DISEASE STATES ASSOCIATED WITH ABDOMINAL PAIN (ACUTE AND CHRONIC)
 Definition
Definition
Acute abdomen is defined as an episode of severe abdominal pain that lasts several hours or longer and requires medical attention. The acute abdomen usually, but not necessarily, has a surgical cause. However, the term “acute abdomen” should not be equated with a need for emergency surgery. The history and physical examination remain the most important aspects of diagnosis. The key feature in the evaluation of patients with acute abdomen is early diagnosis.
 Differential Diagnosis
Differential Diagnosis
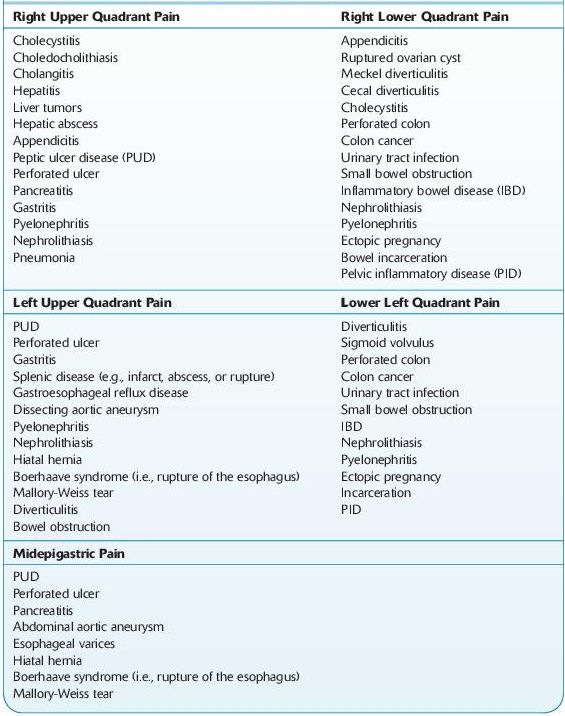
 The differential diagnosis of an acute abdomen is most appropriately considered by its anatomic location (Table 5-1).
The differential diagnosis of an acute abdomen is most appropriately considered by its anatomic location (Table 5-1).
 Common gynecologic causes of lower quadrant pain include mittelschmerz, ovarian cyst, endometriosis, fibroids, ovarian torsion, pelvic inflammatory disease, ovarian tumor, ectopic pregnancy, infection of the uterus, threatened abortion, and round ligament pain secondary to pregnancy.
Common gynecologic causes of lower quadrant pain include mittelschmerz, ovarian cyst, endometriosis, fibroids, ovarian torsion, pelvic inflammatory disease, ovarian tumor, ectopic pregnancy, infection of the uterus, threatened abortion, and round ligament pain secondary to pregnancy.
 Medical conditions that may present as acute abdomen are many. Common examples include lower lobe pneumonias, acute myocardial infarction (MI), DKA, acute hepatitis, porphyria, adrenal hemorrhage, and musculoskeletal problems. Appendicitis is a clinical diagnosis. The triad of right lower quadrant pain, anorexia, and leukocytosis is the most sensitive diagnostic tool. Nausea and vomiting usually follow the onset of pain. The patient may have a low-grade fever and mild leukocytosis. Fevers with higher temperatures or increased WBC counts suggest perforation.
Medical conditions that may present as acute abdomen are many. Common examples include lower lobe pneumonias, acute myocardial infarction (MI), DKA, acute hepatitis, porphyria, adrenal hemorrhage, and musculoskeletal problems. Appendicitis is a clinical diagnosis. The triad of right lower quadrant pain, anorexia, and leukocytosis is the most sensitive diagnostic tool. Nausea and vomiting usually follow the onset of pain. The patient may have a low-grade fever and mild leukocytosis. Fevers with higher temperatures or increased WBC counts suggest perforation.
 Thirty percent of patients with appendicitis have an elevated WBC count, whereas 95% have a left shift.
Thirty percent of patients with appendicitis have an elevated WBC count, whereas 95% have a left shift.
 The intensity of pain is somewhat in proportion to the degree of irritation to the parietal peritoneum. Therefore, a retrocecal appendix (which is the most common location) may cause only a dull ache, given the lack of contact with the parietal peritoneum.
The intensity of pain is somewhat in proportion to the degree of irritation to the parietal peritoneum. Therefore, a retrocecal appendix (which is the most common location) may cause only a dull ache, given the lack of contact with the parietal peritoneum.
TABLE 5–1. Differential Diagnosis of the Acute Abdomen
 Laboratory Findings
Laboratory Findings
 Laboratory studies are undertaken to support a clinical hypothesis. The evaluation generally includes a CBC, liver chemistries, amylase and lipase, coagulation profile, urinalysis, and urine pregnancy test.
Laboratory studies are undertaken to support a clinical hypothesis. The evaluation generally includes a CBC, liver chemistries, amylase and lipase, coagulation profile, urinalysis, and urine pregnancy test.
 Lactic acid level should be obtained for patients with suspected ischemic bowel. An elevated level is associated with tissue hypoperfusion.
Lactic acid level should be obtained for patients with suspected ischemic bowel. An elevated level is associated with tissue hypoperfusion.
 Beta-hCG levels must be obtained for all women of childbearing age to exclude the possibility of ectopic pregnancy.
Beta-hCG levels must be obtained for all women of childbearing age to exclude the possibility of ectopic pregnancy.
 Radiographic studies:
Radiographic studies:
 Chest radiograph should be obtained on all patients with acute abdomen to rule out free air. Pneumonia may present as an acute abdomen.
Chest radiograph should be obtained on all patients with acute abdomen to rule out free air. Pneumonia may present as an acute abdomen.
 Abdominal radiograph is most effective in detecting either bowel obstruction or pneumoperitoneum. An upright and supine view is necessary.
Abdominal radiograph is most effective in detecting either bowel obstruction or pneumoperitoneum. An upright and supine view is necessary.
 Appendicolith can be seen in 15% of patients with appendicitis, whereas renal stones may also be visualized up to 85% of the time.
Appendicolith can be seen in 15% of patients with appendicitis, whereas renal stones may also be visualized up to 85% of the time.
 Other radiographic findings of acute appendicitis include right lower quadrant ileus, loss of psoas shadow, deformity of the cecal outline, free air, and soft tissue density.
Other radiographic findings of acute appendicitis include right lower quadrant ileus, loss of psoas shadow, deformity of the cecal outline, free air, and soft tissue density.
 Abdominal ultrasound is the study of choice in patients with possible acute cholecystitis or ovarian cyst. A sonographic Murphy sign is more sensitive than a clinical Murphy sign for acute cholecystitis. An inflamed appendix can be visualized with compression ultrasound (sensitivity ranges from 80 to 90%).
Abdominal ultrasound is the study of choice in patients with possible acute cholecystitis or ovarian cyst. A sonographic Murphy sign is more sensitive than a clinical Murphy sign for acute cholecystitis. An inflamed appendix can be visualized with compression ultrasound (sensitivity ranges from 80 to 90%).
 CT can also be used to diagnose appendicitis in patients whose clinical symptoms are ambiguous.
CT can also be used to diagnose appendicitis in patients whose clinical symptoms are ambiguous.
 Air in the appendix or a normal-appearing contrast-filled appendix virtually rules out the diagnosis of appendicitis.
Air in the appendix or a normal-appearing contrast-filled appendix virtually rules out the diagnosis of appendicitis.
 CT will provide an alternate diagnosis in 15% of patients when assessing for appendicitis.
CT will provide an alternate diagnosis in 15% of patients when assessing for appendicitis.
 Arteriography is the test of choice for patients with suspected mesenteric ischemia.
Arteriography is the test of choice for patients with suspected mesenteric ischemia.
 DISEASE STATES ASSOCIATED WITH ABDOMINAL PAIN
DISEASE STATES ASSOCIATED WITH ABDOMINAL PAIN
DISORDERS OF THE ESOPHAGUS
MALLORY-WEISS SYNDROME
 Definition
Definition
Mallory-Weiss syndrome is characterized by spontaneous cardioesophageal laceration, usually caused by excessive retching. Laboratory findings are due to hemorrhage from cardioesophageal laceration.
PERFORATION OF THE ESOPHAGUS, SPONTANEOUS
In spontaneous perforation, gastric contents are found in thoracocentesis fluid.
PLUMMER-VINSON SYNDROME
 Definition
Definition
Plummer-Vinson syndrome is an iron deficiency anemia associated with dysphagia, atrophic gastritis, glossitis, and so on. It carries an increased risk of cancer of the esophagus and hypopharynx.
DISORDERS OF THE STOMACH
GASTRITIS, CHRONIC
 A diagnosis of chronic gastritis depends on biopsy of gastric mucosa.
A diagnosis of chronic gastritis depends on biopsy of gastric mucosa.
 Atrophic (Type A Gastritis, Autoimmune Type)
Atrophic (Type A Gastritis, Autoimmune Type)
 Gastric antrum is spared.
Gastric antrum is spared.
 Parietal cell antibodies and intrinsic factor antibodies help identify those patients prone to pernicious anemia (PA).
Parietal cell antibodies and intrinsic factor antibodies help identify those patients prone to pernicious anemia (PA).
 Characteristics include the following:
Characteristics include the following:
 Achlorhydria
Achlorhydria
 Vitamin B12 –deficient megaloblastosis
Vitamin B12 –deficient megaloblastosis
 Hypergastrinemia (due to hyperplasia of gastrin-producing cells)
Hypergastrinemia (due to hyperplasia of gastrin-producing cells)
 Gastric carcinoids
Gastric carcinoids
 Low serum pepsinogen I concentrations
Low serum pepsinogen I concentrations
 Laboratory findings may be due to other accompanying autoimmune diseases (e.g., Hashimoto thyroiditis, Addison disease, Graves disease, myasthenia gravis, hypoparathyroidism, type 1 DM).
Laboratory findings may be due to other accompanying autoimmune diseases (e.g., Hashimoto thyroiditis, Addison disease, Graves disease, myasthenia gravis, hypoparathyroidism, type 1 DM).
 Nonatrophic (Type B Gastritis)
Nonatrophic (Type B Gastritis)
 Gastric antrum is involved.
Gastric antrum is involved.
 Anemia caused by iron deficiency, and malabsorption may occur.
Anemia caused by iron deficiency, and malabsorption may occur.
 Helicobacter pylori infection is detectable in approximately 80% of patients with peptic ulcer and chronic gastritis. Diagnosis is by biopsy, culture, direct Gram staining, urease breath test, and serologic tests.
Helicobacter pylori infection is detectable in approximately 80% of patients with peptic ulcer and chronic gastritis. Diagnosis is by biopsy, culture, direct Gram staining, urease breath test, and serologic tests.
 Hypogastrinemia is caused by destruction of gastrin-producing cells in the antrum.
Hypogastrinemia is caused by destruction of gastrin-producing cells in the antrum.
 Chronic antral gastritis is consistently present in patients with benign gastric ulcer.
Chronic antral gastritis is consistently present in patients with benign gastric ulcer.
 Gastric acid studies are of limited value. Severe hypochlorhydria or achlorhydria after maximal stimulation usually denotes mucosal atrophy.
Gastric acid studies are of limited value. Severe hypochlorhydria or achlorhydria after maximal stimulation usually denotes mucosal atrophy.
 Other Causes
Other Causes
 Infections (other bacteria [syphilis], viral [e.g., CMV], parasitic [e.g., anisakiasis], fungal)
Infections (other bacteria [syphilis], viral [e.g., CMV], parasitic [e.g., anisakiasis], fungal)
 Chemical (e.g., NSAIDs, bile reflux, other drugs)
Chemical (e.g., NSAIDs, bile reflux, other drugs)
 Lymphocytic gastritis
Lymphocytic gastritis
 Eosinophilic gastroenteritis
Eosinophilic gastroenteritis
 Noninfectious granulomatous (e.g., sarcoidosis, Crohn disease)
Noninfectious granulomatous (e.g., sarcoidosis, Crohn disease)
 Ménétrier disease
Ménétrier disease
 Radiation
Radiation
CARCINOMA OF THE STOMACH
 Laboratory Findings
Laboratory Findings
Carcinoma of the stomach should always be searched for by periodic prophylactic screening in high-risk patients, especially those with PA, gastric atrophy, or gastric polyps. Cytology: Exfoliative cytology positive in 80% of patients; false-positive result in <2%.
Tumor markers: Increased serum CEA (>5 ng/dL) in 40–50% of patients with metastases and 10–20% of patients with surgically resectable disease. May be useful for postoperative monitoring for recurrence or to estimate metastatic tumor burden. Increased serum AFP and CA 19-9 in 30% of patients, usually incurable. Markers are not useful for early detection.
Gastric analysis: Normal in 25% of patients. Hypochlorhydria in 25% of patients. Achlorhydria following histamine or betazole in 50% of patients.
Core laboratory: Anemia due to chronic blood loss. Occult blood in stool.
DISORDERS OF THE PANCREAS
CARCINOMA OF THE PANCREAS
BODY OR TAIL
 Laboratory Findings
Laboratory Findings
Imaging studies: Most useful tests are ultrasound or CT scanning followed by ERCP (at which time fluid is also obtained for cytologic and pancreatic function studies). This combination will correctly diagnose or rule out cancer of the pancreas in ≥90% of cases. ERCP with brush cytology has S/S = ≤25%/≤100%. Radioisotope scanning of the pancreas may be done (75Se) for lesions >2 cm.
Histology: Ultrasound-guided needle biopsy has reported sensitivity of 80–90%; false positives are rare.
Tumor markers: Serum markers for tumor (CA 19-9, CEA, and so on) are often normal. In carcinoma of the pancreas, CA 19-9 has S/S = 70%/87%, PPV = 59%, and NPV = 92%; there is no difference in sensitivity between local disease and metastatic disease. Often normal in early stages, they are not useful for screening. Increased values may help differentiate benign disease from cancer. Declines to normal in 3–6 months if cancer is completely removed so may be useful for prognosis and followup. Detects tumor recurrence 2–20 weeks before clinical evidence. Not specific for pancreas because high levels may also occur in other GI cancers, especially those affecting the colon and bile duct. CEA level in bile (obtained by percutaneous transhepatic drainage) was reported increased in 76% of a small group of cases.
Testosterone: Dihydrotestosterone ratio <5 (normal approximately 10) in >70% of men with pancreatic cancer (due to increased conversion by tumor); less sensitive but more specific than CA 19-9 and present in higher proportion of stage I tumors.
Serum amylase and lipase: May be slightly increased in early stages (<10% of cases); with later destruction of the pancreas, they are normal or decreased. They may increase following secretin–pancreozymin stimulation before destruction is extensive; therefore, the increase is less marked with a diabetic glucose tolerance curve. Serum amylase response is less reliable. See Serum Glycoprotein 2.
Glucose tolerance: Curve is of the diabetic type, with overt diabetes in 20% of patients with pancreatic cancer. Flat blood sugar curve with IV tolbutamide tolerance test indicates destruction of islet cell tissue. Unstable, insulin-sensitive diabetes that develops in an older man should arouse suspicion of carcinoma of the pancreas.
Serum LAP: Increased (>300 U) in 60% of patients with carcinoma of the pancreas due to liver metastases or biliary tract obstruction. It may also be increased in chronic liver disease.
Other: Triolein-131I test demonstrates pancreatic duct obstruction with absence of lipase in the intestine, causing flat blood curves and increased stool excretion.
HEAD (SEE JAUNDICE)
 The abnormal pancreatic function tests and increased tumor markers that occur with carcinoma of the body of the pancreas may be evident.
The abnormal pancreatic function tests and increased tumor markers that occur with carcinoma of the body of the pancreas may be evident.
 Laboratory Findings
Laboratory Findings
Core laboratory: Serum bilirubin is increased (12–25 mg/dL), mostly conjugated (increase persistent and nonfluctuating). Serum ALP is increased. Both urine and stool urobilinogen are absent. Increased serum cholesterol (usually >300 mg/dL) with esters not decreased. Other liver function tests are usually normal. See Serum Glycoprotein 2.
Hematology: Increased prothrombin time (PT); normal after IV vitamin K administration.
Other: Secretin–cholecystokinin stimulation evidences duct obstruction when duodenal intubation shows decreased volume of duodenal contents (<10 mL/10-minute collection period) with usually normal bicarbonate and enzyme levels in duodenal contents. Acinar destruction (as in pancreatitis) shows normal volume (20–30 mL/10-minute collection period), but bicarbonate and enzyme levels may be decreased. Abnormal volume, bicarbonate, or both are found in 60–80% of patients with pancreatitis or cancer. In carcinoma, the test result depends on the relative extent and combination of acinar destruction and of duct obstruction.
Histology: Cytologic examination of duodenal contents shows malignant cells in 40% of patients. Malignant cells may be found in up to 80% of patients with periampullary cancer.
CYSTIC FIBROSIS OF THE PANCREAS
Core laboratory: Hypochloremic metabolic alkalosis and hypokalemia. Serum protein electrophoresis shows increasing IgG and IgA with progressive pulmonary disease; IgM and IgD are not appreciably increased. Serum albumin is often decreased (because of hemodilution due to cor pulmonale; may be found before cardiac involvement is clinically apparent). Serum chloride, sodium, potassium, calcium, and phosphorus are normal unless complications occur (e.g., chronic pulmonary disease with accumulation of CO2 ; massive salt loss due to sweating may cause hyponatremia). Urine electrolytes are normal. Excessive loss of electrolytes in sweat and stool. Impaired glucose intolerance in approximately 40% of patients with glycosuria, and hyperglycemia in 8% precedes DM. Protein–calorie malnutrition, hypoproteinemia; fat malabsorption with vitamin deficiency. Stool and duodenal fluid show lack of trypsin digestion of x-ray film gelatin; useful screening test up to age 4; decreased chymotrypsin production.
Saliva findings: Submaxillary saliva is more turbid, with increased calcium, total protein, amylase, chloride, and sodium but not potassium. These changes are not generally found in parotid saliva.
Other findings: Overt liver disease, including cirrhosis, fatty liver, bile duct strictures, and cholelithiasis, in ≤5% of cases. Meconium ileus during early infancy. Chronic or acute and recurrent pancreatitis. Pancreatic insufficiency frequency by age 1 >90%; in adults >95%. Increased incidence of GI tract cancers. GU tract abnormalities with aspermia in 98% due to obstructive changes in the vas deferens and epididymis are confirmed by testicular biopsy.
MACROAMYLASEMIA IN VIVO ARTIFACT
 Definition
Definition
Complex of amylase with IgA, IgG, or other high molecular weight plasma proteins that cannot filter through the glomerulus due to its large size associated with no specific symptoms or disease states.
 Laboratory Findings
Laboratory Findings
Core laboratory: Serum lipase is normal; normal pancreatic-to-salivary amylase ratio. Urine amylase normal or low. Serum amylase persistently increased (often 1–4× normal) without apparent cause. Amylase–creatinine clearance ratio <1% with normal renal function is very useful for this diagnosis; should make the clinician suspect this diagnosis. Macroamylase is identified in serum by special gel filtration or ultracentrifugation technique.
 Limitations
Limitations
 Macroamylase may be found in approximately 1% of randomly selected patients and 2.5% of persons with increased serum amylase level. Same findings may also occur in patients with normal molecular weight hyperamylasemia in which excess amylase is principally salivary gland isoamylase types 2 and 3.
Macroamylase may be found in approximately 1% of randomly selected patients and 2.5% of persons with increased serum amylase level. Same findings may also occur in patients with normal molecular weight hyperamylasemia in which excess amylase is principally salivary gland isoamylase types 2 and 3.
PANCREATITIS
PANCREATITIS, ACUTE
 Laboratory Findings Lipase:
Laboratory Findings Lipase:
Serum lipase increases within 3–6 hours with peak at 24 hours and usually returns to normal over a period of 8–14 days; is superior to amylase; increases to a greater extent and may remain elevated for up to 14 days after amylase returns to normal. In patients with signs of acute pancreatitis, pancreatitis is highly likely (clinical specificity = 85%) when lipase ≥5× upper reference limit (URL); if values change significantly with time, and if amylase and lipase changes are concordant. (Lipase should always be determined whenever amylase is determined.) Urinary lipase is not clinically useful. It has been suggested that a lipase:amylase ratio >3 (and especially >5) indicates alcoholic rather than nonalcoholic pancreatitis. If lipase ≥5× URL, acute pancreatitis or organ rejection is highly likely but unlikely if <3× URL (Figure 5-1).
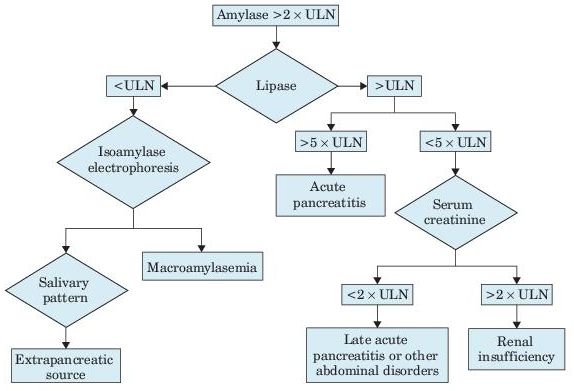
Figure 5–1 Algorithm for increased serum amylase and lipase. ULN, upper limit of normal.
Amylase: Increase begins in 3–6 hours, rises rapidly within 8 hours in 75% of patients, reaches maximum in 20–30 hours, and may persist for 48–72 hours; >95% sensitivity during the first 12–24 hours. The increase may be ≤40× normal, but the height of the increase and rate of fall do not correlate with the severity of the disease, prognosis, or rate of resolution. In patients with signs of acute pancreatitis, amylase >3× ULN or >600 Somogyi units/dL is very suggestive of acute pancreatitis. An increase >7–10 days suggests an associated cancer of the pancreas or pseudocyst, pancreatic ascites, or nonpancreatic etiology. Similar high values may occur in obstruction of the pancreatic duct; they tend to fall after several days; ≤19% of patients with acute pancreatitis (especially when seen more than 2 days after onset of symptoms) may have normal values, especially with an alcoholic etiology and longer duration of symptoms, even when dying of acute pancreatitis. May also be normal in relapsing chronic pancreatitis and patients with hypertriglyceridemia (technical interference with test). Frequently normal in acute alcoholic pancreatitis. Acute abdomen due to GI infarction or perforation rather than acute pancreatitis is suggested by only moderate increase in serum amylase and lipase (<3× URL), evidence of bacteremia. Of patients with acute alcoholic intoxication, 10–40% have elevated serum amylase (about half are salivary type); they often present with abdominal pain, but increased serum amylase is usually <3× URL. Levels >25× URL indicate metastatic tumor rather than pancreatitis. Serum pancreatic isoamylase can distinguish elevations due to salivary amylase that may account for 25% of all elevated values. (In healthy persons, 40% of total serum amylase is pancreatic type and 60% is salivary type.) Only slight increase in serum amylase and lipase values suggests a different diagnosis than acute pancreatitis. Many drugs increase both amylase and lipase in serum.
Increased urinary amylase tends to reflect serum changes by a time lag of 6–10 hours, but sometimes, increased urine levels are higher and of longer duration than serum levels. The 24-hour level may be normal even when some of the 1-hour specimens show increased values. Amylase levels in hourly samples of urine may be useful. Ratio of amylase clearance to creatinine clearance is increased (>5%) and avoids the problem of timed urine specimens; also increased in any condition that decreases tubular reabsorption of amylase (e.g., severe burns, DKA, chronic renal insufficiency, multiple myeloma, acute duodenal perforation). Considered not specific and now discouraged by some but still recommended by others.
Calcium: Serum level is decreased in severe cases 1–9 days after onset (due to binding to soaps in fat necrosis). The decrease usually occurs after amylase and lipase levels have become normal. Tetany may occur. (Rule out hyperparathyroidism if serum calcium is high or fails to fall in hyperamylasemia of acute pancreatitis.)
Bilirubin: Serum levels may be increased when pancreatitis is of biliary tract origin but is usually normal in alcoholic pancreatitis. Serum ALP, ALT, and AST may increase and parallel serum bilirubin rather than amylase, lipase, or calcium levels. Marked amylase increase (e.g., >2,000 U/L) also favors biliary tract origin. Fluctuation >50% in 24 hours of serum bilirubin, ALP, ALT, and AST suggests intermittent biliary obstruction.
Trypsin: Serum level is increased. High sensitivity makes a normal value useful for excluding acute pancreatitis. But low specificity (increased in large proportion of patients with hepatobiliary, bowel, and other diseases and renal insufficiency; increased in 13% of patients with chronic pancreatitis, 50% with pancreatic carcinoma) and RIA technology limit utility.
CRP: Level peaks 3 days after onset of pain; at 48 hours, sensitivity = 65–100%, PPV = 37–77%. Level of 150 mg/L distinguishes mild from severe disease.
Laboratory criteria for severe disease or predictor of mortality:
 PaO2 <60 μmol/L
PaO2 <60 μmol/L
 Creatinine >2 mg/dL after rehydration
Creatinine >2 mg/dL after rehydration
 Blood glucose >250 mg/dL
Blood glucose >250 mg/dL
 Hemoconcentration (Hct >47% or failure to decrease in 24 hours after admission), but Hct may be decreased in severe hemorrhagic pancreatitis
Hemoconcentration (Hct >47% or failure to decrease in 24 hours after admission), but Hct may be decreased in severe hemorrhagic pancreatitis
 GI bleed >500 mL/24 hours
GI bleed >500 mL/24 hours
 Presence, volume, and color of peritoneal fluid
Presence, volume, and color of peritoneal fluid
 Methemalbumin may be increased in serum and ascitic fluid (AF) in hemorrhagic (severe) but not edematous (mild) pancreatitis; may distinguish these two conditions but not useful in diagnosis of acute pancreatitis.
Methemalbumin may be increased in serum and ascitic fluid (AF) in hemorrhagic (severe) but not edematous (mild) pancreatitis; may distinguish these two conditions but not useful in diagnosis of acute pancreatitis.
 WBC is slightly to moderately increased (10,000–20,000/μL).
WBC is slightly to moderately increased (10,000–20,000/μL).
 Glycosuria appears in 25% of patients.
Glycosuria appears in 25% of patients.
 Hypokalemia, metabolic alkalosis, or lactic acidosis may occur.
Hypokalemia, metabolic alkalosis, or lactic acidosis may occur.
Laboratory findings due to predisposing conditions (may be multiple):
 Alcohol abuse accounts for approximately 36% of cases.
Alcohol abuse accounts for approximately 36% of cases.
 Biliary tract disease accounts for 17% of cases.
Biliary tract disease accounts for 17% of cases.
 Idiopathic accounts for >36% of cases.
Idiopathic accounts for >36% of cases.
 Infections (especially viral such as mumps and coxsackievirus, CMV, and AIDS).
Infections (especially viral such as mumps and coxsackievirus, CMV, and AIDS).
 Trauma and postoperative factors account for >8% of cases.
Trauma and postoperative factors account for >8% of cases.
 Drugs (e.g., steroids, thiazides, azathioprine, estrogens, sulfonamides; children taking valproic acid) account for >5% of cases.
Drugs (e.g., steroids, thiazides, azathioprine, estrogens, sulfonamides; children taking valproic acid) account for >5% of cases.
 Hypertriglyceridemia (hyperlipidemia—types V, I, IV) accounts for 7% of cases.
Hypertriglyceridemia (hyperlipidemia—types V, I, IV) accounts for 7% of cases.
 Hypercalcemia from any cause.
Hypercalcemia from any cause.
 Tumors (pancreas, ampulla).
Tumors (pancreas, ampulla).
 Anatomic abnormalities of the ampullary region causing obstruction (e.g., annular pancreas, Crohn disease, duodenal diverticulum).
Anatomic abnormalities of the ampullary region causing obstruction (e.g., annular pancreas, Crohn disease, duodenal diverticulum).
 Hereditary.
Hereditary.
 Renal failure; renal transplantation.
Renal failure; renal transplantation.
 Miscellaneous (e.g., collagen vascular disease, pregnancy, ischemia, scorpion bites, parasites obstructing the pancreatic duct [Ascaris, fluke], Reye syndrome, fulminant hepatitis, severe hypotension, cholesterol embolization).
Miscellaneous (e.g., collagen vascular disease, pregnancy, ischemia, scorpion bites, parasites obstructing the pancreatic duct [Ascaris, fluke], Reye syndrome, fulminant hepatitis, severe hypotension, cholesterol embolization).
Laboratory findings due to complications:
 Pseudocysts of the pancreas.
Pseudocysts of the pancreas.
 Pancreatic infection or abscess diagnosed by increased WBC count, Gram staining, and culture of aspirate.
Pancreatic infection or abscess diagnosed by increased WBC count, Gram staining, and culture of aspirate.
 Polyserositis (peritoneal, pleural, pericardial, synovial surfaces). Ascites may develop cloudy or bloody or “prune juice” fluid, 0.5–2.0 L in volume, containing increased amylase with a level higher than that of serum amylase. No bile is evident (unlike in perforated ulcer). Gram stain shows no bacteria (unlike infarct of the intestine). Protein >3 g/dL and marked increase in amylase.
Polyserositis (peritoneal, pleural, pericardial, synovial surfaces). Ascites may develop cloudy or bloody or “prune juice” fluid, 0.5–2.0 L in volume, containing increased amylase with a level higher than that of serum amylase. No bile is evident (unlike in perforated ulcer). Gram stain shows no bacteria (unlike infarct of the intestine). Protein >3 g/dL and marked increase in amylase.
 Adult respiratory distress syndrome (with pleural effusion, alveolar exudate, or both) may occur in approximately 40% of patients; arterial hypoxemia is present.
Adult respiratory distress syndrome (with pleural effusion, alveolar exudate, or both) may occur in approximately 40% of patients; arterial hypoxemia is present.
 DIC.
DIC.
 Hypovolemic shock.
Hypovolemic shock.
 Others.
Others.
 Prognostic Laboratory Findings
Prognostic Laboratory Findings
 On admission
On admission
WBC >16,000/μL
Blood glucose >200 mg/dL
Serum LD >350 U/L
Serum AST >250 U/L
Age >55 years
 Within 48 hours
Within 48 hours
>10% decrease in Hct
Serum calcium <8.0 mg/dL
Increase in BUN >5 mg/dL
Arterial pO2 <60 mm Hg
Metabolic acidosis with base deficit >4 mEq/L
 Mortality
Mortality
1%, if 3 signs are positive
15%, if 3 to 4 signs are positive
40%, if 5 to 6 signs are positive
100%, if ≥7 signs are positive
 Degree of amylase elevation has no prognostic significance.
Degree of amylase elevation has no prognostic significance.
 CT scan, MRI, and ultrasound are useful for confirming diagnosis or identifying causes or other conditions.
CT scan, MRI, and ultrasound are useful for confirming diagnosis or identifying causes or other conditions.
Suggested Readings
Papachristou GI, Whitcomb DC. Inflammatory markers of disease severity in acute pancreatitis. Clin Lab Med. 2005;25:17.
Whitcomb DC. Acute pancreatitis. N Engl J Med. 2006;354:2142.
PANCREATITIS, CHRONIC
 See also Malabsorption.
See also Malabsorption.
 Laboratory Findings
Laboratory Findings
Laboratory findings are often normal.
Imaging studies: CT, ultrasound, and ERCP are most accurate for diagnosing and staging chronic pancreatitis. Radioactive scanning of the pancreas (selenium) yields variable findings in different clinics.
Cholecystokinin–secretin test: Measures the effect of IV administration of cholecystokinin and secretin on volume, bicarbonate concentration, and amylase output of duodenal contents and increase in serum lipase and amylase. This is the most sensitive and reliable test (gold standard) for chronic pancreatitis especially in the early stages. However, it is technically difficult and is often not performed accurately; gastric contamination must be avoided. Some abnormality occurs in >85% of patients with chronic pancreatitis. Amylase output is the most frequent abnormality. When all three are abnormal, there is a greater frequency of abnormality in the tests listed below.
 Normal duodenal contents:
Normal duodenal contents:
 Volume: 95–235 mL/hour
Volume: 95–235 mL/hour
 Bicarbonate concentration: 74–121 mEq/L
Bicarbonate concentration: 74–121 mEq/L
 Amylase output: 87,000–276,000 mg
Amylase output: 87,000–276,000 mg
 Serum amylase and lipase increase after administration of cholecystokinin and secretin in approximately 20% of patients with chronic pancreatitis. They are more often abnormal when duodenal contents are normal. Normally serum lipase and amylase do not rise above normal limits.
Serum amylase and lipase increase after administration of cholecystokinin and secretin in approximately 20% of patients with chronic pancreatitis. They are more often abnormal when duodenal contents are normal. Normally serum lipase and amylase do not rise above normal limits.
 Fasting serum amylase and lipase are increased in 10% of patients with chronic pancreatitis.
Fasting serum amylase and lipase are increased in 10% of patients with chronic pancreatitis.
Serum pancreolauryl test: Fluorescein dilaurate with breakfast is acted on by a pancreas-specific cholesterol ester hydrolase–releasing fluorescein, which is absorbed from gut and measured in serum; preceded by administration of secretin and followed by metoclopramide. Reported S/S = 82%/91%.
Glucose tolerance test (GTT): In 65% of patients with chronic pancreatitis and frank diabetes in >10% of patients with chronic relapsing pancreatitis. When GTT is normal in the presence of steatorrhea, the cause should be sought elsewhere than in the pancreas.
Laboratory findings due to malabsorption: Occurs when >90% of exocrine function is lost.
 Bentiromide test is usually abnormal with moderate to severe pancreatic insufficiency but often normal in early cases.
Bentiromide test is usually abnormal with moderate to severe pancreatic insufficiency but often normal in early cases.
 Schilling test may show mild malabsorption of vitamin B12 (no longer performed).
Schilling test may show mild malabsorption of vitamin B12 (no longer performed).
 Xylose tolerance test and small bowel biopsy are not usually done but are normal.
Xylose tolerance test and small bowel biopsy are not usually done but are normal.
 Chemical determination of fecal fat demonstrates steatorrhea. It is more sensitive than tests using triolein-131I.
Chemical determination of fecal fat demonstrates steatorrhea. It is more sensitive than tests using triolein-131I.
 Triolein-131I is abnormal in one third of patients with chronic pancreatitis.
Triolein-131I is abnormal in one third of patients with chronic pancreatitis.
 Starch tolerance test is abnormal in 25% of patients with chronic pancreatitis.
Starch tolerance test is abnormal in 25% of patients with chronic pancreatitis.
Laboratory findings due to chronic pancreatitis and pancreatic exocrine insufficiency:
 Alcohol in 60–70%
Alcohol in 60–70%
 Idiopathic in 30–40%
Idiopathic in 30–40%
 Obstruction of pancreatic duct (e.g., trauma, pseudocyst, pancreas divisum, cancer, or obstruction of duct or ampulla)
Obstruction of pancreatic duct (e.g., trauma, pseudocyst, pancreas divisum, cancer, or obstruction of duct or ampulla)
 Others occasionally (e.g., CF, primary hyperparathyroidism, heredity, malnutrition, miscellaneous [Z-E syndrome, Shwachman syndrome, alpha1 -antitrypsin deficiency, trypsinogen deficiency, enterokinase deficiency, hemochromatosis, parenteral hyperalimentation])
Others occasionally (e.g., CF, primary hyperparathyroidism, heredity, malnutrition, miscellaneous [Z-E syndrome, Shwachman syndrome, alpha1 -antitrypsin deficiency, trypsinogen deficiency, enterokinase deficiency, hemochromatosis, parenteral hyperalimentation])
PSEUDOCYST OF THE PANCREAS
 Laboratory Findings
Laboratory Findings
Imaging studies: Detected by ultrasound or CT scan.
Core laboratory: Serum conjugated bilirubin is increased (>2 mg/dL) in 10% of patients. Serum ALP is increased in 10% of patients. Fasting blood sugar is increased in <10% of patients.
Secretin–pancreozymin stimulation: Duodenal contents usually show decreased bicarbonate content (<70 mEq/L) but normal volume and normal content of amylase, lipase, and trypsin.
Pancreatic cyst fluid findings: High fluid viscosity and CEA indicate mucinous differentiation and exclude pseudocyst, serous cystadenoma, other nonmucinous cysts, or cystic tumors. Pancreatic enzymes, leukocyte esterase, and NB/70K are increased in pseudocyst fluid. Increased CA 72-4, CA 15-3, and tissue polypeptide antigen are markers of malignancy; if all are low, pseudocyst or serous cystadenoma is most likely. CA 125 is increased in serous cystadenoma.
Other: Laboratory findings due to conditions preceding acute pancreatitis are noted (e.g., alcoholism, trauma, duodenal ulcer, cholelithiasis), infection, perforation, and hemorrhage by erosion of blood vessel or into a viscus.
DYSPEPSIA AND PEPTIC ULCER DISEASE
 Definition
Definition
 Dyspepsia encompasses any or all of a great variety of upper abdominal symptoms, including upper abdominal pain or discomfort, nausea, bloating, heartburn, early satiety, regurgitation, and belching.
Dyspepsia encompasses any or all of a great variety of upper abdominal symptoms, including upper abdominal pain or discomfort, nausea, bloating, heartburn, early satiety, regurgitation, and belching.
 Nonulcerative dyspepsia is defined as persistent or recurrent abdominal pain or discomfort centered in the upper abdomen without definite structural or biochemical explanation. By definition, nonulcerative dyspepsia is a diagnosis of exclusion. Possible mechanisms include dysmotility of the stomach or small intestine, heightened visceral sensitivity, altered intestinal or gastric reflexes, and psychological distress.
Nonulcerative dyspepsia is defined as persistent or recurrent abdominal pain or discomfort centered in the upper abdomen without definite structural or biochemical explanation. By definition, nonulcerative dyspepsia is a diagnosis of exclusion. Possible mechanisms include dysmotility of the stomach or small intestine, heightened visceral sensitivity, altered intestinal or gastric reflexes, and psychological distress.
 Peptic ulcer disease (PUD)
Peptic ulcer disease (PUD)
 Epigastric abdominal pain is the most common symptom. Pain is nonradiating and is described as a “gnawing” or “hunger pain.” Pain occurs 1–2 hours postprandially and is relieved characteristically by food or antacids.
Epigastric abdominal pain is the most common symptom. Pain is nonradiating and is described as a “gnawing” or “hunger pain.” Pain occurs 1–2 hours postprandially and is relieved characteristically by food or antacids.
 Nocturnal pain is more specific for PUD and is due to the physiologic increase in acid secretion, which occurs in the early morning hours.
Nocturnal pain is more specific for PUD and is due to the physiologic increase in acid secretion, which occurs in the early morning hours.
 Asymptomatic:
Asymptomatic:
 Patients with PUD induced by NSAIDs are frequently asymptomatic.
Patients with PUD induced by NSAIDs are frequently asymptomatic.
 As many as 60% of patients who develop bleeding as a complication of PUD are also asymptomatic.
As many as 60% of patients who develop bleeding as a complication of PUD are also asymptomatic.
 Dyspepsia is typically a chronic relapsing condition. Between 65% and 86% of patients with dyspepsia will experience dyspeptic symptoms, at least intermittently, 2 to 3 years after the initial presentation. Long duration of symptoms and intermittent symptoms can also occur in PUD and esophagitis; therefore, these characteristics are not reassuring as to the absence of pathology.
Dyspepsia is typically a chronic relapsing condition. Between 65% and 86% of patients with dyspepsia will experience dyspeptic symptoms, at least intermittently, 2 to 3 years after the initial presentation. Long duration of symptoms and intermittent symptoms can also occur in PUD and esophagitis; therefore, these characteristics are not reassuring as to the absence of pathology.
 Gastroesophageal reflux disease (GERD) and dyspepsia have similar symptoms. Gastroesophageal reflux is a normal physiologic process that occurs daily in all individuals. GERD (expressed clinically as heartburn).
Gastroesophageal reflux disease (GERD) and dyspepsia have similar symptoms. Gastroesophageal reflux is a normal physiologic process that occurs daily in all individuals. GERD (expressed clinically as heartburn).
 Helicobacter pylori infection is clearly implicated in the etiology of recurrent PUD, yet its role in nonulcerative dyspepsia remains unclear. Between 30% and 60% of patients with nonulcerative dyspepsia have H. pylori. However, the background prevalence in the general population is also high.
Helicobacter pylori infection is clearly implicated in the etiology of recurrent PUD, yet its role in nonulcerative dyspepsia remains unclear. Between 30% and 60% of patients with nonulcerative dyspepsia have H. pylori. However, the background prevalence in the general population is also high.
 Recommended Tests
Recommended Tests
 Laboratory investigation may not be necessary in young patients (<45 years of age) who have a normal examination and no indicators for organic disease. The etiology of dyspepsia is presented in Table 5-2.
Laboratory investigation may not be necessary in young patients (<45 years of age) who have a normal examination and no indicators for organic disease. The etiology of dyspepsia is presented in Table 5-2.
 In older patients at increased risk, the minimal laboratory workup should include a CBC, electrolytes, calcium, and liver chemistries.
In older patients at increased risk, the minimal laboratory workup should include a CBC, electrolytes, calcium, and liver chemistries.
 Thyroid tests, hCG, amylase, and stool studies should be ordered if specific features of the history or examination are suggestive.
Thyroid tests, hCG, amylase, and stool studies should be ordered if specific features of the history or examination are suggestive.
 Additional studies
Additional studies
 Upper endoscopy (i.e., esophagogastroduodenoscopy [EGD]): In the majority of cases, this is the study of first choice when further evaluation of dyspepsia is required, including the ability to obtain biopsies. As many as two thirds of endoscopies are completely normal in younger patients (i.e., <45 years of age). Therefore, it is best applied to older patients and to younger patients with classic symptoms.
Upper endoscopy (i.e., esophagogastroduodenoscopy [EGD]): In the majority of cases, this is the study of first choice when further evaluation of dyspepsia is required, including the ability to obtain biopsies. As many as two thirds of endoscopies are completely normal in younger patients (i.e., <45 years of age). Therefore, it is best applied to older patients and to younger patients with classic symptoms.
 Upper GI radiography: This test is less accurate than upper endoscopy and cannot provide tissue diagnosis. It is best reserved for situations where endoscopy expertise is unavailable, for patients who refuse endoscopy or have low pretest probability of disease, and in situations where endoscopy might be considered unsafe.
Upper GI radiography: This test is less accurate than upper endoscopy and cannot provide tissue diagnosis. It is best reserved for situations where endoscopy expertise is unavailable, for patients who refuse endoscopy or have low pretest probability of disease, and in situations where endoscopy might be considered unsafe.
 Helicobacter pylori testing
Helicobacter pylori testing
 Gastric emptying studies: Gastric scintigraphy and gastroduodenal manometry studies generally do not influence medical management and are reserved for patients with normal laboratory tests and a normal EGD, yet who continue to have frequent or protracted vomiting suggestive of a motility disorder. Even in these cases, empiric treatment with prokinetic agents should probably be tried first. Disorders of the gallbladder (see Biliary Extrahepatic Obstruction, Complete).
Gastric emptying studies: Gastric scintigraphy and gastroduodenal manometry studies generally do not influence medical management and are reserved for patients with normal laboratory tests and a normal EGD, yet who continue to have frequent or protracted vomiting suggestive of a motility disorder. Even in these cases, empiric treatment with prokinetic agents should probably be tried first. Disorders of the gallbladder (see Biliary Extrahepatic Obstruction, Complete).
TABLE 5–2. Differential Diagnosis of Dyspepsia
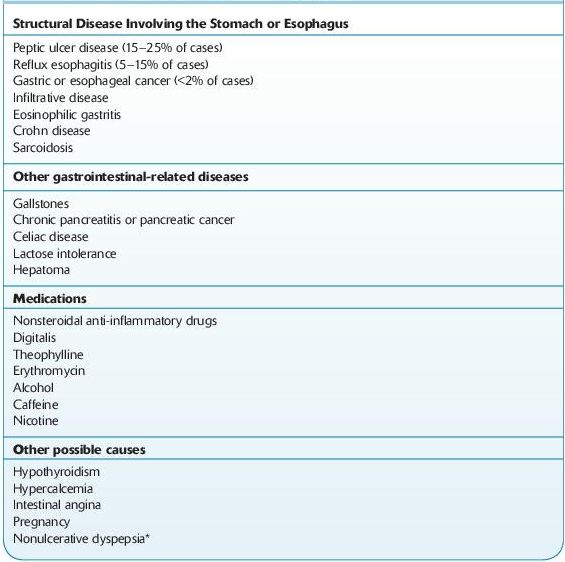
*Nonulcerative dyspepsia occurs in up to 60% of cases, but the diagnosis requires the exclusion of other diagnostic entities.
Suggested Readings
Dominguez-Munoz JE, Malfertheiner P. Optimized serum pancreolauryl test for differentiating patients with and without chronic pancreatitis. Clin Chem. 1998;44:869.
Ferry GD. Causes of acute abdominal pain in children. www.uptodate.com, May 2009.
Khan F, Sachs H, Pechet L, et al. Guide to Diagnostic Testing. Philadelphia, PA: Lippincott Williams & Wilkins; 2002.
Penner RM, Majumdar SR. Diagnostic approach to abdominal pain in adults. www.uptodate.com, May 2009.
 ASCITES
ASCITES
 Definition
Definition
 Ascites is a collection of free fluid in the peritoneal cavity.
Ascites is a collection of free fluid in the peritoneal cavity.
 Etiology
Etiology
 Chronic liver disease (infectious hepatitis and alcoholism) causes 80% of cases of ascites (see Hepatomegaly, Jaundice).
Chronic liver disease (infectious hepatitis and alcoholism) causes 80% of cases of ascites (see Hepatomegaly, Jaundice).
 Multiple causes, including cirrhosis, peritoneal carcinomatosis, or tuberculous peritonitis, account for 3–5% of cases.
Multiple causes, including cirrhosis, peritoneal carcinomatosis, or tuberculous peritonitis, account for 3–5% of cases.
 Carcinomatosis causes <10% of cases of ascites.
Carcinomatosis causes <10% of cases of ascites.
 Heart failure is responsible for <3–5% of cases, and nephritic syndrome is a rare cause of ascites.
Heart failure is responsible for <3–5% of cases, and nephritic syndrome is a rare cause of ascites.
 Cryptogenic cirrhosis may account for up to 10% of cases.
Cryptogenic cirrhosis may account for up to 10% of cases.
 Classification
Classification
 Ascites is currently classified as high gradient or low gradient, depending on the serum ascites albumin gradient (SAAG). Calculation of SAAG involves the difference (not the ratio) between serum values and AF values.
Ascites is currently classified as high gradient or low gradient, depending on the serum ascites albumin gradient (SAAG). Calculation of SAAG involves the difference (not the ratio) between serum values and AF values.
 High-gradient ascites results from portal hypertension, whether on the basis of cirrhosis or noncirrhosis. Nephrotic syndrome is an exception and will usually cause low-gradient ascites due to marked hypoalbuminemia.
High-gradient ascites results from portal hypertension, whether on the basis of cirrhosis or noncirrhosis. Nephrotic syndrome is an exception and will usually cause low-gradient ascites due to marked hypoalbuminemia.
 Low-gradient ascites usually occurs as the result of cardiac failure, malignant carcinomatosis of the peritoneum, infections (such as TB), perforation of the bowel, connective tissues diseases, SLE, and chemical inflammation as in pancreatitis.
Low-gradient ascites usually occurs as the result of cardiac failure, malignant carcinomatosis of the peritoneum, infections (such as TB), perforation of the bowel, connective tissues diseases, SLE, and chemical inflammation as in pancreatitis.
 Laboratory Findings (Figure 5-2)
Laboratory Findings (Figure 5-2)
 Culture: Bedside inoculation of AF in blood culture bottles has increased the positive bacterial yield to interpreted in concert with the cell count. A Gram stain should also be done.
Culture: Bedside inoculation of AF in blood culture bottles has increased the positive bacterial yield to interpreted in concert with the cell count. A Gram stain should also be done.
Imaging studies: Ultrasonography is useful for detecting the presence of ascites as well as for determining the etiology. It may reveal evidence of chronic liver disease, malignancy, hepatomegaly, and pancreatic disorder.
Ascites fluid findings: AF examination is the principle diagnostic tool. Using abdominal paracentesis to obtain and study the fluid is crucial to making a diagnosis.
 Transparent to pale fluid: Is seen in cases of portal hypertension. Neutrophilia in excess of 1,000/mL results in opalescence. A concentration of RBCs in excess of 10,000/mL gives a faint pink tinge, and cell counts >20,000/mL color it red. A traumatic tap is evident by a streak of blood rather than homogeneously red fluid and the tendency to clot. Hepatocellular carcinoma and, rarely, metastatic disease; can cause a bloody tap. TB is only a rare cause of hemorrhagic ascites.
Transparent to pale fluid: Is seen in cases of portal hypertension. Neutrophilia in excess of 1,000/mL results in opalescence. A concentration of RBCs in excess of 10,000/mL gives a faint pink tinge, and cell counts >20,000/mL color it red. A traumatic tap is evident by a streak of blood rather than homogeneously red fluid and the tendency to clot. Hepatocellular carcinoma and, rarely, metastatic disease; can cause a bloody tap. TB is only a rare cause of hemorrhagic ascites.
 Chylous or milky ascites: Has a higher triglyceride concentration than serum and >200 mg/dL. It is rarely seen and is usually an indication of cirrhosis rather than lymphoma or TB as was previously thought. The triglycerides are >1,000 mg/dL in truly milky ascites. Dark-brown ascites may be seen in significant hyperbilirubinemia, biliary perforation (when ascitic bilirubin is higher than serum bilirubin), pancreatitis, and, rarely, in malignant melanoma.
Chylous or milky ascites: Has a higher triglyceride concentration than serum and >200 mg/dL. It is rarely seen and is usually an indication of cirrhosis rather than lymphoma or TB as was previously thought. The triglycerides are >1,000 mg/dL in truly milky ascites. Dark-brown ascites may be seen in significant hyperbilirubinemia, biliary perforation (when ascitic bilirubin is higher than serum bilirubin), pancreatitis, and, rarely, in malignant melanoma.
 Bloody ascites fluid: Once a traumatic tap has been ruled out, 50% of cases are due to hepatocellular carcinoma. TB rarely causes bloody fluid.
Bloody ascites fluid: Once a traumatic tap has been ruled out, 50% of cases are due to hepatocellular carcinoma. TB rarely causes bloody fluid.
 Staining: Gram staining has low yield. Even with centrifugation, it has 10% sensitivity in spontaneous bacterial peritonitis. AFB smear for TB have very low sensitivity. In an appropriate clinical setting of low-grade fever, malaise, and weight loss, a high cell count with lymphocytic predominance and low SAAG is suggestive of TB ascites.
Staining: Gram staining has low yield. Even with centrifugation, it has 10% sensitivity in spontaneous bacterial peritonitis. AFB smear for TB have very low sensitivity. In an appropriate clinical setting of low-grade fever, malaise, and weight loss, a high cell count with lymphocytic predominance and low SAAG is suggestive of TB ascites.
 Protein concentration of AF categorized ascites into exudative (ascitic protein >2.5 g/dL) or transudative (ascitic protein <2.5 g/dL). The significance of this has never been evaluated adequately and objectively.
Protein concentration of AF categorized ascites into exudative (ascitic protein >2.5 g/dL) or transudative (ascitic protein <2.5 g/dL). The significance of this has never been evaluated adequately and objectively.
 Cell count and differential: In uncomplicated cirrhosis, the total WBC count is <500 cells/μL with <250 neutrophils/μL. After diuresis, the total cell count may go up, but the neutrophil count remains below 250 cells/μL. In spontaneous bacterial peritonitis, the total WBC count and neutrophil count are usually, but not always, raised. In TB and carcinomatosis, the cell count rises but with a predominance of lymphocytes. In traumatic taps, for every 250 RBCs, one neutrophil is subtracted from the total WBC count.
Cell count and differential: In uncomplicated cirrhosis, the total WBC count is <500 cells/μL with <250 neutrophils/μL. After diuresis, the total cell count may go up, but the neutrophil count remains below 250 cells/μL. In spontaneous bacterial peritonitis, the total WBC count and neutrophil count are usually, but not always, raised. In TB and carcinomatosis, the cell count rises but with a predominance of lymphocytes. In traumatic taps, for every 250 RBCs, one neutrophil is subtracted from the total WBC count.
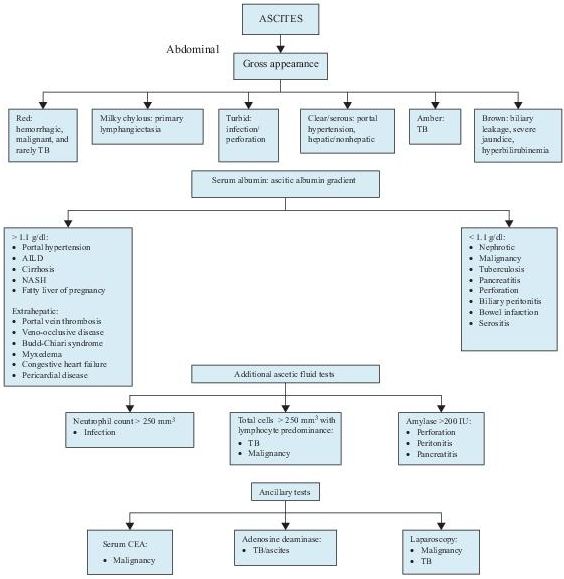
Figure 5–2 Algorithm for the workup of patients with ascites. AILD, alcohol-induced liver disease; CEA, carcinogenic embryonic antigen; NASH, nonalcoholic steatohepatitis; TB, tuberculosis; TNC, total neutrophil count.
Core laboratory: The serum and AF glucose concentrations are nearly the same in uncomplicated portal hypertension (large numbers of WBCs, bacteria, or tumor cells consume glucose and may lead to diminished levels). Amylase values may be about 3–5 times higher than the serum values. LD levels rise because of release of LD from the neutrophils. The rise occurs in cases of secondary peritonitis, TB, and pancreatitis.
Cytology: Has limitations in the diagnosis of malignant ascites and has been replaced largely laparoscopic examination of the peritoneum along with biopsy and culture.
 Limitations
Limitations
 Errors may occur if serum albumin is very low or when serum and ascitic samples are not obtained within a short space of time from each other.
Errors may occur if serum albumin is very low or when serum and ascitic samples are not obtained within a short space of time from each other.
 A high globulin level in serum may also give a false result.
A high globulin level in serum may also give a false result.
DISORDERS OF THE PERITONEUM ASSOCIATED WITH ASCITES
CHRONIC LIVER DISEASE (SEE P. 198)
 This disease differs from ascites caused by malignancy.
This disease differs from ascites caused by malignancy.
 Laboratory Findings
Laboratory Findings
Albumin: Almost always ≥1.1 g/dL in cirrhosis (most common cause), alcoholic hepatitis, massive liver metastases, fulminant hepatic failure, portal vein thrombosis, Budd-Chiari syndrome, cardiac ascites, fatty liver, acute fatty liver of pregnancy, myxedema, mixed (e.g., cirrhosis with peritoneal TB). May be falsely low if serum albumin <1.1 g/dL or the patient in shock. May be falsely high with chylous ascites (lipid interferes with albumin assay). Albumin levels <1.1 g/dL in >90% of cases of peritoneal carcinomatosis (most common cause), TB, pancreatic or biliary ascites, nephrotic syndrome, bowel infarction or obstruction, and serositis in patients without cirrhosis.
Ascites fluid findings: AF total protein >2.5 mg/dL in cancer is only 56% accurate because of high protein content in 12–19% of these ascites as well as changes caused by albumin infusion and diuretic therapies. AF/serum albumin ratio <0.5 in cirrhosis (>90% accuracy). AF/serum ratio of LD (>0.6) or protein (>0.5) is not more accurate (approximately 56%) than only total protein for diagnosis of exudate. AF cholesterol <55 mg/dL in cirrhosis (94% accuracy). Albumin gradient (serum albumin minus AF albumin) reflects portal pressure. Total WBC count is usually <300/μL (50% of cases) and PMN <25% (50% of cases).
Core laboratory: Liver function tests are abnormal.
Other: Cirrhosis findings are similar with or without hepatocellular carcinoma. Cardiac ascites is associated with a blood–AF albumin gradient >1.1 g/dL, but malignant AF shows blood–AF albumin gradient <1.1 g/dL in 93% of cases.
INFECTED ASCITIC FLUID
 Laboratory Findings
Laboratory Findings
Culture: AF in blood culture bottles has 85% sensitivity.
Ascites fluid findings:
 WBC count >250/μL: sensitivity = 85%, specificity = 93%, and neutrophils >50% are presumptive of bacterial peritonitis.
WBC count >250/μL: sensitivity = 85%, specificity = 93%, and neutrophils >50% are presumptive of bacterial peritonitis.
 pH <7.35 and arterial–AF pH difference >0.10; both these findings are virtually diagnostic of bacterial peritonitis and the absence of the above findings virtually excludes bacterial peritonitis.
pH <7.35 and arterial–AF pH difference >0.10; both these findings are virtually diagnostic of bacterial peritonitis and the absence of the above findings virtually excludes bacterial peritonitis.
 Lactate >25 mg/dL and arterial–AF difference >20 mg/dL are often present. LD is markedly increased. Phosphate, potassium, and gamma-glutamyltransferase may also be increased. Glucose is unreliable for diagnosis. Total protein <1.0 g/dL indicates high risk for SBP.
Lactate >25 mg/dL and arterial–AF difference >20 mg/dL are often present. LD is markedly increased. Phosphate, potassium, and gamma-glutamyltransferase may also be increased. Glucose is unreliable for diagnosis. Total protein <1.0 g/dL indicates high risk for SBP.
 Gram stain shows few bacteria in spontaneous bacterial peritonitis (SBP) but many when caused by intestinal perforation. Culture sensitivity = 50% for SBP and approximately 80% for secondary peritonitis. TB acid-fast stain sensitivity = 20–30% and TB culture sensitivity = 50–70%.
Gram stain shows few bacteria in spontaneous bacterial peritonitis (SBP) but many when caused by intestinal perforation. Culture sensitivity = 50% for SBP and approximately 80% for secondary peritonitis. TB acid-fast stain sensitivity = 20–30% and TB culture sensitivity = 50–70%.
SECONDARY PERITONITIS
 This condition shows polymicrobial infection, total protein >1.0 g/dL, AF/LD greater than serum upper limit of normal, and glucose <50 mg/dL compared with spontaneous bacterial peritonitis (SPB).
This condition shows polymicrobial infection, total protein >1.0 g/dL, AF/LD greater than serum upper limit of normal, and glucose <50 mg/dL compared with spontaneous bacterial peritonitis (SPB).
 Prevalence of SBP 15%; due to Escherichia coli approximately 50%, Klebsiella, and other gram-negative bacteria; gram-positive bacteria approximately 25% (especially streptococci).
Prevalence of SBP 15%; due to Escherichia coli approximately 50%, Klebsiella, and other gram-negative bacteria; gram-positive bacteria approximately 25% (especially streptococci).
CONTINUOUS AMBULATORY PERITONEAL DIALYSIS
Monitor dialysate for the following:
 Infection: Peritonitis is defined as WBC count >100/μL, usually with >50% PMNs (normal is <50 WBC/μL, usually mononuclear cells), or positive Gram stain or culture (most prevalent: coagulase-negative staphylococci, Staphylococcus aureus, Streptococcus sp.; multiple organisms, especially mixed aerobes and anaerobes occur with bowel perforation). Successful therapy causes fall in WBC count within first 2 days and a return to <100/μL in 4–5 days; differential returns to predominance of monocytes in 4–7 days with increased eosinophils in 10% of cases. Patients check outflow bags for turbidity. Turbid dialysate can occur occasionally without peritonitis during the first few months of placing catheter (due to catheter hypersensitivity) with WBC count 100–8,000/μL, 10–95% eosinophils, sometimes increased PMNs, and negative cultures. Occasional RBCs may be seen during menstruation or with ovulation at midcycle. Because of low WBC decision level, manual hemocytometer count rather than an automated instrument must be used.
Infection: Peritonitis is defined as WBC count >100/μL, usually with >50% PMNs (normal is <50 WBC/μL, usually mononuclear cells), or positive Gram stain or culture (most prevalent: coagulase-negative staphylococci, Staphylococcus aureus, Streptococcus sp.; multiple organisms, especially mixed aerobes and anaerobes occur with bowel perforation). Successful therapy causes fall in WBC count within first 2 days and a return to <100/μL in 4–5 days; differential returns to predominance of monocytes in 4–7 days with increased eosinophils in 10% of cases. Patients check outflow bags for turbidity. Turbid dialysate can occur occasionally without peritonitis during the first few months of placing catheter (due to catheter hypersensitivity) with WBC count 100–8,000/μL, 10–95% eosinophils, sometimes increased PMNs, and negative cultures. Occasional RBCs may be seen during menstruation or with ovulation at midcycle. Because of low WBC decision level, manual hemocytometer count rather than an automated instrument must be used.
 Metabolic change: Assay dialysate for creatinine and glucose; calculate ultra-filtrate volume by weighing dialysate fluid after 4-hour dwell time and subtracting it from preinfusion weight using specific gravity of 1.0.
Metabolic change: Assay dialysate for creatinine and glucose; calculate ultra-filtrate volume by weighing dialysate fluid after 4-hour dwell time and subtracting it from preinfusion weight using specific gravity of 1.0.
PANCREATIC DISEASE
 AF amylase level greater than serum amylase level is specific for pancreatic disease, but both levels are normal in 10% of cases.
AF amylase level greater than serum amylase level is specific for pancreatic disease, but both levels are normal in 10% of cases.
 Methemalbumin in serum or AF and total protein >4.5 g/dL indicate poor prognosis.
Methemalbumin in serum or AF and total protein >4.5 g/dL indicate poor prognosis.
MALIGNANT ASCITES
 Increased fluid cholesterol (>45 mg/dL) and fibronectin (>10 mg/dL) have S/S 90%/82%.
Increased fluid cholesterol (>45 mg/dL) and fibronectin (>10 mg/dL) have S/S 90%/82%.
 Positive cytology has S/S 70%/100%.
Positive cytology has S/S 70%/100%.
 Increased AF CEA (>2.5 mg/dL) has S/S 45%/100%.
Increased AF CEA (>2.5 mg/dL) has S/S 45%/100%.
ASCITES IN FETUS OR NEONATE
 Causes
Causes
 Nonimmune (occurs in 1 in 3,000 pregnancies)
Nonimmune (occurs in 1 in 3,000 pregnancies)
 Cardiovascular abnormalities causing CHF (e.g., structural, arrhythmias) (40% of cases)
Cardiovascular abnormalities causing CHF (e.g., structural, arrhythmias) (40% of cases)
 Chromosomal (e.g., Turner and Down syndromes are most common; trisomy 13, 15, 16, and 18) (10–15% of cases)
Chromosomal (e.g., Turner and Down syndromes are most common; trisomy 13, 15, 16, and 18) (10–15% of cases)
 Hematologic disorders (any severe anemia) (10% of cases)
Hematologic disorders (any severe anemia) (10% of cases)
 Inherited (e.g., α-thalassemia, hemoglobinopathies, G6PD deficiency)
Inherited (e.g., α-thalassemia, hemoglobinopathies, G6PD deficiency)
 Acquired (e.g., fetal–maternal hemorrhage, twin-to-twin transfusion, congenital infection [parvovirus B19], methemoglobinemia)
Acquired (e.g., fetal–maternal hemorrhage, twin-to-twin transfusion, congenital infection [parvovirus B19], methemoglobinemia)
 Congenital defects of the chest and abdomen.
Congenital defects of the chest and abdomen.
 Structural (e.g., diaphragmatic hernia, jejunal atresia, volvulus, intestinal malrotation) Peritonitis caused by GI tract perforation, congenital infection (e.g., syphilis, TORCH [toxoplasmosis, other agents, rubella, CMV, and herpes simplex], hepatitis), meconium peritonitis
Structural (e.g., diaphragmatic hernia, jejunal atresia, volvulus, intestinal malrotation) Peritonitis caused by GI tract perforation, congenital infection (e.g., syphilis, TORCH [toxoplasmosis, other agents, rubella, CMV, and herpes simplex], hepatitis), meconium peritonitis
 Lymphatic duct obstruction
Lymphatic duct obstruction
 Biliary atresia
Biliary atresia
 Nonstructural (e.g., congenital nephrotic syndrome, cirrhosis, cholestasis, hepatic necrosis, GI tract obstruction)
Nonstructural (e.g., congenital nephrotic syndrome, cirrhosis, cholestasis, hepatic necrosis, GI tract obstruction)
 Lower GU tract obstruction (e.g., posterior urethral valves, urethral atresia, and ureterocele) is most common cause
Lower GU tract obstruction (e.g., posterior urethral valves, urethral atresia, and ureterocele) is most common cause
 Inherited skeletal dysplasias (enlarged liver causing extramedullary hematopoiesis)
Inherited skeletal dysplasias (enlarged liver causing extramedullary hematopoiesis)
 Fetal tumors, most often teratomas and neuroblastomas
Fetal tumors, most often teratomas and neuroblastomas
 Vascular placental abnormalities
Vascular placental abnormalities
 Genetic metabolic disorders (e.g., Hurler syndrome, Gaucher disease, Niemann-Pick disease, GM1 gangliosidosis type I, I-cell disease, β-glucuronidase deficiency)
Genetic metabolic disorders (e.g., Hurler syndrome, Gaucher disease, Niemann-Pick disease, GM1 gangliosidosis type I, I-cell disease, β-glucuronidase deficiency)
 Immune (maternal antibodies reacting to fetal antigens [e.g., Rh, C, E, Kell])
Immune (maternal antibodies reacting to fetal antigens [e.g., Rh, C, E, Kell])
PERITONITIS, ACUTE
 See Figures 5-3 and 5-4.
See Figures 5-3 and 5-4.
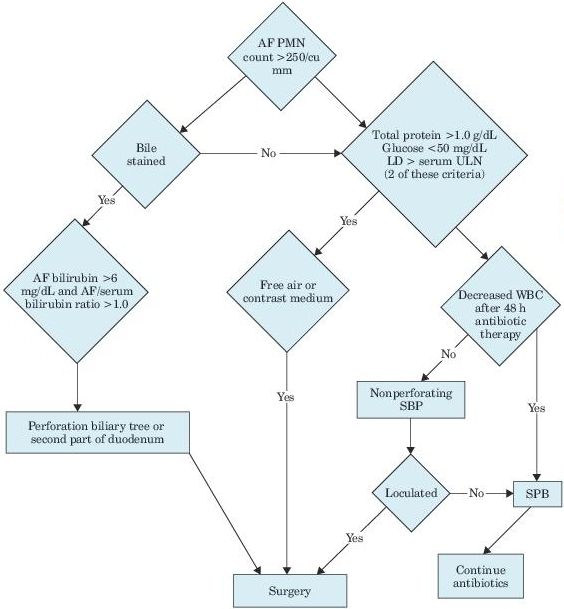
Figure 5–3 Algorithm for differentiating secondary from spontaneous bacterial peritonitis. AF, ascitic fluid; PMN, polymorphonuclear leukocytes; LD, lactate dehydrogenase; ULN, upper limit of normal; WBC, white blood cell; SBP, spontaneous bacterial peritonitis.
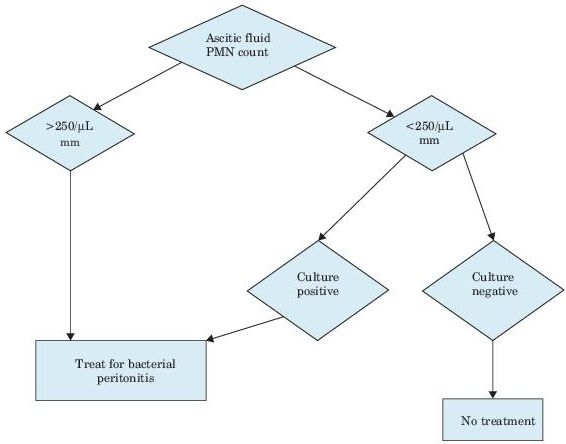
Figure 5–4 Algorithm for spontaneous bacterial peritonitis. PMN, polymorphonuclear leukocytes.
PRIMARY PERITONITIS
Ascites fluid findings: Gram stain of direct smear and culture of peritoneal fluid usually shows streptococci in children. In adults, it is caused by E. coli (40–60%) or S. pneumoniae (15%), other gram-negative bacilli, and enterococci; usually one organism. May be caused by Mycobacterium tuberculosis. Marked increase in WBC (≤50,000/μL) and PMN (80–90%).
Peritoneal lavage fluid findings: Shows WBC count >200/μL in 99% of cases. Other: Laboratory findings due to nephrotic syndrome and post–necrotic cirrhosis and occasionally bacteremia in children and cirrhosis with ascites in adults.
SECONDARY PERITONITIS
Occurs and recurs very frequently in continuous ambulatory peritoneal dialysis.
Laboratory findings due perforation of hollow viscus (e.g., appendicitis, perforated ulcer).
Dialysate findings: Turbid (indicates >300 WBC/μL); Gram stain, culture, and leukocytosis may be absent. Caused by gram-positive bacteria in approximately 70%, enteric gram-negative bacilli and P. aeruginosa in 20–30%, others in 10–20%, and sterile in 10–20%. If more than one pathogen is found, rule out perforated viscus. Usually more than one organism is found.
Suggested Readings
Cárdenas A, Gelrud A, Chopra S. Chylous, bloody, and pancreatic ascites. www.uptodate.com, May 2009.
Khan F, Sachs H, Pechet L, et al. Guide to Diagnostic Testing. Philadelphia, PA: Lippincott Williams & Wilkins; 2002.
Runyon B. Diagnosis and evaluation of patients with ascites. www.uptodate.com, May 2009.
Runyon B. Diagnosis of spontaneous bacterial peritonitis. www.uptodate.com, May 2009.
 DIARRHEA
DIARRHEA
 Definition
Definition
 Diarrhea is defined as >200 g of stool or an increase in the frequency or fluidity of normal stools. It may be acute or chronic, and it is considered chronic when it lasts at least 4 weeks.
Diarrhea is defined as >200 g of stool or an increase in the frequency or fluidity of normal stools. It may be acute or chronic, and it is considered chronic when it lasts at least 4 weeks.
 Etiology
Etiology
Diarrhea can result from any of the following mechanisms.
1.Osmosis: Molecules not normally present in the intestinal lumen increase the osmolality of chime, drawing water into the lumen (i.e., lactose)
2.Secretion: Substances can cause intestinal cells to secrete sodium and water (i.e., cholera toxin).
3.Inflammation results in denuding of the intestinal lining, which in turn disrupts normal absorption, thereby allowing compounds from the lining to leak into the lumen resulting in an increased osmosis.
4.Motility: Hypermotility leads to an increased stool volume. Hypomotility can lead to bacterial overgrowth, which causes diarrhea through several different mechanisms.
5.Anal sphincter dysfunction causes fecal incontinence, which can be interpreted by the patient as diarrhea.
 Differential Diagnosis
Differential Diagnosis
1.Laxative abuse accounts for approximately 15% of all chronic causes. It should be suspected in patients with a mental health disorder.
2.Sorbitol can cause diarrhea. In one study, approximately 17% of people had diarrhea following the ingestion of 4–5 minutes containing sorbitol.
3.Both bile salts and fatty acids cause secretion of chloride followed by water into the colon. Excess bile salts also lead to a mild degree of fat malabsorption.
4.Bacterial overgrowth can occur secondary to diabetes, blind loop syndrome, amyloidosis, diverticulitis, and scleroderma, among other causes.
5.Irritable bowel syndrome classically presents with diarrhea alternating with constipation, but it can also occur in a diarrhea-predominant form.
6.Gastric surgery syndrome results in a decreased contact time with the luminal surface and decreased digestive juices mixing with the chyme.
7.Hyperthyroidism usually has increased frequency and amount of diarrhea but not fluidity. Diarrhea is present in approximately 25% of hyperthyroid cases.
8.Inflammatory bowel disease (IBD):
 Ulcerative colitis is a relapsing and remitting disease that leads to acute inflammation of the colorectal mucosa. The rectum is involved in 55% of cases. In severe cases, bloody diarrhea often leads to weight loss, anemia, and electrolyte imbalance.
Ulcerative colitis is a relapsing and remitting disease that leads to acute inflammation of the colorectal mucosa. The rectum is involved in 55% of cases. In severe cases, bloody diarrhea often leads to weight loss, anemia, and electrolyte imbalance.
 Crohn disease is a chronic relapsing disorder characterized by transmural, asymmetric, and segmental inflammation. It typically involves the ileum, colon, or perianal region; right lower quadrant pain associated with bloody diarrhea is present in 80% of patients.
Crohn disease is a chronic relapsing disorder characterized by transmural, asymmetric, and segmental inflammation. It typically involves the ileum, colon, or perianal region; right lower quadrant pain associated with bloody diarrhea is present in 80% of patients.
9. Neoplasia:
 Villous adenoma produces prostaglandins, which stimulate chloride and water secretion from the colon.
Villous adenoma produces prostaglandins, which stimulate chloride and water secretion from the colon.
 Serotonin from carcinoid cells stimulates gut motility and increases intestinal secretion.
Serotonin from carcinoid cells stimulates gut motility and increases intestinal secretion.
 Tumor-associated calcitonin stimulates gut motility.
Tumor-associated calcitonin stimulates gut motility.
 Gastrinoma leads to increased gastric acid, which directly causes fluid secretion.
Gastrinoma leads to increased gastric acid, which directly causes fluid secretion.
10. Infection:
 Refer to p. 624, Foodborne Infectious Illnesses, and see other sections on specific agents that cause diarrheal disease.
Refer to p. 624, Foodborne Infectious Illnesses, and see other sections on specific agents that cause diarrheal disease.
 Laboratory Findings
Laboratory Findings
Endoscopy: Lower endoscopy may help. One series has a 20% yield in identifying a pathologic diagnosis. In non–HIV-infected patients, the role of sigmoidoscopy versus colonoscopy is unclear. When clinically suspected, even if no gross abnormalities are noted, consider doing blind biopsies looking for lymphocytic and collagenous colitis. The yield of biopsy with no gross abnormalities ranges from 6% to 42%. Upper endoscopy is useful for making the diagnosis of sprue, Whipple disease, and other small bowel infiltrative processes.
Radiology: An upper GI series with small bowel follow-through is most commonly used when evaluating for Crohn disease. Enteroclysis is superior, with 100% sensitivity and 98% specificity for small bowel involvement with Crohn disease.
Recommended laboratory tests of stool:
 Fecal leukocytes.
Fecal leukocytes.
 Stool for osmolality gap: The osmolality gap is calculated by the following formula: 2 (stool Na + K). The accuracy is fair in distinguishing between osmotic (if gap is 50) and secretory (if gap is >50) diarrhea.
Stool for osmolality gap: The osmolality gap is calculated by the following formula: 2 (stool Na + K). The accuracy is fair in distinguishing between osmotic (if gap is 50) and secretory (if gap is >50) diarrhea.
 Stool for pH: For carbohydrate intolerance (e.g., lactose or sorbitol), one small study found the pH <5.6. For bile acid–induced diarrhea, the pH is usually over 6.8.
Stool for pH: For carbohydrate intolerance (e.g., lactose or sorbitol), one small study found the pH <5.6. For bile acid–induced diarrhea, the pH is usually over 6.8.
 Stool for fecal fat: This test is used to detect steatorrhea on the basis of malabsorption.
Stool for fecal fat: This test is used to detect steatorrhea on the basis of malabsorption.
 Qualitative: Sensitivity is 97–100%, but the specificity varies from 56% to 86%.
Qualitative: Sensitivity is 97–100%, but the specificity varies from 56% to 86%.
 Quantitative: Based upon a 72-hour collection, the patient should be on a 75- to 100-g fat diet. A nutritional consult is advised to maximize compliance.
Quantitative: Based upon a 72-hour collection, the patient should be on a 75- to 100-g fat diet. A nutritional consult is advised to maximize compliance.
 Test(s) for infectious agents (e.g., stool culture, O&P examination, rotavirus detection) based on clinical presentation.
Test(s) for infectious agents (e.g., stool culture, O&P examination, rotavirus detection) based on clinical presentation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


