CHARACTERISTIC FEATURES OF EPITHELIAL CELLS
Intercellular Adhesion & Other Junctions
SPECIALIZATIONS OF THE APICAL CELL SURFACE
Despite its complexity, the human body is composed of only four basic types of tissue: epithelial, connective, muscular, and nervous. These tissues, which all contain cells and molecules of the extracellular matrix (ECM), exist in association with one another and in variable proportions and morphologies, forming the different organs of the body. The main characteristics of these basic types of tissue are shown in Table 4–1. Also of great functional importance are the free cells found in body fluids such as blood and lymph.
TABLE 4–1 Main characteristics of the four basic types of tissues.

Connective tissue is characterized by the abundant ECM produced by its cells; muscle tissue is composed of elongated cells specialized for contraction and movement; and nervous tissue is composed of cells with long, fine processes specialized to receive, generate, and transmit nerve impulses. Most organs can be divided into parenchyma, which is composed of the cells responsible for the organ’s specialized functions, and stroma, the cells of which have a supporting role in the organ. Except in the brain and spinal cord, the stroma is always connective tissue.
Epithelial tissues are composed of closely aggregated polyhedral cells with strong adhesion to one another and attached to a thin layer of ECM. Epithelia are cellular sheets that line the cavities of organs and cover the body surface.
The principal functions of epithelial (Gr. epi, upon + thele, nipple) tissues include the following:
 Covering, lining, and protecting surfaces (eg, epidermis)
Covering, lining, and protecting surfaces (eg, epidermis)
 Absorption (eg, the intestinal lining)
Absorption (eg, the intestinal lining)
 Secretion (eg, parenchymal cells of glands)
Secretion (eg, parenchymal cells of glands)
Specific cells of certain epithelia may be contractile (myoepithelial cells) or specialized sensory cells, such as those of taste buds or the olfactory epithelium. Because epithelial cells line all external and internal surfaces of the body, all substances that enter or leave tissues and organs must cross an epithelium.
CHARACTERISTIC FEATURES OF EPITHELIAL CELLS
The shapes and dimensions of epithelial cells are quite variable, ranging from tall columnar to cuboidal to low squamous cells. The cells’ size and morphology are generally dictated by their function. Epithelial cell nuclei vary in shape and may be elliptic (oval), spherical, or flattened. Nuclear shape corresponds roughly to cell shape; tall cells have elongated nuclei and squamous cells have flattened nuclei. Cuboidal or pyramidal cells usually have more spherical nuclei (Figure 4–1).
FIGURE 4–1 Epithelia and adjacent connective tissue.
Because the lipid-rich membranes of epithelial cells are frequently indistinguishable with the light microscope, the number and shape of the stained nuclei can indicate cell density and shape. The nuclei also allow one to determine the number of cell layers in an epithelium, a primary morphologic criterion for classifying epithelia.
Most epithelia rest on connective tissue that contains the microvasculature bringing nutrients and O2 to both tissues. Even thick epithelia do not themselves normally contain blood vessels. The connective tissue that underlies the epithelia lining the organs of the digestive, respiratory, and urinary systems is called the lamina propria. The area of contact between the epithelium and connective tissue may be increased by irregularities at the interface in the form of small evaginations called papillae (L. diminutive of papula, nipple; singular papilla). Papillae occur most frequently in epithelial tissues subject to friction, such as the covering of the skin or tongue.
Epithelial cells generally show polarity, with organelles and membrane proteins distributed unevenly within the cell. The region of the cell contacting the connective tissue is called the basal pole and the opposite end, usually facing a space, is the apical pole. The two poles of epithelial cells differ in both structure and function. Regions of cuboidal or columnar cells that adjoin the neighboring cells are the lateral surfaces; cell membranes here often have numerous infoldings to increase the area of that surface, increasing its functional capacity.
Basement Membranes
All epithelial cells in contact with subjacent connective tissue have at their basal surfaces a specialized, feltlike sheet of extracellular material referred to as the basement membrane (Figure 4–1). Glycoproteins and other components in this structure can be stained and make it visible beneath epithelia with the light microscope (Figure 4–2).
FIGURE 4–2 Basement membranes.
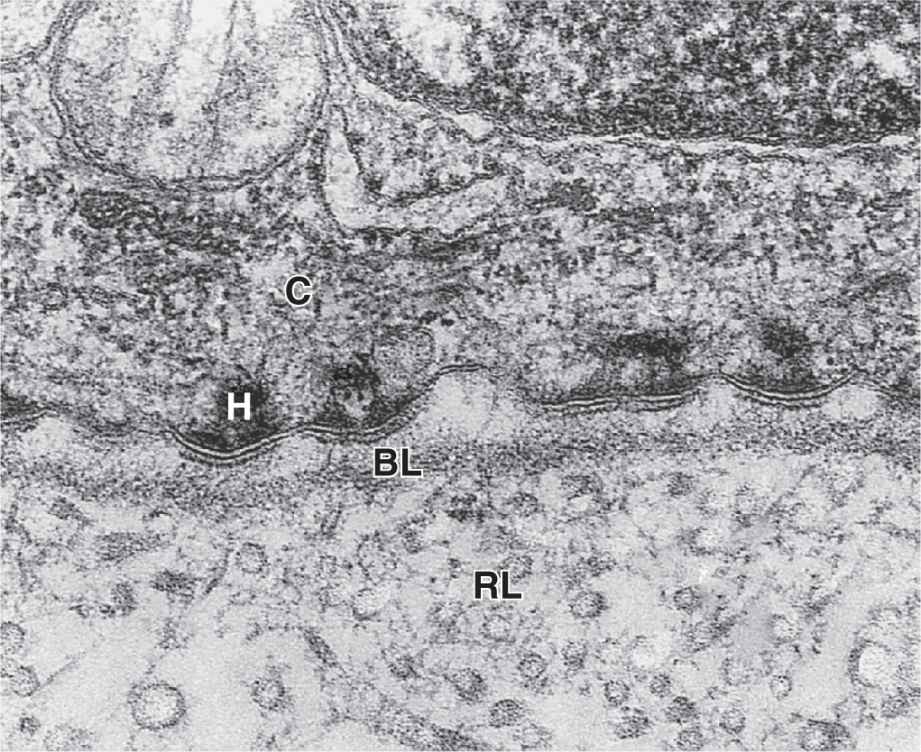
With the transmission electron microscope (TEM) the basement membrane may be resolved into two structures. Nearest the epithelial basal poles is an electron-dense layer, 20-100 nm thick, consisting of a network of fine fibrils that comprise the basal lamina (Figure 4–3). Beneath this layer is often a more diffuse and fibrous reticular lamina.
FIGURE 4–3 Basal and reticular laminae.
The terms “basement membrane” and “basal lamina” are often used indiscriminately, causing confusion. Most authors use “basal lamina” to denote the extracellular epithelial layer seen ultrastructurally and “basement membrane” for the entire structure below an epithelium visible with the light microscope.
The macromolecules of the basal lamina are secreted at the basal poles of the epithelial cells and form three-dimensional arrays. ECM components are described more fully in Chapter 5, but those of basal laminae include the following:
 Laminin: These are large glycoproteins that self-assemble as a lacelike network immediately below the cells’ basal poles where they are held in place by the transmembrane integrins.
Laminin: These are large glycoproteins that self-assemble as a lacelike network immediately below the cells’ basal poles where they are held in place by the transmembrane integrins.
 Type IV collagen: Monomers of type IV collagen contain three polypeptide chains and self-assemble further to form a feltlike layer.
Type IV collagen: Monomers of type IV collagen contain three polypeptide chains and self-assemble further to form a feltlike layer.
 The laminin and type IV collagen networks are held together by the adhesive glycoprotein entactin/nidogen, and by perlecan, a proteoglycan.
The laminin and type IV collagen networks are held together by the adhesive glycoprotein entactin/nidogen, and by perlecan, a proteoglycan.
These components of basal laminae are also produced by other cells besides those of epithelia, including muscle cells, adipocytes (fat-storing cells), and the cells supporting peripheral neurons. Surrounding these cells, this external lamina binds factors important for interactions with other cells and serves as semipermeable barriers further regulating macromolecular exchanges between the enclosed cells and connective tissue.
The more diffuse meshwork of reticular laminae contains type III collagen and is bound to the basal laminae by anchoring fibrils of type VII collagen. These components of the basement membrane are produced by cells of the connective tissue and are also visible by TEM (Figure 4–3).
Basement membranes have many functions. They provide structural support and polarity to epithelial cells and attach epithelia to underlying connective tissue. Proteins of the layered meshwork help filter substances entering the epithelium from below, concentrate mitogenic growth factors, and form a scaffold for epithelial repair and regeneration. Basal lamina components help organize proteins in the plasma membrane of epithelial cells, affecting endocytosis, signal transduction, and other activities. Basement membrane proteins also mediate many cell-to-cell interactions involving epithelia and mark routes for certain cell migrations along epithelia.
Intercellular Adhesion & Other Junctions
Several membrane-associated structures provide adhesion and communication between cells. Some are present in other tissues but all are particularly numerous and prominent in epithelia. Epithelial cells adhere strongly to neighboring cells and basal laminae, particularly in epithelia subject to friction or other mechanical forces.
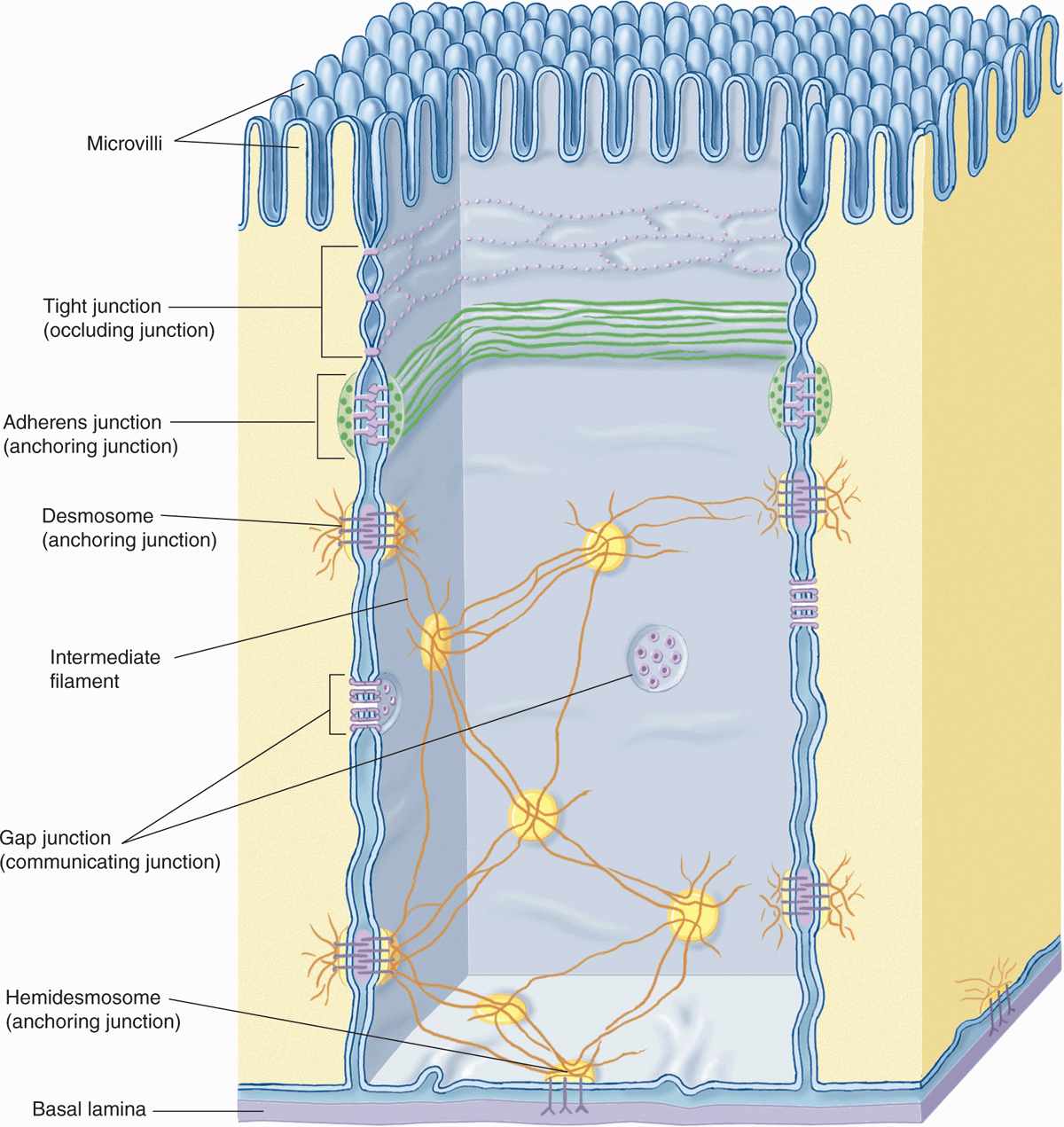
As shown in Figure 4–4 and summarized in Table 4–2, lateral surfaces of epithelial cells exhibit several specialized intercellular junctions, which serve different functions:
FIGURE 4–4 Junctional complexes of epithelial cells.
TABLE 4–2 Epithelial cell junctions, their major structural features and functions, and medical significance.

 Tight or occluding junctions form a seal between adjacent cells.
Tight or occluding junctions form a seal between adjacent cells.
 Adherent or anchoring junctions are sites of string cell adhesion.
Adherent or anchoring junctions are sites of string cell adhesion.
 Gap junctions are channels for communication between adjacent cells.
Gap junctions are channels for communication between adjacent cells.
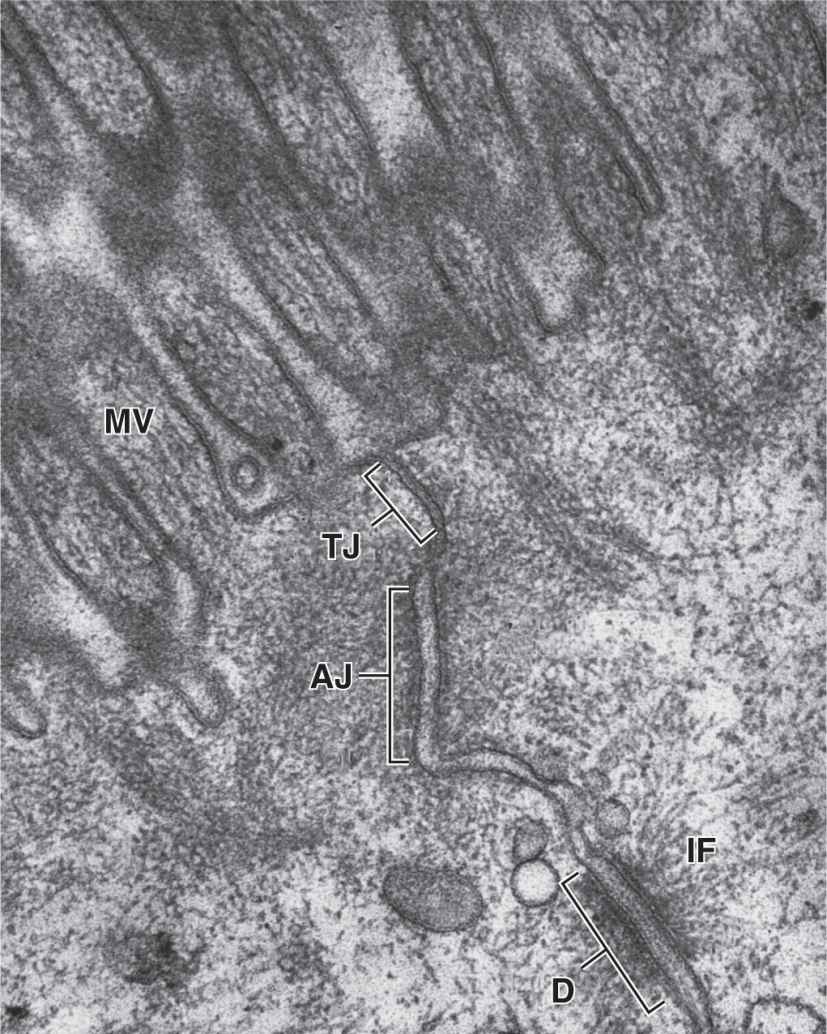
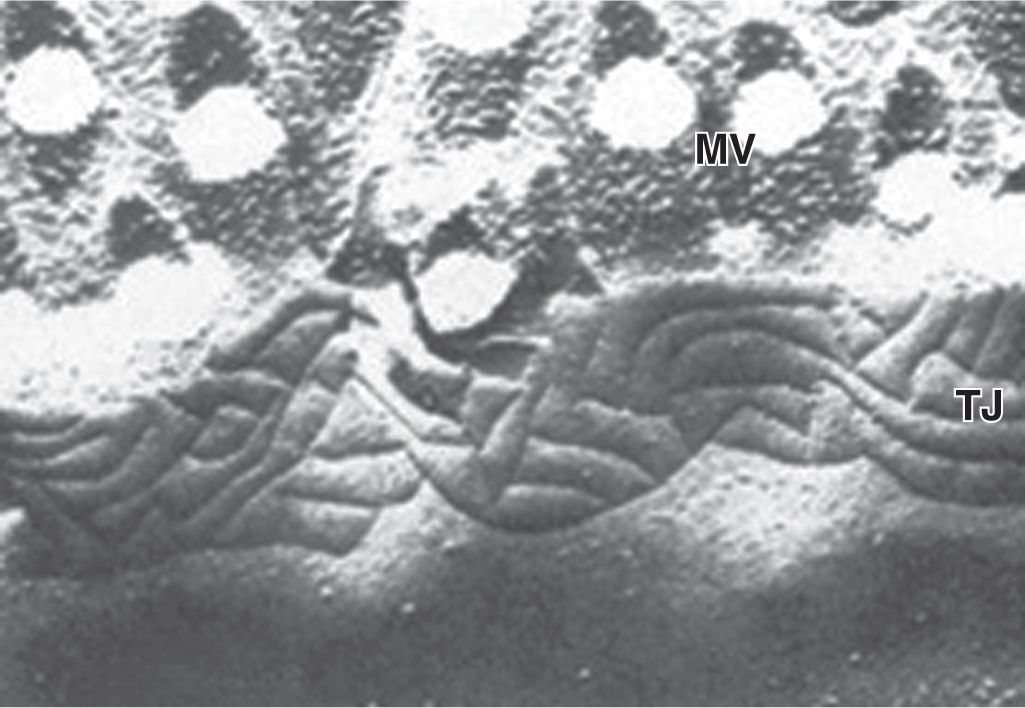
In many epithelia these junctions are present in a definite order at the apical end of the cells. Tight junctions, or zonulae occludens, are the most apical of the junctions. The term “zonula” indicates that the junction forms a band completely encircling each cell. In TEM the adjacent membranes at these junctions appear fused or very tightly apposed (Figures 4–5). The seal between the membranes is due to interactions between the transmembrane proteins claudin and occludin of each cell. Zonulae occludens are clearly seen after cryofracture (Figure 4–6), where they appear as a band of branching strands in the membrane around each cell’s apical end. The intercellular seal of this junctional type ensures that molecules crossing an epithelium in either direction do so by going through the cells (transcellular path) rather than between them (paracellular pathway). Epithelia with one or very few fused sealing strands (eg, proximal renal tubule) are more permeable to water and solutes than are epithelia with numerous fusion sites (eg, the lining of the urinary bladder).
FIGURE 4–5 Epithelial cell junctional complex.
FIGURE 4–6 View of a tight junction after cryofracture.
Tight junctions also serve a related purpose in epithelia. These continuous zones around epithelial cells prevent membrane proteins at the apical cell surface from moving in the membrane to the basal and lateral surfaces, and vice versa. This produces two membrane domains (apical and basolateral) with different protein populations, which allows the two sides of the epithelium to maintain different receptors and to function differently. Apical cell membranes of epithelia are part of the luminal compartment of a tissue or organ, while the basolateral domains of the epithelial cells are part of a basal compartment that also encompasses the underlying connective tissue.
The second type of junction is the zonula adherens (Figures 4–4 and 4–5), which also encircles the epithelial cell, usually immediately below the zonula occludens. This is an adherent junction, firmly anchoring a cell to its neighbors. Cell adhesion here is mediated by cadherins, transmembrane glycoproteins of each cell that interact in the presence of Ca2+. At their cytoplasmic ends, cadherins bind catenin that is linked via actin-binding proteins to actin filaments. The actin filaments linked to the zonula adherens form part of the terminal web, a cytoskeletal feature at the apical pole in many epithelial cells with a role in cytoplasmic motility and other functions. Together, the tight and adherent junctions encircling the apical ends of epithelial cells function like the plastic form that holds a six-pack of canned drinks together.
A related adherent junction is the desmosome or macula adherens (L. macula, spot). As the latter name implies, this junction resembles a single “spot-weld” and does not form a belt around the cell. Maculae adherens or desmosomes are discshaped structures at the surface of one cell that are matched with identical structures at an adjacent cell surface (Figures 4–4 and 4–5). Desmosomes contain larger members of the cadherin family called desmoglein and desmocollin. On the cytoplasmic side of each cell membrane, these proteins inset into a dense attachment plaque of anchoring proteins (plakoglobin and desmoplakin) that bind intermediate filaments rather than actin filaments. Cable-like filaments of cytokeratin are most common in desmosomes of epithelia. Because intermediate filaments of the cytoskeleton are very strong, desmosomes provide firm adhesion among the cells. In nonepithelial cells, the intermediate filaments attached to desmosomes are composed of other proteins, such as desmin or vimentin.
Gap junctions mediate communication rather than adhesion or occlusion between cells (Figure 4–7). Abundant in many epithelia, gap junctions are also functionally important in nearly all mammalian tissues. Cryofracture preparations show that gap junctions consist of aggregated transmembrane protein complexes that form circular patches in the plasma membrane (Figure 4–7b).
FIGURE 4–7 Gap junctions.
The gap junction proteins, called connexins, form hexameric complexes called connexons, each of which has a central hydrophilic pore about 1.5 nm in diameter. When two cells attach, connexins in the adjacent cell membranes move laterally and align to form connexons between the two cells (Figure 4–4), with each junction having dozens or hundreds of aligned connexon pairs. Gap junctions permit intercellular exchange of molecules with small (<1.5 nm) diameters. Some molecules mediating signal transduction, such as cyclic nucleotides and ions, move rapidly through gap junctions, allowing cells in many tissues to act in a coordinated manner rather than as independent units. For example, in heart and visceral muscles gap junctions help produce rhythmic contractions.
The basal domain of an epithelial cell (Figure 4–4) attaches to the subjacent basal lamina by junctions called hemidesmosomes (Gr. hemi, half + desmos + soma), which can be observed by TEM (Figure 4–3). These adhesive structures resemble a half-desmosome ultrastructurally, but, unlike desmosomes, they contain abundant integrins rather than cadherins. The transmembrane integrin proteins bind the extracellular macromolecules laminin and collagen type IV.
SPECIALIZATIONS OF THE APICAL CELL SURFACE
The apical ends of many tall or cuboidal epithelial cells face an organ’s lumen and often have specialized projecting structures. These function either to increase the apical surface area for absorption or to move substances along the epithelial surface.
Microvilli
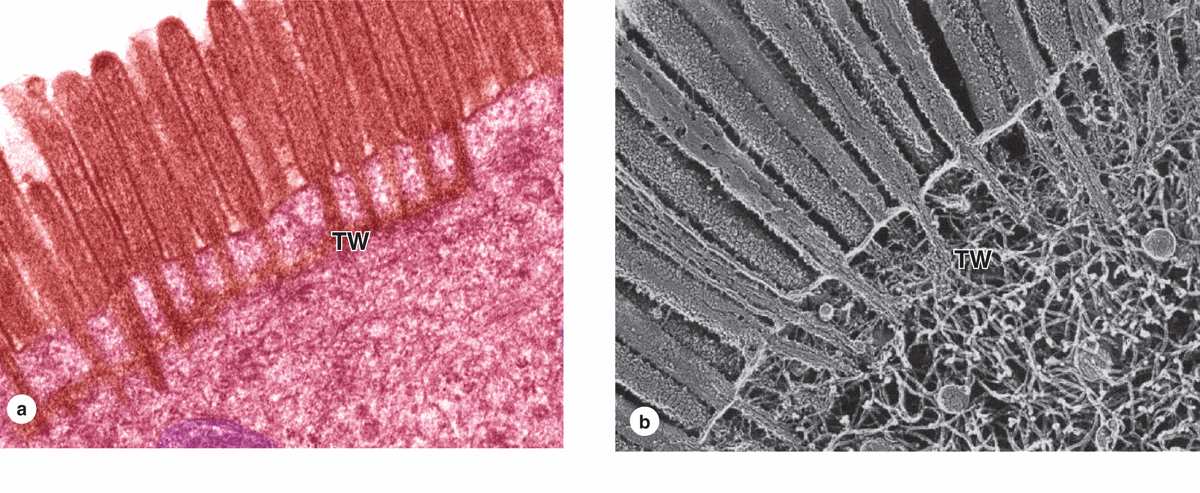
Many cells are seen to have cytoplasmic projections visible with the electron microscope. Such extensions usually reflect cytoplasmic movements and activity of actin filaments and are both temporary and variable in their length, shape, and number. However, in epithelial cells specialized for absorption, the apical surfaces present an array of projections called microvilli (L. villus, tuft). In cells such as those lining the small intestine, apical surfaces are densely covered with uniform microvilli, which are visible as a brush or striated border on these cells (Figure 4–8). The average microvillus is about 1 μm long and 0.1 μm wide, but with hundreds or thousands present on the end of each absorptive cell, the total surface area can be increased by 20- or 30-fold. Glycocalyx covering intestinal microvilli is thick and includes enzymes for digestion of certain macromolecules.
Each microvillus contains many bundles of actin filaments capped and cross-linked to each other and to the surrounding plasma membrane by many different actin-binding proteins (Figure 4–8). Although microvilli are often relatively stable, the microfilament arrays are dynamic and undergo various myosin-based movements, which help maintain optimal conditions for absorption via numerous channels, receptors, and other proteins in the plasmalemma. The actin filaments insert into the terminal web of similar filaments at the base of the microvilli.
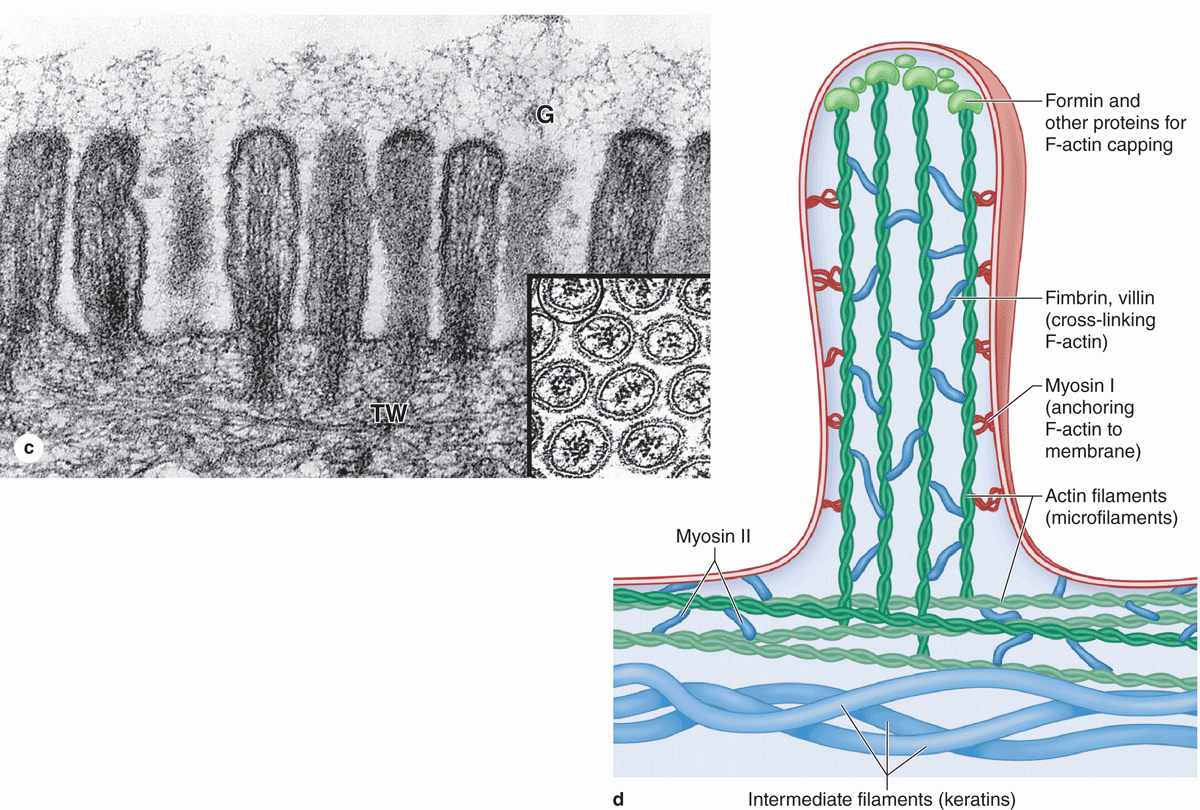
FIGURE 4–8 Microvilli.
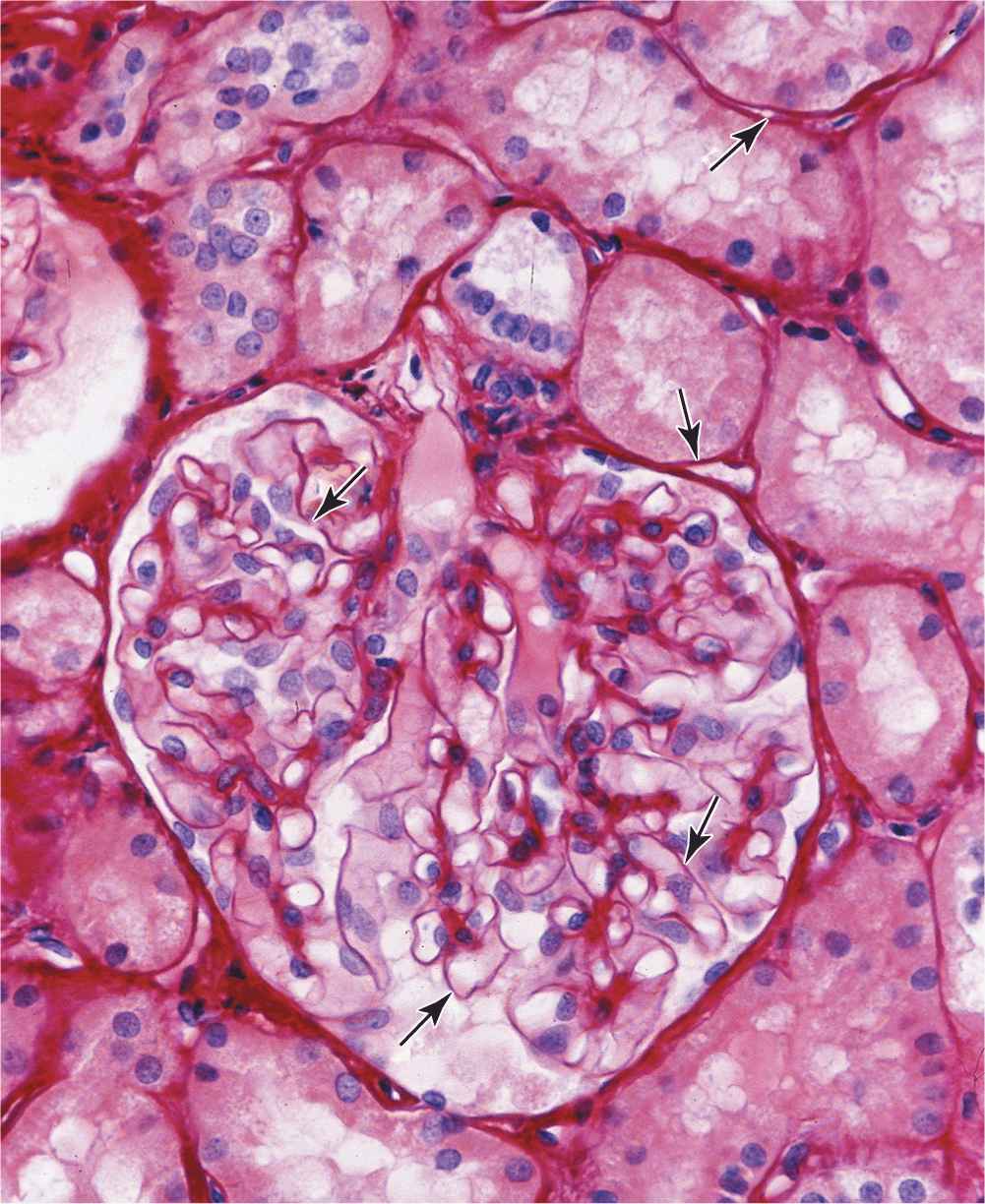
 MEDICAL APPLICATION
MEDICAL APPLICATION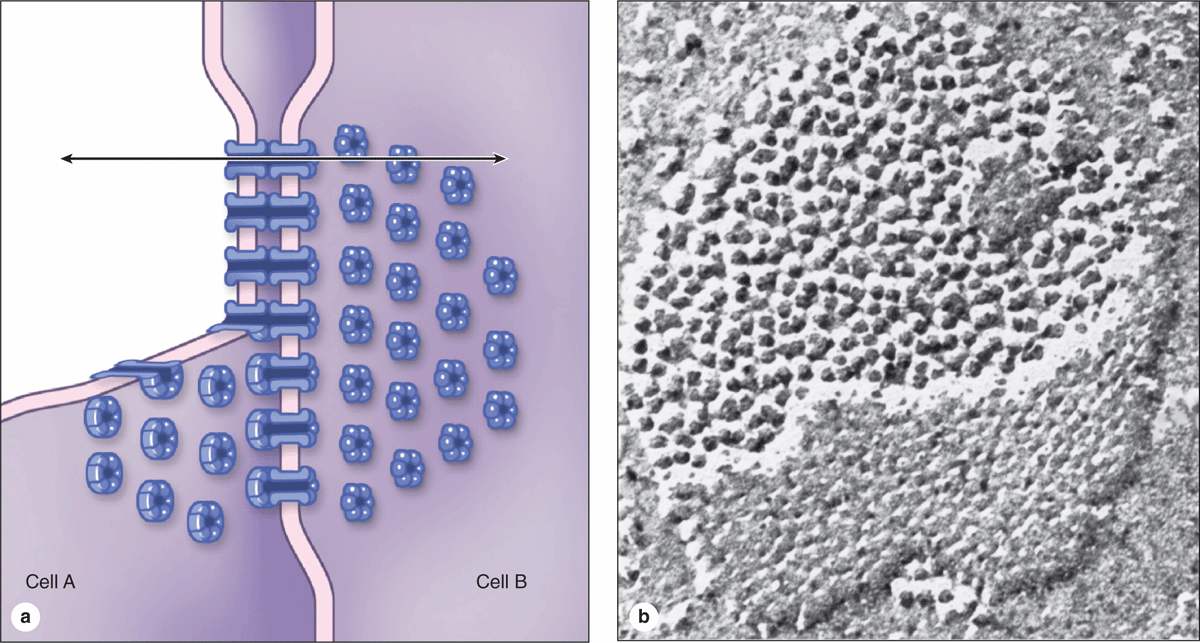
 MEDICAL APPLICATION
MEDICAL APPLICATION