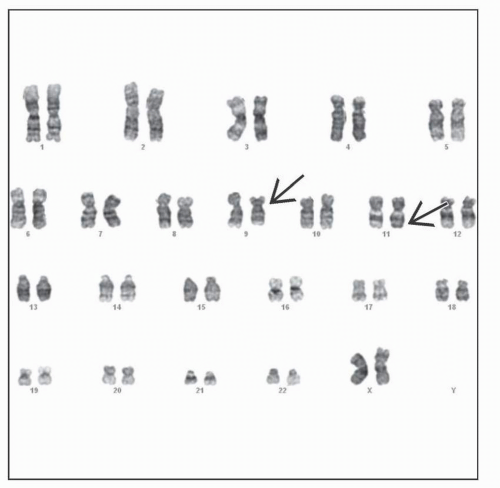
Acute Myeloid Leukemia with t(9;11)(p22;q23), MLLT3-MLL
Kaaren K. Reichard, MD
Key Facts
Terminology
Only AML with MLL translocation diagnosed as distinct biologic subtype
Clinical Issues
Extramedullary disease
Skin, CNS
Disseminated intravascular coagulation
Microscopic Pathology
≥ 20% blasts/blast equivalents in PB &/or BM
Typically monoblasts and promonocytes
Hypercellular bone marrow
Minimal residual hematopoiesis
Ancillary Tests
Cytochemistry
MPO(-), NSE(+)
Immunophenotyping
Monocytic differentiation: CD4 (weak +), CD33 (bright +), CD36/CD64 coexpression, CD14 and CD163 (variable)
Blast markers often negative: CD34, CD117
Genetics
Cytogenetics reveal classic translocation
FISH useful to document MLL rearrangement
Molecular detection of MLL-MLLT3 fusion for MRD
Top Differential Diagnoses
G-CSF/GM-CSF therapy
Nonclonal
CMML
< 20% blasts/blast equivalents
Therapy-related myeloid neoplasm with t(9;11)
History of chemotherapy or radiation
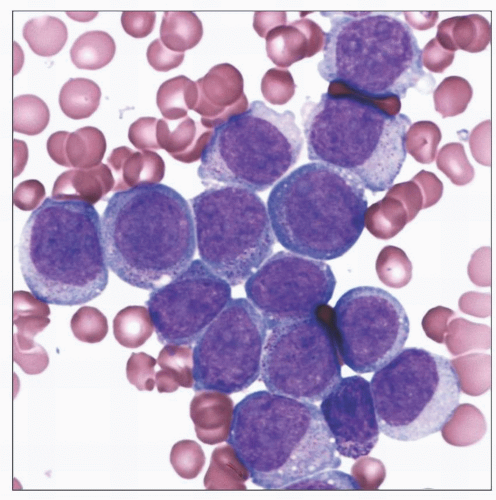 This bone marrow aspirate smear demonstrates the classic cytology of AML with t(9;11). The cells are monoblasts with an abundant rim of cytoplasm containing MPO-negative azurophilic granules. |
TERMINOLOGY
Abbreviations
AML with t(9;11)
Synonyms
Acute myeloid leukemia with MLL/11q23 abnormality
Definitions
Acute myeloid leukemia with t(9;11)(p22;q23) or variant &/or molecular evidence of MLL-MLLT3 fusion
Strong association with AML M5 and M4 subtypes (FAB classification)
Usually AML with monoblastic/monocytic differentiation
Blasts/blast equivalents ≥ 20%
Monoblasts and promonocytes
ETIOLOGY/PATHOGENESIS
Epidemiology
May occur at any age
˜ 5-20% of pediatric AML
˜ 4-5% of adult AML
Rare cases may originate from pre-B-cell harboring MLL-MLLT3 fusion
MLL
Mixed lineage leukemia gene
Human homologue of the trithorax gene in Drosophila
Encodes DNA-binding protein
Transcription regulatory factor (e.g., HOX genes)
Methylates histone H3
Chromatin remodeling
Plays major role in hematopoiesis
MLLT3
a.k.a. AF9
Involved in erythroid and megakaryocytic production
Involved in regulation of transcription
CLINICAL ISSUES
Presentation
Leukocytosis
Blood abnormality, NOS
Monocytosis
Extramedullary disease
Skin involvement, CNS symptoms, gingival hyperplasia
Disseminated intravascular coagulation (DIC)
Treatment
Drugs
Chemotherapy
Prognosis
Intermediate compared with other AMLs
Better than AMLs with other MLL (non MLLT3) rearrangements
Minimal residual disease monitoring
Early achievement of molecular negativity for MLL-MLLT3
May portend longer remission status
Increasing MLL-MLLT3 transcripts
May herald early disease relapse
MICROSCOPIC PATHOLOGY
Predominant Pattern/Injury Type
Diffuse
Predominant Cell/Compartment Type
Blast
Hematopoietic stem cell
Morphology
Peripheral blood
Variable WBC count
Often leukocytosis
Anemia
Thrombocytopenia
Circulating monoblasts and promonocytes
Usually > 20% of WBCs
Circulating myeloid blasts
Some cases show variable, usually small (< 5-10%) population of otherwise typical myeloblasts
Monoblasts
Large
Round nuclear contours
Fine chromatin
Prominent nucleolus
Abundant variably basophilic cytoplasm
Cytoplasmic azurophilic granules (MPO negative)
No Auer rods
Promonocytes
Intermediate to large
Smaller, variably prominent nucleoli
Abundant lightly basophilic/bluer cytoplasm
Gently lobulated nuclei with delicate nuclear folding
More even/dispersed chromatin pattern than typical monocyte
No significant granulocytic or platelet dysplasia
Bone marrow aspirate smears
Hypercellular
Usually sheets of monoblasts and promonocytes
Comprise > 80% of marrow nucleated cells
No significant background dysplasia
No significant basophilia or eosinophilia
Bone marrow core biopsy
Hypercellular
Predominant population of blasts/promonocytes
May see scattered residual hematopoietic elements
No significant, disease-related bony changes
Extramedullary Disease
Myeloid sarcoma
Sites most affected: Skin, gingiva, CNS
ANCILLARY TESTS
Cytochemistry
Myeloperoxidase (MPO)
Typically negative in monoblasts and promonocytes
Nonspecific esterase (NSE) (α naphthyl butyrate esterase)
Typically diffusely positive in monoblasts and promonocytes
Flow Cytometry
Monoblasts and promonocytes
Immature (blast) markers: CD34 and CD117 usually negative
Myeloid-associated markers: Positive for CD13 (variable), CD33 (bright)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree