Role of the Thymus in T-Cell Maturation & Selection
MUCOSA-ASSOCIATED LYMPHOID TISSUE
Role of Lymph Nodes in the Immune response
Functions of Splenic White & red Pulp
The immune system provides defense or immunity against infectious agents ranging from viruses to multicellular parasites. Histologically this system consists of a large, diverse population of leukocytes located within every tissue of the body and lymphoid organs interconnected only by the blood and lymphatic circulation. Immunity obviously has tremendous medical importance, one part of which focuses on autoimmune diseases in which immune cells begin to function abnormally and attack molecular components of the body’s own organs.
Immunologists recognize two partially overlapping lines of defense against invaders and/or other abnormal, potentially harmful cells: innate immunity and adaptive immunity. The first of these is nonspecific, involves a wide variety of effector mechanisms, and is evolutionarily older than the second type. Among the cells mediating innate immunity are most of the granulocytes and other leukocytes described in Chapters 12 and 13. Conversely, adaptive immunity aims at specific microbial invaders, is mediated by lymphocytes and antigen-presenting cells (APCs) discussed in this chapter, and produces memory cells that permit a similar, very rapid response if that specific microbe appears again.
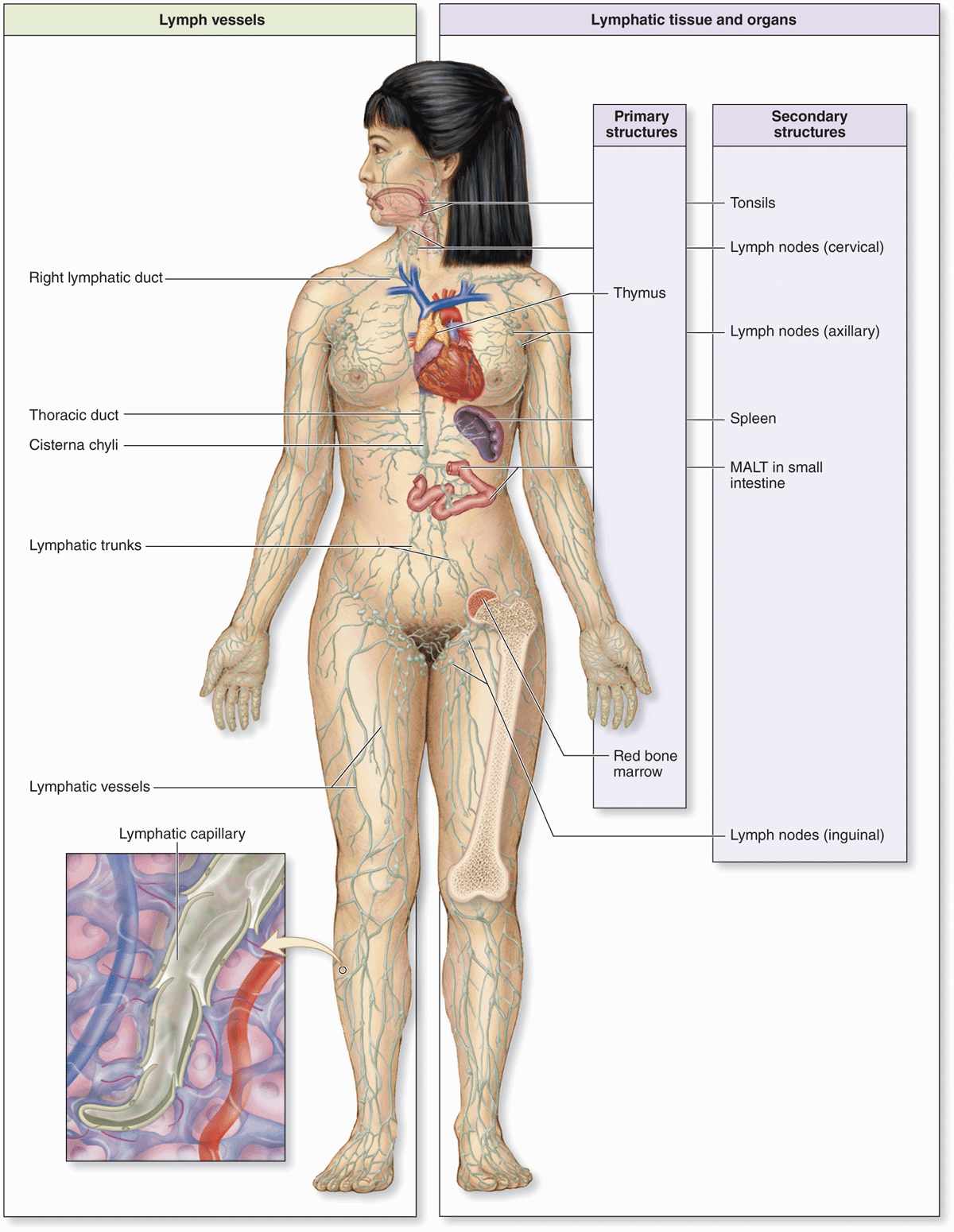
The lymphocytes and APCs for adaptive immunity are distributed throughout the body in the blood, lymph, and epithelial and connective tissues. Lymphocytes are formed initially in primary lymphoid organs (the thymus and bone marrow), but most lymphocyte activation and proliferation occur in secondary lymphoid organs (the lymph nodes, the spleen, and diffuse lymphoid tissue found in the mucosa of the digestive system, including the tonsils, Peyer patches, and appendix). The immune cells located diffusely in the digestive, respiratory, or urogenital mucosae comprise what is collectively known as mucosa-associated lymphoid tissue (MALT). Proliferating B lymphocytes in the secondary structures of MALT are arranged in small spherical lymphoid nodules. The wide distribution of immune system cells and the constant traffic of lymphocytes through the blood, lymph, connective tissues, and secondary lymphoid structures provide the body with an elaborate and efficient system of surveillance and defense (Figure 14–1).
FIGURE 14–1 The lymphoid organs and main paths of lymphatic vessels.
INNATE & ADAPTIVE IMMUNITY
The system of defenses termed innate immunity involves immediate, nonspecific actions, including physical barriers such as the skin and mucous membranes of the gastrointestinal, respiratory, and urogenital tracts that prevent infections or penetration of the host body. Bacteria, fungi, and parasites that manage to penetrate these barriers are quickly removed by neutrophils and other leukocytes in the adjacent connective tissue. Toll-like receptors (TLRs) on leukocytes allow the recognition and binding of surface components of such invaders. Other leukocytes orchestrate the defenses at sites of penetration. Natural killer (NK) cells destroy various unhealthy host cells, including those infected with virus or bacteria, as well as certain potentially tumorigenic cells.
Leukocytes and specific cells of the tissue barriers also produce a wide variety of antimicrobial chemicals that also form a major part of innate immunity, including the following:
 Hydrochloric acid (HCl) and organic acids in specific regions lower the pH locally to either kill entering microorganisms directly or inhibit their growth.
Hydrochloric acid (HCl) and organic acids in specific regions lower the pH locally to either kill entering microorganisms directly or inhibit their growth.
 Defensins, short cationic polypeptides produced by neutrophils and various epithelial cells that kill bacteria by disrupting the cell walls.
Defensins, short cationic polypeptides produced by neutrophils and various epithelial cells that kill bacteria by disrupting the cell walls.
 Lysozyme, an enzyme made by neutrophils and cells of epithelial barriers, which hydrolyzes bacterial cell wall components, killing those cells.
Lysozyme, an enzyme made by neutrophils and cells of epithelial barriers, which hydrolyzes bacterial cell wall components, killing those cells.
 Complement, a system of proteins in blood plasma, mucus, and macrophages that react with bacterial surface components to aid removal of bacteria.
Complement, a system of proteins in blood plasma, mucus, and macrophages that react with bacterial surface components to aid removal of bacteria.
 Interferons, paracrine factors from leukocytes and virus-infected cells that signal NK cells to kill such cells and adjacent cells to resist viral infection.
Interferons, paracrine factors from leukocytes and virus-infected cells that signal NK cells to kill such cells and adjacent cells to resist viral infection.
Adaptive immunity, acquired gradually by exposure to microorganisms, is more specific, slower to respond, and an evolutionarily more recent development than innate immunity. The adaptive immune response involves B and T lymphocytes, whose origins are described in this chapter, which become activated against specific invaders by being presented with specific molecules from those cells by APCs, which are usually derived from monocytes. Unlike innate immunity, adaptive immune responses are aimed at specific microbial invaders and involve production of memory lymphocytes so that a similar response can be mounted very rapidly if that invader ever appears again.
CYTOKINES
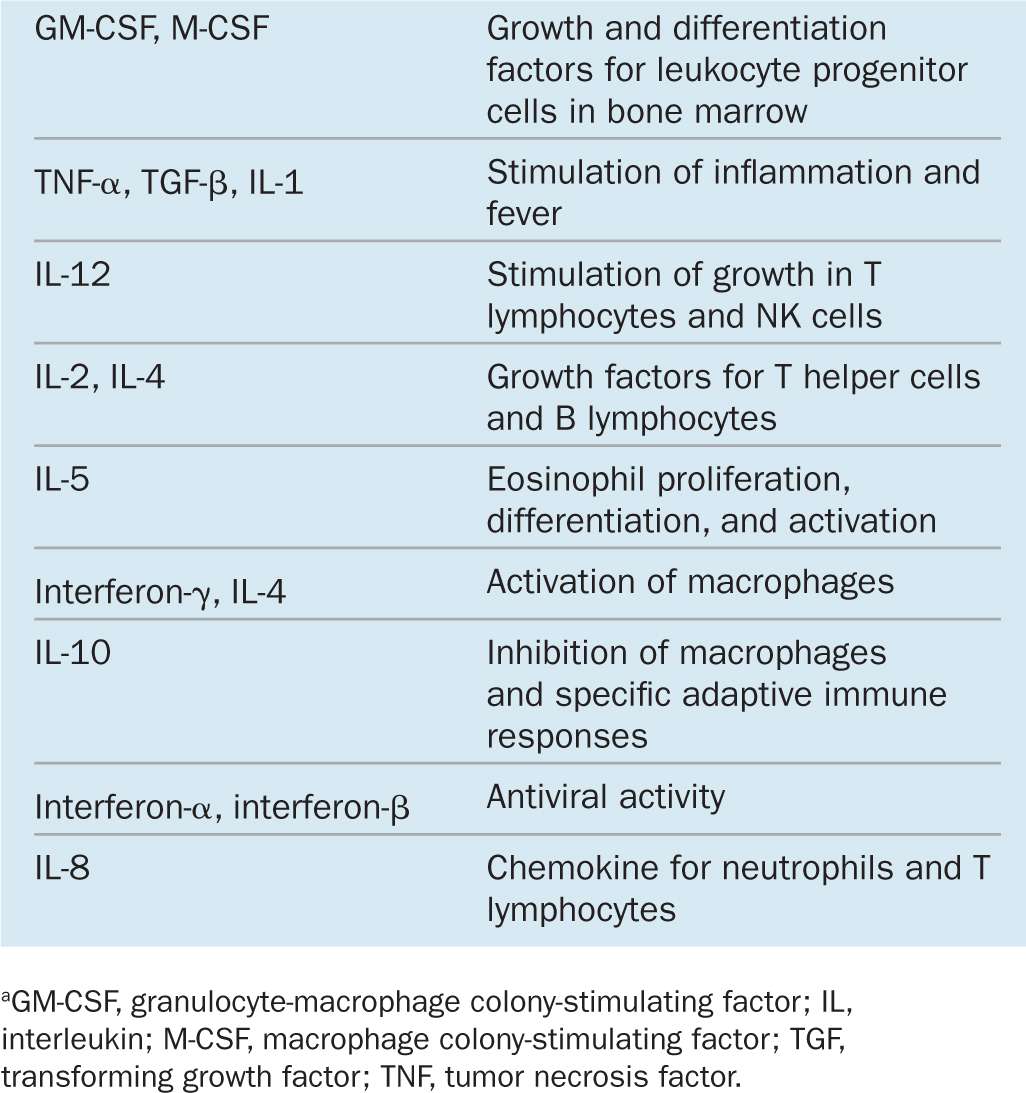
Within lymphoid organs and during inflammation at sites of infection or tissue injury cells in the immune system communicate with each other primarily via cytokines to coordinate defensive measures. Involved in both innate and adaptive immunity, cytokines are a diverse group of peptides and glycoproteins, usually with low molecular masses (between 8 and 80 kDa) and a paracrine mode of action. They coordinate cell activities in the innate and adaptive immune responses. Examples of several important cytokines are given in Table 14–1. Major responses induced in target cells by such factors are the following:
TABLE 14–1 Examples of cytokines, grouped by their main function.
 Directed cell movements, or chemotaxis, toward and cell accumulation at sites of inflammation, for example, during diapedesis. Cytokines producing this effect are also called chemokines.
Directed cell movements, or chemotaxis, toward and cell accumulation at sites of inflammation, for example, during diapedesis. Cytokines producing this effect are also called chemokines.
 Increased mitotic activity in certain leukocytes, both locally and in the bone marrow.
Increased mitotic activity in certain leukocytes, both locally and in the bone marrow.
 Stimulation or suppression of lymphocyte activities in adaptive immunity. A group of cytokines with such effects were named interleukins because they were thought to be produced by and to target only leukocytes.
Stimulation or suppression of lymphocyte activities in adaptive immunity. A group of cytokines with such effects were named interleukins because they were thought to be produced by and to target only leukocytes.
 Stimulated phagocytosis or directed cell killing by innate immune cells.
Stimulated phagocytosis or directed cell killing by innate immune cells.
Most cytokines have multiple target cells in which they exert several effects. Some are produced by and target cells besides immune cells, including endothelial cells, certain autonomic neurons, and cells of the endocrine system. The broad range of cytokine actions greatly extends the physiologic effects of infections and other stressors.
ANTIGENS & ANTIBODIES
A molecule that is recognized by cells of the adaptive immune system is called an antigen and typically elicits a response from these cells. Antigens may consist of soluble molecules (such as proteins or polysaccharides) or molecules that are still components of intact cells (bacteria, protozoa, or tumor cells). Immune cells recognize and react to small molecular domains of the antigen known as antigenic determinants or epitopes. The immune response to antigens may be cellular (in which lymphocytes are primarily in charge of eliminating the antigen), humoral (in which antibodies are primarily responsible for the response), or both.
An antibody is a glycoprotein of the immunoglobulin family that interacts specifically with an antigenic determinant. Antibodies are secreted by plasma cells that arise by terminal differentiation of clonally proliferating B lymphocytes whose receptors recognize and bind specific epitopes. Antibodies either accumulate in the blood plasma and interstitial fluid of tissues or are transported across epithelia into the secretion of glands such as mucous, salivary, and mammary glands. Other antibodies are membrane proteins on the surface of B lymphocytes or other leukocytes. In all these situations each antibody combines with the epitope that it specifically recognizes.
Immunoglobulins of all antibody molecules have a common design, consisting of two identical light chains and two identical heavy chains bound by disulfide bonds (Figure 14–2). The isolated carboxyl-terminal portion of the heavy-chain molecules is called the constant Fc region. The Fc regions of some immunoglobulins are recognized by cell surface receptors on basophils and mast cells, localizing these antibodies to the surface of these cells. The first 110 amino acids near the amino-terminal ends of the light and heavy chains vary widely among different antibody molecules, and this region is called the variable region. The variable portions of one heavy and one light chain make up an antibody’s antigen-binding site. DNA sequences coding for these regions undergo recombination and rearrangement after B lymphocytes are activated against a specific antigen and the progeny of those cells all produce antibodies that specifically bind that antigen. Each antibody has two antigen-binding sites, both for the same antigen.
FIGURE 14–2 Basic structure of an immunoglobulin (antibody).
Classes of Antibodies
Immunoglobulins of humans fall into five major classes, listed in Table 14–2 with their structural features, abundance in plasma, major locations, and functions. The classes are called immunoglobulin G (IgG), IgA, IgM, IgE, and IgD, and key aspects for each include the following:
TABLE 14–2 Important features of the antibody classes in humans.
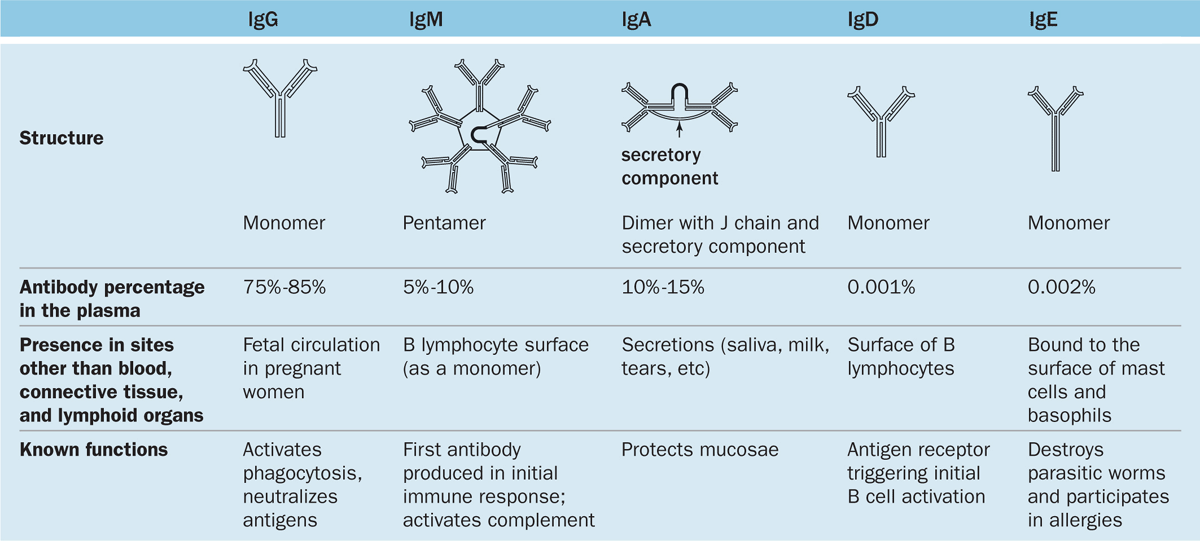
 IgG is the most abundant class representing 75% to 85% of the immunoglobulin in blood. Production increases during immune responses following infections, etc. Unlike the other classes of antibodies, IgG is highly soluble, stable (half-life >3 weeks), and crosses the placental barrier into the fetal circulation. This confers passive immunity against certain infections until the newborn’s own adaptive immune system is acquired.
IgG is the most abundant class representing 75% to 85% of the immunoglobulin in blood. Production increases during immune responses following infections, etc. Unlike the other classes of antibodies, IgG is highly soluble, stable (half-life >3 weeks), and crosses the placental barrier into the fetal circulation. This confers passive immunity against certain infections until the newborn’s own adaptive immune system is acquired.
 IgA is present in almost all exocrine secretions as a dimeric form in which the heavy chains of two monomers are united by a polypeptide called the J chain. IgA is produced by plasma cells in mucosae of the digestive, respiratory, and reproductive tracts. Another protein bound to this immunoglobulin, the secretory component, is released by the epithelial cells as IgA undergoes transcytosis. The resulting structure is relatively resistant to proteolysis and reacts with microorganisms in milk, saliva, tears, and mucus coating the mucosae in which it is made.
IgA is present in almost all exocrine secretions as a dimeric form in which the heavy chains of two monomers are united by a polypeptide called the J chain. IgA is produced by plasma cells in mucosae of the digestive, respiratory, and reproductive tracts. Another protein bound to this immunoglobulin, the secretory component, is released by the epithelial cells as IgA undergoes transcytosis. The resulting structure is relatively resistant to proteolysis and reacts with microorganisms in milk, saliva, tears, and mucus coating the mucosae in which it is made.
 IgM constitutes 5% to 10% of blood immunoglobulin and usually exits in a pentameric form united by a J chain. IgM is mainly produced in an initial response to an antigen. IgM bound to antigen is the most effective antibody class in activating the complement system.
IgM constitutes 5% to 10% of blood immunoglobulin and usually exits in a pentameric form united by a J chain. IgM is mainly produced in an initial response to an antigen. IgM bound to antigen is the most effective antibody class in activating the complement system.
 IgE, usually a monomer, is much less abundant in the circulation and exists bound at its Fc region to receptors on the surface of mast cells and basophils. When this IgE encounters the antigen that elicited its production, the antigen-antibody complex triggers the liberation of several biologically active substances, such as histamine, heparin, and leukotrienes. This characterizes an allergic reaction, which is thus mediated by the binding of cell-bound IgE with the antigens (allergens) that stimulated the IgE to be synthesized initially (see Mast Cells in Chapter 5).
IgE, usually a monomer, is much less abundant in the circulation and exists bound at its Fc region to receptors on the surface of mast cells and basophils. When this IgE encounters the antigen that elicited its production, the antigen-antibody complex triggers the liberation of several biologically active substances, such as histamine, heparin, and leukotrienes. This characterizes an allergic reaction, which is thus mediated by the binding of cell-bound IgE with the antigens (allergens) that stimulated the IgE to be synthesized initially (see Mast Cells in Chapter 5).
 IgD, the least abundant immunoglobulin in plasma, is also the least understood class of antibody. Monomers of IgD are bound to the surface of B lymphocytes where they (along with IgM monomers) act as antigen receptors in triggering B-cell activation.
IgD, the least abundant immunoglobulin in plasma, is also the least understood class of antibody. Monomers of IgD are bound to the surface of B lymphocytes where they (along with IgM monomers) act as antigen receptors in triggering B-cell activation.
Actions of Antibodies
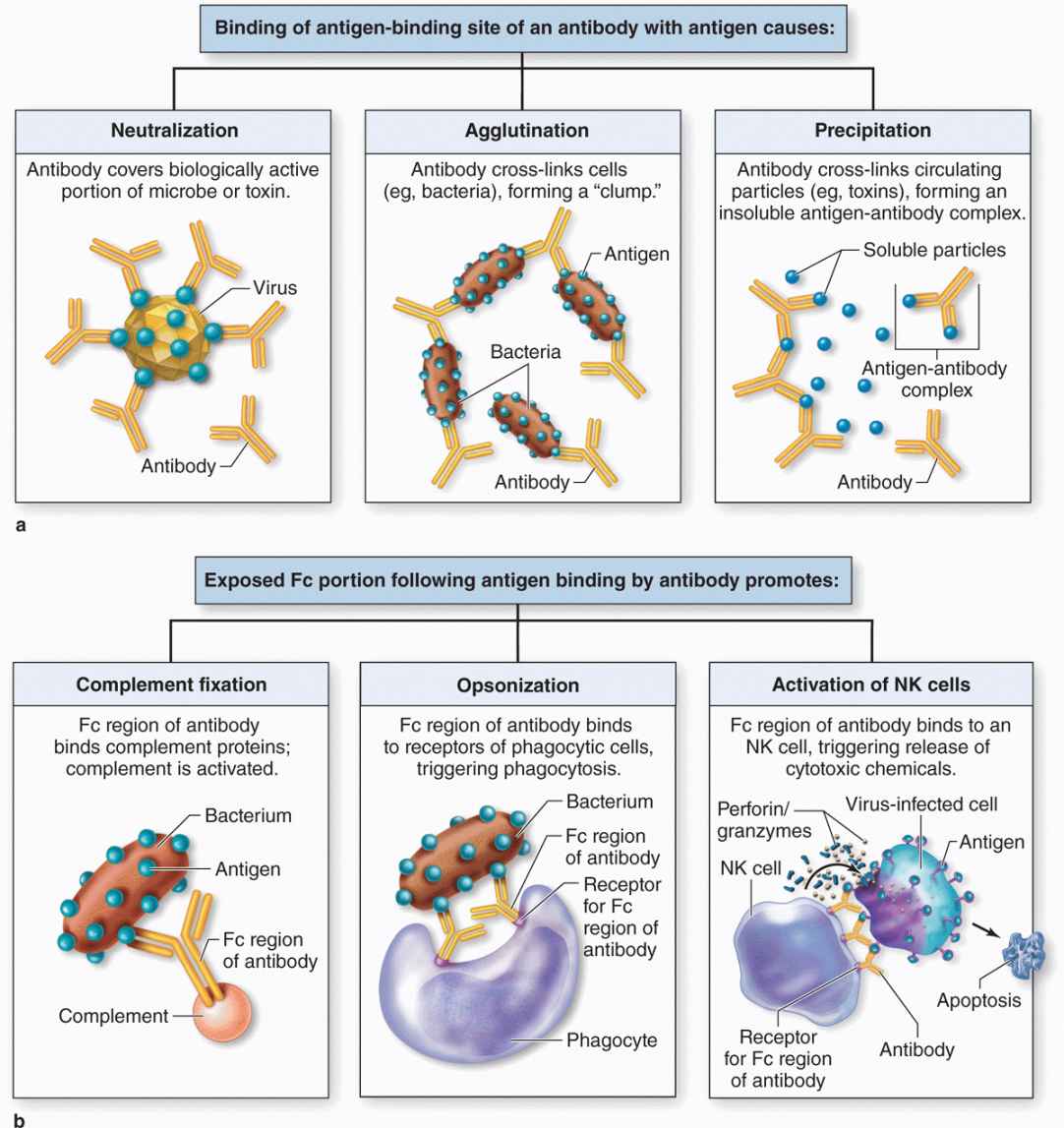
As shown in Figure 14–3a, antigen-binding sites of IgG and IgA antibodies are able to bind specifically and neutralize certain viral particles and bacterial toxins, agglutinate many bacterial cells, and precipitate most soluble antigens. In addition, the Fc portions of these and other antibodies also bind receptors for this sequence and thereby optimize three important actions of innate immunity (Figure 14–3b):
FIGURE 14–3 Various specific and nonspecific functions of antibodies.
 Complement activation: Antigen-antibody complexes containing IgG or IgM bind polypeptides of the complement system, a group of around 20 plasma proteins produced mainly in the liver, and activate them through a cascade of enzymatic reactions. After activation, specific complement components bind and rupture membranes of invading cells, clump antigen-bearing bacteria or cells, and elicit arrival of relevant leukocytes.
Complement activation: Antigen-antibody complexes containing IgG or IgM bind polypeptides of the complement system, a group of around 20 plasma proteins produced mainly in the liver, and activate them through a cascade of enzymatic reactions. After activation, specific complement components bind and rupture membranes of invading cells, clump antigen-bearing bacteria or cells, and elicit arrival of relevant leukocytes.
 Opsonization: This refers to the ability of receptors on macrophages, neutrophils, and eosinophils to recognize and bind the Fc portions of antibodies attached to surface antigens of microorganisms. Opsonization greatly increases the efficiency of phagocytosis by these leukocytes at sites of infection.
Opsonization: This refers to the ability of receptors on macrophages, neutrophils, and eosinophils to recognize and bind the Fc portions of antibodies attached to surface antigens of microorganisms. Opsonization greatly increases the efficiency of phagocytosis by these leukocytes at sites of infection.
 NK cells activation: Antibodies bound to antigens on virus-infected cells of the body are recognized by the primitive lymphocytes called NK cells, which are then activated to kill the infected cell by releasing perforin and various granzymes. These two proteins together enter the infected cell via other receptors and cause apoptosis.
NK cells activation: Antibodies bound to antigens on virus-infected cells of the body are recognized by the primitive lymphocytes called NK cells, which are then activated to kill the infected cell by releasing perforin and various granzymes. These two proteins together enter the infected cell via other receptors and cause apoptosis.
ANTIGEN PRESENTATION
Antigens recognized by lymphocytes are often bound to specialized integral membrane protein complexes on cell surfaces. These abundant antigen-presenting proteins are parts of the major histocompatibility complex (MHC) that includes the two key types called MHC class I and class II. As the name implies, these proteins were first recognized by their roles in the immune rejection of grafted tissue or organs. Proteins of both classes, which on human cells are often called human leukocyte antigens (HLAs), are encoded by genes in large chromosomal loci having very high degrees of allelic variation between different individuals. T lymphocytes are specialized to recognize both classes of MHC proteins and the antigens they present. If the MHCs on cells of a tissue graft are not similar to those that T lymphocytes encountered during their development, the grafted cells will induce a strong immune reaction by T cells of the recipient. To these lymphocytes, the unfamiliar MHC epitopes on the graft’s cells are recognized as markers of potentially tumorigenic, infected, or otherwise abnormal (“non-self”) cells that they must eliminate.
Like all integral membrane protein complexes, MHC molecules are made in the rough ER and Golgi apparatus. Before leaving the ER, MHC class I proteins bind a wide variety of proteasome-derived peptide fragments representing the range of all proteins synthesized in that cell. All nucleated cells produce and expose on their surfaces MHC class I molecules presenting such “self-antigens,” which T cells recognize as a signal to ignore those cells. By this same mechanism, some virally infected cells or cells with proteins altered by gene mutation also have MHC class I proteins displaying peptides that T cells do not recognize as “self,” helping lead to the elimination of such cells.
MHC class II proteins are synthesized and transported to the cell surface similarly but only in cells of the mononuclear phagocyte system and certain other cells under some conditions. Before joining the plasmalemma, the Golgi-derived vesicles with the MHC class II complexes first fuse with endolysosomal vesicles containing antigens ingested by receptor-mediated endocytosis, pinocytosis, or phagocytosis. This allows the class II proteins to bind fragments of whatever proteins the cells had ingested, including those from dead, infected, or abnormal cells and atypical proteins of all kinds. At the surface of these cells, the class II complexes display the peptides from these potentially pathogenic cells, signaling T lymphocytes and activating their responses against sources of these antigens.
CELLS OF ADAPTIVE IMMUNITY
Described in Chapter 12 with blood, lymphocytes and the monocyte-derived cells specialized for antigen presentation to lymphocytes are the major players in adaptive immune responses.
Antigen-Presenting Cells
Most specialized antigen-presenting cells (APCs) are part of the mononuclear phagocyte system, including all types of macrophages and specialized dendritic cells in lymphoid organs. Features common to all APCs are an active endocytotic system and expression of MHC class II molecules for presenting peptides of exogenous antigens. Besides dendritic cells (not to be confused with cells of nervous tissue) and all monocyte-derived cells, “professional” APCs include the epithelial reticular cells of the thymus (discussed below) and B lymphocytes.
During inflammation transient expression of MHC class II is induced by interferon-γ in certain local cells that can be considered “nonprofessional” APCs, including fibroblasts and vascular endothelial cells.
Lymphocytes
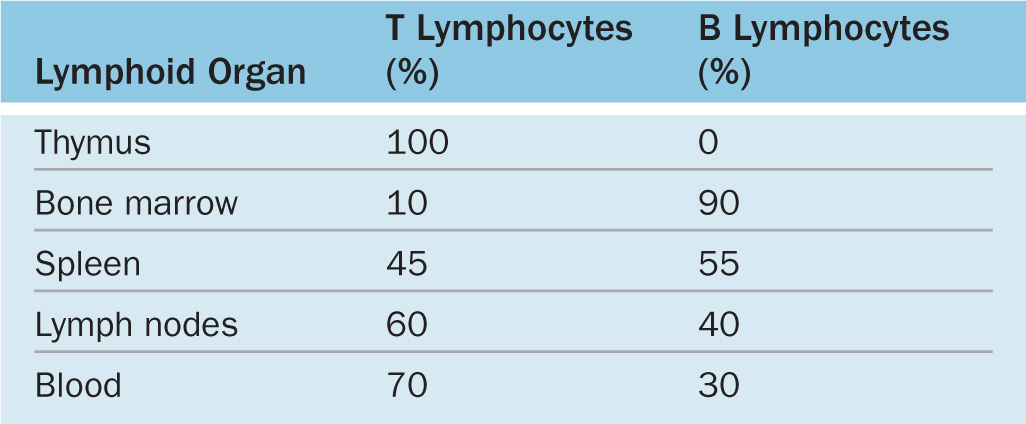
Lymphocytes both regulate and carry out adaptive immunity. In adults stem cells for all lymphocytes are located in the red bone marrow, but cells of the major lymphoid lineages mature and become functional in two different central or primary lymphoid organs. Cells destined to become B lymphocytes remain and differentiate further in the bone marrow. Progenitors of T lymphocytes move via the circulation into the developing thymus. After maturation in these primary structures, B and T cells circulate to the peripheral secondary lymphoid organs, which include the MALT, the lymph nodes, and the spleen (see Figure 14–1). Lymphocytes do not stay long in the lymphoid organs; they continuously recirculate through the body in connective tissues, blood, and lymph. Because of the constant mobility of lymphocytes and APCs, the cellular locations and microscopic details of lymphoid organs differ from one day to the next. However, the relative percentages of T and B lymphocytes in these compartments are relatively steady (Table 14–3).
TABLE 14–3 Approximate percentages of B and T cells in lymphoid organs.
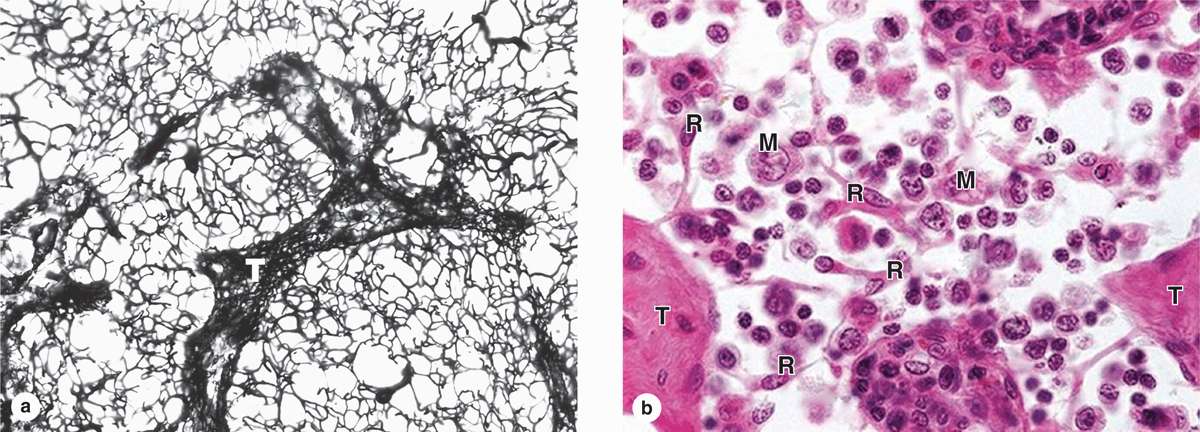
Lymphoid tissue is usually reticular connective tissue filled with large numbers of lymphocytes. It can be either diffuse within areas of loose connective tissue or surrounded by capsules, forming discrete (secondary) lymphoid organs. Because lymphocytes have prominent basophilic nuclei and very little cytoplasm, lymphoid tissue packed with such cells usually stains dark blue in hematoxylin and eosin (H&E)-stained sections. In all secondary lymphoid tissue the lymphocytes are supported by a rich reticulin fiber network of type III collagen (Figure 14–4a). The fibers are produced by fibroblastic reticular cells, which extend numerous processes along and around the fibers (Figure 14–4b). Besides lymphocytes and reticular cells, lymphoid tissue typically contains various APCs and plasma cells.
FIGURE 14–4 Reticular fibers and cells of lymphoid tissue.
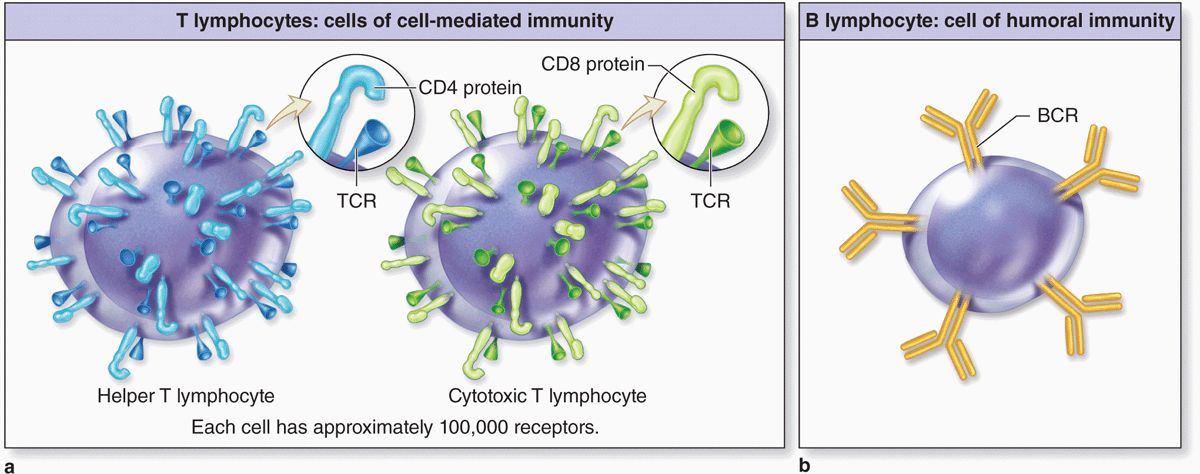
Although most lymphocytes are morphologically indistinguishable in either the light or electron microscope, various surface proteins (“cluster of differentiation” or CD markers) allow them to be distinguished as B cells and subcategories of T cells by immunocytochemical methods. Key features of B and T lymphocytes also include the surface receptors involved in activating their different responses to antigens (Figure 14–5). Receptors of B cells are immunoglobulins that bind antigens directly; those on T cells react only with antigen on MHC molecules and this requires the additional cell surface proteins CD4 or CD8.
FIGURE 14–5 Specific receptors on T and B lymphocytes.
Lymphocytes in the marrow and thymus of a newborn infant not yet exposed to antigens are immunocompetent but naive and unable to recognize antigens. After circulating to the various secondary lymphoid structures, lymphocytes are exposed to antigens on APCs and become activated, proliferating to produce a clone of lymphocytes all able to recognize that antigen.
T Lymphocytes
T cells are long-lived lymphocytes and constitute nearly 75% of the circulating lymphocytes. They recognize antigenic epitopes via surface protein complexes termed T-cell receptors (TCRs). Most TCRs include two glycoproteins called the α and β chains, each with variable regions produced similarly to those of immunoglobulins. Because TCRs only recognize antigenic peptides when presented as part of MHC molecules (interacting with both the MHC and the peptide it presents), T lymphocytes are said to be MHC restricted.
Several types of T lymphocytes exist, with various functions. Important subpopulations of T cells include the following:
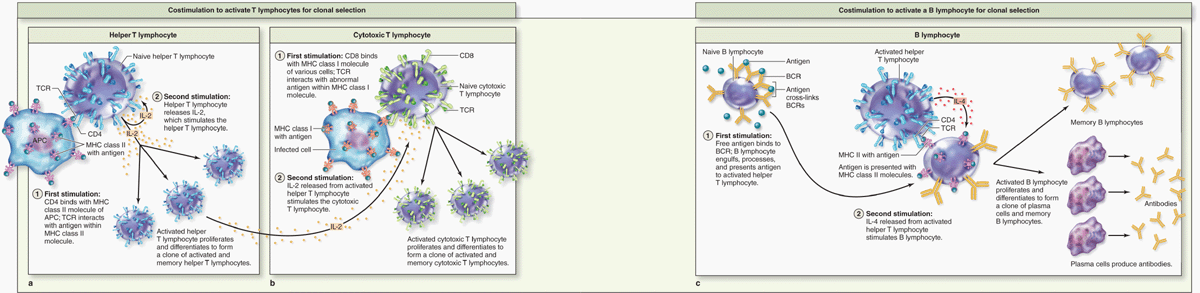
 Helper T cells (Th cells) are characterized by CD4, the coreceptor with the TCR for binding MHC class II molecules and the peptides they are presenting (Figure 14–6a). Activated by such binding, helper T cells greatly assist immune responses by producing cytokines that promote differentiation of B cells into plasma cells, activate macrophages to become phagocytic, activate cytotoxic T lymphocytes (CTLs), and induce many parts of an inflammatory reaction. Some specifically activated helper T cells persist as long-lived memory helper T cells, which allow a more rapid response if the antigen appears again later.
Helper T cells (Th cells) are characterized by CD4, the coreceptor with the TCR for binding MHC class II molecules and the peptides they are presenting (Figure 14–6a). Activated by such binding, helper T cells greatly assist immune responses by producing cytokines that promote differentiation of B cells into plasma cells, activate macrophages to become phagocytic, activate cytotoxic T lymphocytes (CTLs), and induce many parts of an inflammatory reaction. Some specifically activated helper T cells persist as long-lived memory helper T cells, which allow a more rapid response if the antigen appears again later.
FIGURE 14–6 Activation of lymphocytes.
 MEDICAL APPLICATION
MEDICAL APPLICATION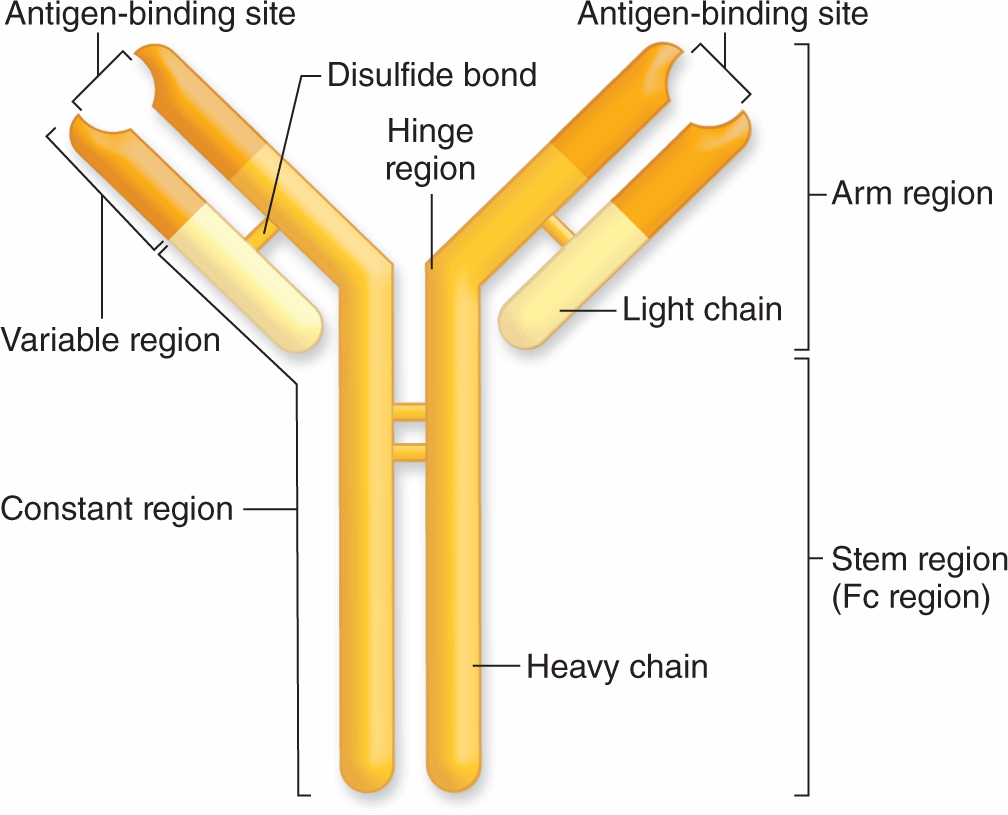
 MEDICAL APPLICATION
MEDICAL APPLICATION