, similar to that proposed by the Joint Committee on Infant Hearing (1982), has proved useful; it includes the following factors: 1. Family history of childhood hearing impairment 2. Congenital perinatal infections 3. Anatomical malformations involving head and neck 4. Birth-weight less than 1500 g The Joint Committee on Infant Hearing (1982) has estimated the incidence of moderate to profound hearing loss at 2.5–5% in the ‘at risk’ group. The incidence of risk factors is under 5%, and, as most babies with risk factors 1 and 3 (above) are to be found in the normal wards, the high-risk group is not necessarily concentrated among those infants treated in special-care baby units. In these groups, the incidence of severe hearing loss increases by at least tenfold, and that of some degree of hearing loss increases by much more. Some risk factors, for example, recessive familial hearing loss, viral infections and asphyxia depend on the investigation pattern for detection, and are especially prone to variability. About 95% of babies do not fall into a risk category; however, one should bear in mind that only half of the children with a hearing loss manifest a known risk factor (Simmons 1980, Feinmesser et al 1982, Stein et al 1983). This emphasizes the need for general awareness of the possibility of hearing loss. Observational audiometry and automated behavioural screening tests, such as the auditory-response cradle, identify neonates with severe and profound hearing loss fairly reliably (Feinmesser & Tell 1976, Bennett 1979, 1980, Weiss 1983, Bhattacharya et al 1984). Several reports have indicated high error rates with the standard behavioural tests and with the automated auditory-response cradle (Feinmesser & Tell 1976, Northern & Downs 1978, Davies 1984, McCormick et al 1984). Auditory-evoked potentials have been used with enthusiasm by many investigators. ECochG is undesirable because it is an invasive procedure requiring general anaesthesia. The ABR, easily recorded in sleeping neonates and infants, seems to be superior to other tests in the screening of mild to profound hearing loss using broad-band clicks (Fig. 12.1, A, B, C). Frequency specificity can be improved by masking parts of the cochlea with band-stop or high-pass masking noise. Fig. 12.1 (A) The Algotek screener in use on a neonate. Acoustic stimuli are delivered by plastic tubes to a circumaural, sealed cavity. This avoids the closure of the ear canal, which can occur with earphones. (B) ABR responses being recorded from a neonate. A miniature insert transducer is used to deliver the stimulus. (C) ABR screening of a 4-month-old child. The mother holds the earphone a few centimetres from the ear. The postauricular muscle response (PAM) has been used for screening neonates and infants (Douek et al 1973, Fraser et al 1978). PAM depends on the tonus of the scalp muscles and is elicited only at relatively high intensities. It gives a high false-positive error rate, which limits its usefulness. The middle-latency responses (MLR) and 40 Hz MLR have limited application in neonatal hearing screening. It has been reported that the MLR may be difficult to recognize in a sleeping neonate, and is less consistent and more difficult to record than ABR (Ozdamar & Kraus 1983, Rotteveel et al 1986). Frye-Osier & Hirsch (1980) tested normal neonates and recorded MLRs in response to 500; 2000; and 4000 Hz tone bursts at levels of 10, 30, and 50 dBnHL. The MLR was recordable at the 10 dB level for the 500 Hz stimulus and at the 30 dB level for the 4000 Hz stimulus, but only at the 50 dB level for the 2000 Hz stimulus. There is clear evidence of maturational changes in the MLR as it is symmetrically distributed over the scalp in adults, but show marked asymmetry in neonates (Wolf & Goldstein 1978). Both the MLR and the 40 Hz MLR thresholds are reported to increase in the sleeping infant. However, both the MLR and the 40 Hz MLR are more frequency-specific than ABR when using tone pips, and can be of value to assess hearing in the low frequencies. The middle-latency responses, including MLR, 40 Hz MLR, and SN10 can be considered in some difficult cases to supplement hearing investigation in a test battery. In conclusion, ABR is the test of choice for screening neonates and infants with broad-band frequency clicks. If a more detailed audiometric contour is required, then some frequency specificity can be achieved by masking with the techniques of band-stop or high-pass masking noise. Other more frequency-specific MLR potentials can be recorded to assess hearing loss at low audiometric frequencies. Available ERA tests for screening along with the recommended parameters are shown in Table 12.1.
ERA in hearing screening in neonates and infants
CLINICAL PROBLEM
Population
CHOICE OF SCREENING TESTS
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Basicmedical Key
Fastest Basicmedical Insight Engine

