Esophagus: Normal Structures, Developmental Abnormalities, and Miscellaneous Disorders
STRUCTURE OF THE ESOPHAGUS
Anatomy
The esophagus is a muscular tube that is lined with stratified squamous epithelium. Its average length is about 11 cm in newborn and about 25 cm in adults. The upper boundary of the esophagus consists of cricopharyngeal muscle that forms the upper esophageal sphincter at the level of the sixth cervical vertebra. The esophagus enters the stomach at an oblique angle (angle of His) several centimeters below the diaphragm.
Endoscopists measure esophageal landmarks as the number of centimeters from the incisors. The upper esophageal sphincter is located 15 to 18 cm from the incisors. The gastroesophageal junction is classically described as being approximately 40 cm (range 37-42 cm) from the incisors, but this location varies considerably according to the individual’s height and whether a hiatal hernia is present (Fig. 9-1). In the latter instance, the gastroesophageal junction is located, on average, 36 cm from the incisors; however, in a short person with a hiatal hernia, it may be located as little as 30 cm from the incisors.
The endoscopic determination of the location of the gastroesophageal junction is based on visualizing the zigzag ora serrata, known more popularly as the Z-line; this is the interface between the pearly, gray-white squamous epithelium of the esophagus and the pink-orange columnar epithelium of the stomach (Fig. 9-2). It straddles the region of the lower esophageal sphincter (LES), which appears endoscopically as an indented or contracted part of the distal esophagus. There may not be a strict correlation between the location of the LES zone and the histologic esophagogastric (squamocolumnar) mucosal
junction. This is reflected in the endoscopic appearance of the Z-line. Gastric mucosa may thus normally extend into the esophagus in this irregular (Z-line) fashion. Endoscopically the junction of tubular esophagus with the saccular stomach is defined by the upper border of convergence of the gastric folds, as neither the squamocolumnar junction nor the lower sphincter defined by manometry is considered reliable or accurate.
junction. This is reflected in the endoscopic appearance of the Z-line. Gastric mucosa may thus normally extend into the esophagus in this irregular (Z-line) fashion. Endoscopically the junction of tubular esophagus with the saccular stomach is defined by the upper border of convergence of the gastric folds, as neither the squamocolumnar junction nor the lower sphincter defined by manometry is considered reliable or accurate.
The blood supply to the esophagus is from the inferior thyroid artery in the upper segment; bronchial arteries, direct aortic branches, accessory branch of subclavian artery, and sometimes branches from the intercostal arteries in the midsegment; and the left gastric artery and the left phrenic artery in the distal segment. The venous drainage arises from intercommunicating plexuses that are both superficial and deep to the muscle coats. The upper third of the esophagus drains through the inferior thyroid veins into the superior vena cava. The middle third of the esophagus drains into the azygous system, and the lower third into gastric veins and ultimately into the portal vein.
The innervation of the esophagus is from both the vagus nerve and the sympathetic nervous system. Most of the esophageal musculature is supplied from the vagus nerve. Its fibers anastomose with short postganglionic fibers in the myenteric (Auerbach’s) plexuses. The sympathetic fibers to the esophagus are derived from cervical and thoracic sympathetic ganglia. As is true throughout the gastrointestinal (GI) tract, regulatory peptides interact with neural and muscle elements in the control of normal function.1
The mucosal and submucosal lymphatics communicate with longitudinal lymphatic channels in the muscle coats. The upper third of the esophagus drains into the cervical nodes, the middle third into the paraesophageal and paratracheal nodes of the mediastinum, and the lower third into the para-aortic nodes, nodes in the celiac axis, or both.
Histology
Mucosa. The mucosal lining consists of stratified squamous epithelium of the nonkeratinized type (Fig. 9-3). Occasionally, keratohyalin particles are present, although a well-developed granular cell layer is absent, which differentiates it from the squamous
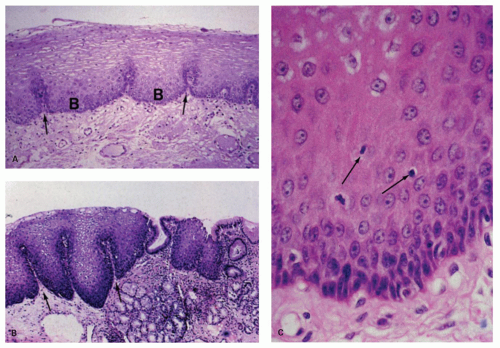
epithelium of skin. With electron microscopy the cell layers have been characterized as basal, prickle, and functional, the last referring to the flattened surface cells.2 The epithelium is generally 15 cell layer thick (350-450 µm). In clinical practice, one need to only distinguish between the basal and overlying nonbasal cells. The basal cell zone is one to four cells thick and does not exceed 15% of the thickness of the squamous layer except in the most distal centimeters of the esophagus.3, 4 The cells above the basal zone contain flattened nuclei and a glycogen-rich cytoplasm. The cells at the surface are elongated, with a very narrow margin of cytoplasm surrounding pyknotic-appearing nuclei. Basal cells are distinguished from nonbasal cells by the fact that the former do not contain glycogen. In clinical practice, using conventionally stained sections, basal cells appear juxtaposed. One rule of thumb used to define the upper margin of the basal layer is to determine where nuclei are separated by a distance of at least one nuclear diameter.5 Sometimes the tongues of columnar mucosa of the gastric type normally extend several centimeters on either side of the apparent LES zone. In addition, glands similar to those of the gastric cardia, with mucous glands and scattered fundic gland elements, commonly extend up into the lamina propria of the esophagus for a short distance.
epithelium of skin. With electron microscopy the cell layers have been characterized as basal, prickle, and functional, the last referring to the flattened surface cells.2 The epithelium is generally 15 cell layer thick (350-450 µm). In clinical practice, one need to only distinguish between the basal and overlying nonbasal cells. The basal cell zone is one to four cells thick and does not exceed 15% of the thickness of the squamous layer except in the most distal centimeters of the esophagus.3, 4 The cells above the basal zone contain flattened nuclei and a glycogen-rich cytoplasm. The cells at the surface are elongated, with a very narrow margin of cytoplasm surrounding pyknotic-appearing nuclei. Basal cells are distinguished from nonbasal cells by the fact that the former do not contain glycogen. In clinical practice, using conventionally stained sections, basal cells appear juxtaposed. One rule of thumb used to define the upper margin of the basal layer is to determine where nuclei are separated by a distance of at least one nuclear diameter.5 Sometimes the tongues of columnar mucosa of the gastric type normally extend several centimeters on either side of the apparent LES zone. In addition, glands similar to those of the gastric cardia, with mucous glands and scattered fundic gland elements, commonly extend up into the lamina propria of the esophagus for a short distance.
Mitoses can be observed in the parabasal cell layer and are associated with increased proliferative activity. No mitoses are detected in the basal cell layer, and presumably the cells move higher up once they enter mitotic cycle. The exact time for cells to move from basal zone to surface for shedding is believed to be <10 days, while in mice and rats this is between 4 and 7 days.
Melanocytes have also been described in some esophagi.6 Melanocytes are believed to occur in between 4% and 8% of esophagi. The latter are likely relevant to the rare primary malignant melanomas of the esophagus (see Chapter 12), in which melanosis may surround the neoplasms.7 The number of melanocytes seems to be increased in many esophageal resections (27%-34%) performed for squamous cell carcinoma for yet unclear reasons. Pseudomelanosis of the esophagus has been reported, but the cause is still unknown.8
Endocrine cells can be observed in the esophageal epithelium as well. The incidence is reported to be about 28%. Mostly these cells are found in the basal cell layer of squamous epithelium. There is an ongoing discussion whether these cells contribute to the regulation of esophageal sphincters. Langerhans cells are also present in the esophageal squamous epithelium that can be highlighted with S100 and CD1a immunostains. Their function is likely to be antigen presentation similar to their role in skin, and their
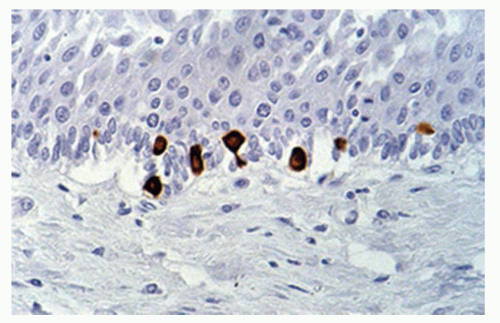
number seems to increase in esophagitis. Merkel cells are also found in the esophageal mucosa.9 Compared to fetuses their numbers are slightly increased in adults (1.2/cm vs. 2.2/cm). The numbers of Merkel cells vary markedly among individuals (Fig. 9-4). Immunohistochemically Merkel cells are positive for CK20. Merkel cells with their long dendritic processes can be identified from the 13th week of gestation onward. They are associated with light touch and in the skin can discriminate shape and textures, but it is unclear if they can do this in the esophagus.
number seems to increase in esophagitis. Merkel cells are also found in the esophageal mucosa.9 Compared to fetuses their numbers are slightly increased in adults (1.2/cm vs. 2.2/cm). The numbers of Merkel cells vary markedly among individuals (Fig. 9-4). Immunohistochemically Merkel cells are positive for CK20. Merkel cells with their long dendritic processes can be identified from the 13th week of gestation onward. They are associated with light touch and in the skin can discriminate shape and textures, but it is unclear if they can do this in the esophagus.
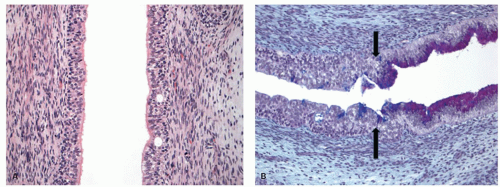
Lymphocytes may be scattered throughout the squamous cell layer, interspersed between epithelial cells. Their nuclei may be compressed (squiggle cells) or lobulated and thus may be mistaken for neutrophils (Fig. 9-5A). Some tiny cell fragments in the epithelium may defy identification of their origin. Immunocytochemical studies show that these are mostly T lymphocytes10 and are CD8 immunoreactive. Intraepithelial mast cells and B cells can also be found.11
The basal cell zone is invaginated by extensions of the lamina propria termed dermal papillae (see Fig. 9-3B). They generally do not extend more than two-thirds of the thickness of the epithelium, except in the distal 2 to 3 cm of the esophagus.3, 4 These papillae contain narrow capillaries; sometimes they are engorged with erythrocytes.
The lamina propria consists of connective tissue, which contains scattered plasma cells and occasional lymphoid nodules (see Fig. 9-3B). Sparsely distributed mucous glands are present in the lamina propria of the distal esophagus. These have been termed esophageal cardiac glands. Near the gastroesophageal junction, these glands may contain oxyntic mucosa glands, such as parietal cells.12
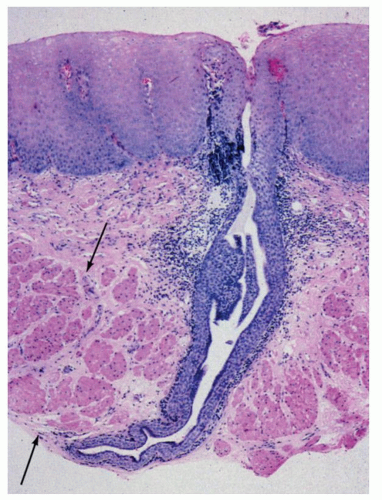
The muscularis mucosae become progressively thicker in the distal part of the esophagus. In biopsy specimens, the unwary morphologist may make the frightening mistake of confusing the thick muscularis mucosae for the muscularis propria (Fig. 9-5). The muscularis mucosa is particularly thick in cases of Barrett’s associated distal esophageal or GE junction cancers and can form multiple layers (duplication of muscularis mucosa).13, 14
Submucosa and muscularis propria. The submucosa consists of loose connective tissue containing blood vessels, nerve fibers, scattered lymphocytes, and small lymphoid follicles. Ganglion cells of Meissner’s submucosal plexus are very sparse in the esophagus. Submucosal esophageal mucous glands are distributed throughout the esophagus (Fig. 9-3B). Ducts from these glands penetrate the muscularis mucosae and end in the epithelium. The ducts are covered with a cuboidal epithelium and then by a stratified epithelium in their terminal parts (Fig. 9-5).15 Focal cystic dilatation of these ducts and glands and surrounding lymphoid collections may be seen in some autopsy specimens of “normal” esophagi.16
The muscle coats that lie outside the submucosa consist of striated muscle in the upper one-quarter to one-third of the esophagus and of smooth muscle in the lower two-thirds of the esophagus. Much of the striated muscle portion (except for the upper esophageal sphincter) is actually a blend of striated and smooth muscles.17 Individual muscle fibers are arranged in a spiral fashion, with inner horizontally curved bands and outer bands with a more longitudinal orientation.15, 17 The ganglion cells of the myenteric plexus (Auerbach’s) are more dense in the distal esophagus. The number of cells of Cajal is related to the plexus density. In achalasia cells of Cajal are more sparse.18 Thus, it is proposed in the esophagus also that cells of Cajal are necessary for a neural-controlled motor function.19, 20
ESOPHAGEAL FUNCTION
Food is transported through the esophagus into the stomach at a speed of about 2 to 4 cm/s by peristalsis. The gastroesophageal sphincter relaxes for few seconds before and after the passage of food. Repeated swallowing results in the relaxation of the sphincter for a longer period. During vomiting the LES relaxes and the gastric content is transported upward due to contractions of abdominal musculature and stomach. Numerous transmitters (prostaglandins, VIP, 5HT3, nitrogen oxide, dopaminergic and H2 receptors, gastrin, motilin, cholecystokinin, etc.) are responsible for the physiological motility of the esophagus and the sphincter activity.
Age-Dependent Changes
The data on age-dependent esophageal changes are very limited. It is known that the smooth musculature does not change with age, but in the elderly the number of ganglia in the Auerbach’s plexus is reduced.23 With age the esophageal lumen is believed to become wider, and the wall consistency and sensitivity to stretching are said to decrease.24
EMBRYOLOGY AND DEVELOPMENT OF THE ESOPHAGUS
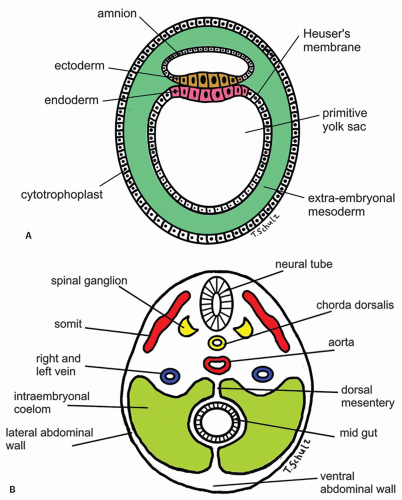
Prenatal life is divided into embryonic (until 9 weeks) and fetal (9 weeks to birth) periods. Around 14th day the embryo develops into a bilaminar disk of ectoderm and endoderm (Fig. 9-6A). The endoderm forms the lining of the yolk sac and is the scaffold for the future GI tract. The mesoderm appears as the third layer around 15th day and gives rise to the connective tissue, vessels, smooth muscle, and serosal layers of the GI tract. Proliferation and segmentation of the mesoderm into somites that takes place between the endoderm and ectoderm induces numerous transformations in the endoderm. During this time the embryo continues to elongate craniocaudally and folds laterally. The embryo thus becomes a cylindrical tube lined with endoderm, which divides the yolk sac into intraembryonic and extraembryonic parts (Fig. 9-6). The intraembryonic part forms the GI tract and its accessory glands (pancreas, gall bladder, and liver). The extraembryonic part regresses and disappears around 12th week. At this point, based on the arterial supply the primitive digestive tube can be divided into foregut, midgut, and hindgut supplied by celiac, superior mesenteric, and inferior mesenteric arteries, respectively.
Development of the GI tract takes place in four axes: Anterior-posterior, dorsal-ventral, left-right, and craniocaudal. Development along each axis is based on the epithelial-mesenchymal interactions mediated by specific molecular pathways and growth factors such as Wnt5a (expressed by mesoderm), endodermal proteins Six2/Sox2, as well as Hoxa-2, Hoxa-3, and Hoxb-4, which control esophageal development in the anterior-posterior axis. These factors affect both the esophageal environment and the migration of the neural crest cells.
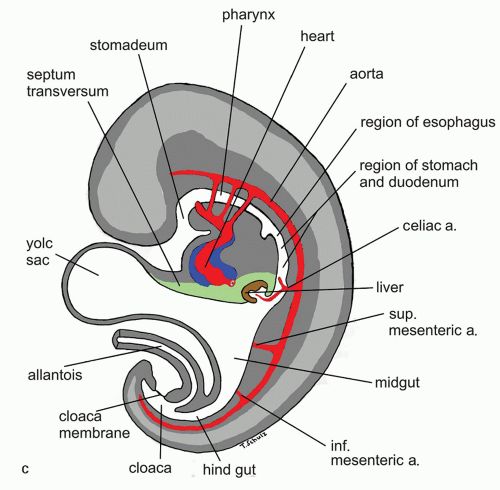 Figure 9-6. (Continued) C: Saggital cut of the embryo at 5th week of gestation showing various developing vascular structures and their relationship to the gut. |
Between the 3rd and 4th weeks of gestation the esophagus becomes recognizable as distinct from the stomach and the pharynx. During week 4, the foregut develops a small diverticulum on its ventral surface adjacent to the pharyngeal gut. The mesenchyme between the esophagus and this diverticulum forms the esophagotracheal septum, which separates the esophagus from trachea during subsequent development. This tracheobronchial diverticulum subsequently elongates caudally and forms the primitive respiratory tract, including the lungs around 26th day.
The remaining part of the foregut rapidly elongates along with the craniocaudal growth of the embryo. Initially the esophagus is lined by ciliated columnar epithelium (Fig. 9-7), and during the 14th week to 4th month it starts getting replaced by stratified squamous epithelium starting from the middle third of the esophagus, a process that continues until birth.25 Portions of ciliated columnar epithelium can persist after birth and even till adulthood.26, 27, 28 In the proximal esophagus so-called inlet patches (gastric heterotopia) likely represent columnar mucosa that develops into gastric mucosa seen in more than 10% of all individuals undergoing upper GI endoscopy.29 Histologically and immunohistochemically fetal esophageal epithelium differs from cardia-epithelium and gastric heterotopia in the proximal esophagus.30 At this time it is strongly positive for CK19 and negative for CK7 and CK20. The esophageal epithelium during the fetal period undergoes marked proliferation, nearly obliterating the entire lumen. Unlike other species, complete occlusion of the foregut by this process has not been observed in human embryos. By the 10th week, vacuolation appears in the luminal cells of the foregut, which coalesce to form a central esophageal lumen lined by a superficial layer of ciliated epithelial cells. During the 4th month the longitudinal mucosal folds develop.31 The primitive foregut endoderm is the origin for both the future esophageal epithelium and submucosal glands. Esophageal glands develop during the 7th month from the esophageal epithelium.
During the 6th week of gestation, the circular muscle coat and ganglion cells of the myenteric plexus form. During the 7th week, blood vessels enter the submucosa. The muscularis propria develops around the 6th week from the surrounding mesenchyme. Interestingly, the inner circular layer develops a month earlier than the outer longitudinal layer. During 4th to 7th months the muscularis mucosae start developing from distal to proximal.
The vagal nerve innervates the esophageal musculature. By the 5th month fully developed neural plexus can be found (Meissner and Auerbach), and ganglia
can also be identified by the 7th week.25 From animal experiments it is known that interstitial cells of Cajal in the neural plexus and the circular muscle build networks for neural motor function. At time of birth it is concluded that neural networks are more mature in esophagus and stomach. Esophageal development of nerve and Cajal cell activity precede that in the remaining gut but have not achieved adult organization at time of birth. The timepoint of migration of Cajal cells into esophageal tissue is not yet known in detail. These findings explain that gut motility at birth does not show an adult pattern.32 The smooth muscle of the lower esophagus and the LES is derived from the mesenchyme of the somites surrounding the foregut. The striated muscle in the muscularis propria of the upper part of the esophagus and the upper esophageal sphincter is derived from mesenchyme of the fourth, fifth, and sixth branchial arches. This explains the innervation of the upper esophageal sphincter by the vagal nerve (the nerve of fifth branchial arch) and by the recurrent laryngeal nerve (a branch of the vagus nerve and the nerve of sixth branchial arch). The embryologic origin of the gastroesophageal junction is still controversial, but gastric rotation, together with augmentation of the fundus of the stomach, is believed to determine its formation.
can also be identified by the 7th week.25 From animal experiments it is known that interstitial cells of Cajal in the neural plexus and the circular muscle build networks for neural motor function. At time of birth it is concluded that neural networks are more mature in esophagus and stomach. Esophageal development of nerve and Cajal cell activity precede that in the remaining gut but have not achieved adult organization at time of birth. The timepoint of migration of Cajal cells into esophageal tissue is not yet known in detail. These findings explain that gut motility at birth does not show an adult pattern.32 The smooth muscle of the lower esophagus and the LES is derived from the mesenchyme of the somites surrounding the foregut. The striated muscle in the muscularis propria of the upper part of the esophagus and the upper esophageal sphincter is derived from mesenchyme of the fourth, fifth, and sixth branchial arches. This explains the innervation of the upper esophageal sphincter by the vagal nerve (the nerve of fifth branchial arch) and by the recurrent laryngeal nerve (a branch of the vagus nerve and the nerve of sixth branchial arch). The embryologic origin of the gastroesophageal junction is still controversial, but gastric rotation, together with augmentation of the fundus of the stomach, is believed to determine its formation.
DEVELOPMENTAL AND CONGENITAL ANOMALIES
Esophageal Atresia and Tracheoesophageal Fistulas
Esophageal atresia along with tracheoesophageal fistula is the most frequent esophageal anomaly (1:3,000-5,000 births), although isolated esophageal atresia is quite rare.33 It is unclear whether this results from lack of proper separation of foregut into respiratory and intestinal tracts by the growth of tracheoesophageal septum or improper recanalization of the esophagus following esophageal epithelial proliferation.25 The use of teratogenic substances during pregnancy has been implicated in the development of esophageal atresia. Frequently additional anomalies involving other organ systems are also present (Table 9-1; Fig. 9-8).
Esophageal atresia occurs in five different forms (Table 9-2).34, 35 Unfortunately, the terminology in the literature is not consistent and many different classification schemes are in use resulting in some confusion (A-E, Type I-V, Type 1-5, Type I-IIIc). The most common variant is type C (Table 9-2) with a proximal blind esophageal pouch and a fistula between the trachea and the distal esophagus. The fistula often enters the trachea close to the carina. The second most common anomaly is type A—pure esophageal atresia without tracheoesophageal fistula.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree