Fig. 2.1
The adequacy algorithm shows The Paris System’s recommendation for the proper relationship between specimen source, cytological diagnosis, urine volume, urothelial cellularity, and obscuring features. Obscuring features include non-urothelial cells such as vaginal contaminants, bacteria, acute inflammation, sperm, and crystals which, when copious, may obscure the finding and characterization of urothelial cells. “*”: Cut-offs for appropriate benign urothelial cellularity should be validated for both instrumented and non-instrumented sources
The adequacy algorithm does not currently take into consideration the method used for preparing urinary tract specimens. This is intentional given the incomplete nature of evidence on this topic (see Chap. 10). Based on the established differences in gynecological cytopathology adequacy criteria, which depend on specimen preparation type, we expect to see preparation-dependent cut-offs at each point in the algorithm. However, the generic approach to adequacy should be uniform regardless of the specimen preparation method used in any particular laboratory.
Volume and Adequacy
There is a common misperception that cells in body fluids are solutes in a homogenous solution. However, urine is a heterogeneous mixture of non-solute particulates: crystals, microorganisms, decaying cell remnants, and cells. It is heterogeneous because the particulates are not evenly distributed throughout the volume. Cells are denser than most aqueous solutions, so they sink. If only the supernatant, which is acellular or paucicellular, is examined because the first drops of a urine stream were not captured in the specimen container or because the specimen was inadvertently decanted, the results will be suboptimal.
Volume-only factors into the adequacy of urine specimens for voided urines in the urinary tract adequacy algorithm for obvious reasons—instrumented specimens have artificial volumes, and their adequacy is based on operator skill and measured by cellularity. Conversely urine volume is important for voided urines for two main reasons (Fig. 2.2). First, in at least one study [2] there is a clear correlation between low-volume specimens and the lack of malignant diagnoses, suggesting that some benign diagnoses in small-volume specimens are due to undersampling of the potential voided urine volume rather than an absence of disease. This phenomenon is well known in effusion cytology, a field in which there can be up to a fivefold difference in the malignancy prevalence that is only explainable on the basis of the volume submitted to the laboratory. Second, the volume received by a laboratory can be evidence of specimen manipulation at some point in the collection. In a single study, the data suggests that a cut-off of 30 mL is appropriate in a laboratory that uses SurePath® preparation performed on fresh unfixed voided urines [2].
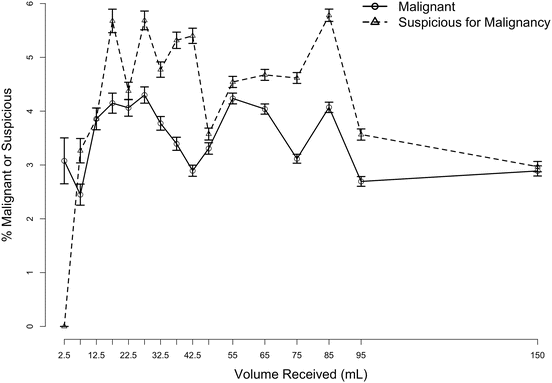





Fig. 2.2
Relationship of volume to the prevalence of malignant and suspicious diagnosis for demographically comparable populations. There were no suspicious samples in the lowest volume bin, and there was a nearly linear increase in the suspicious fraction up to the bin centered on 15 mL. A specimen was nearly twofold more likely to be suspicious when more than 15 mL was received than when less than 5 mL was received. The global maximum for the malignancy fraction was seen at 27.5 mL (range: 25–30 mL) which was also the location of a local maximum for the suspicious fraction. The global maximum for the suspicious curve was centered at 85 mL, and the difference between the global maximum (5.8, 95 % CI: 5.6–5.9) and the local maximum at 27.5 mL (5.7, 95 % CI: 5.5–5.9) was not statistically significant. Based on this analysis, we concluded that at least 30 mL are necessary to consider a urine specimen fully adequate when processed with SurePath (From VandenBussche et al. [2] with kind permission of John Wiley & Sons)Copyright © 1999-2015 John Wiley & Sons, Inc.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


