Bladder
THE UNREMARKABLE AND NONNEOPLASTIC BLADDER ANATOMY AND HISTOLOGY
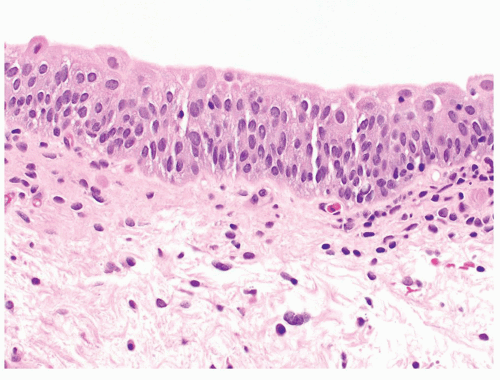
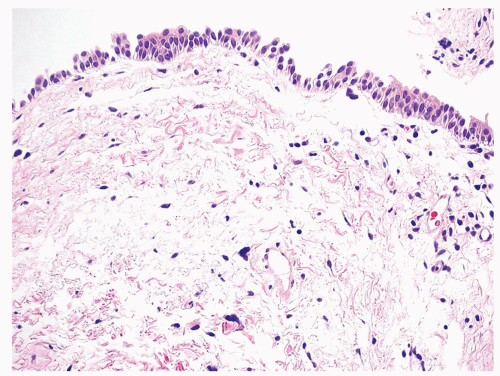
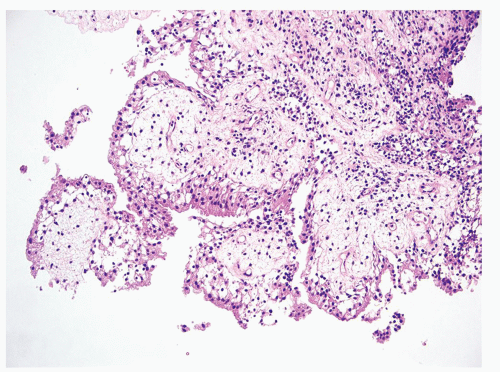
The urinary bladder is a hollow viscus situated in the deep pelvis, which can expand to hold up to 500 mL of urine. Expansion of the bladder is permitted by interlacing bundles of smooth muscle and its multilayered urothelial lining which can fold into the bladder when empty. Urothelium is present throughout the urinary tract, starting from the renal pelvis, extending to the ureters, bladder, and terminating in the urethra, where it transitions to squamous epithelium. The general histology of these anatomic areas is similar, with a 5 to 7 cell layer-thick urothelium lined by umbrella cells. Beneath the urothelium lies the lamina propria, loose connective tissue containing numerous small lymphatics, and the wispy muscle fibers of the muscularis mucosae (Figures 2.1,2.2,2.3,2.4). Of note, the trigone muscle layer is derived from the bladder detrusor muscle and the ureter muscle and, therefore, lacks muscularis mucosae. The detrusor muscle, or muscularis propria, of the bladder is the deepest layer, containing thick fascicles of smooth muscle that interdigitate to allow for expansion and contraction of the bladder. The bladder is loosely encased in a layer of adipose tissue, the perivesical fat, which permits expansion as the bladder fills.1
The bladder is a roughly pyramid-shaped organ, with the point of the pyramid aligned inferiorly toward the urethra and the flat “dome” situated superiorly. The other anatomic regions of the bladder include the trigone, a flattened triangular area of urothelium located between the ureteral orifices and urethra on the posterior wall. The ureteral orifices, also denoted as the ureterovesical junctions, are located on the right and left sidewalls of the bladder, and the ureter courses through the muscularis propria within the bladder for some distance before emerging into the retroperitoneum and terminating in the renal pelvis.
PEARLS AND PITFALLS: Normal Elements of Bladder Wall
Fat can be identified at any level within the bladder wall (Figure 2.5)
Presence of fat in a TURBT (transurethral resection of bladder tumor) specimen should not prompt concern for perforation or involvement of perivesical tissue
Ganglia and paraganglia are a normal component of bladder and may be identified in the muscularis propria (Figure 2.6)
THE NEAR-NORMAL BLADDER
von Brunn Nests
von Brunn nests (VBN) are rounded clusters of normal urothelium located within the lamina propria. They occur in both ureter and bladder. VBN represent downward invagination and budding of the urothelial surface and as such are a normal part of the urothelial mucosa. They are usually well circumscribed with focal attachment to the surface urothelium, depending on the plane of section. They tend to appear at roughly the same horizontal depth in the lamina propria, without irregular extension beyond that level.2 VBN proliferations are common in the ureter and should not be confused for a neoplasm in a small biopsy specimen (Figure 2.7). However, there is morphologic overlap with proliferation of VBN and superficial sampling of nested variant of urothelial carcinoma, which will be discussed later in the chapter (Figure 2.8). One should be especially cautious of diagnosing nested variant of urothelial carcinoma in the ureter because of the frequency of proliferation of VBN in this location.
SAMPLE NOTE: Superficial Sampling of Atypical Urothelial Proliferation (See Note)
Note: The specimen is composed of a superficial sampling of urothelium with rounded nests of urothelial cells present in the lamina propria. The differential diagnosis includes proliferation of VBN and nested variant of urothelial carcinoma. Recommend clinical correlation and repeat sampling if warranted.
 Figure 2.8. Higher power image of von Brunn nests (VBN) from Figure 2.7 showing bland cytology, focal cystic change (top right of image). In small and superficial biopsy specimens, a proliferation of small urothelial nests such as this raises the differential diagnosis of VBN and small nested urothelial carcinoma. A diagnosis of “atypical urothelial proliferation” is warranted for these types of cases. |
Chronic irritation may lead to squamous metaplasia of the urothelium. The usual transitional type epithelium is replaced with squamous epithelium (Figure 2.9). Common causes include chronic urinary tract infections, chronic catheterization, and some specialized infections such as schistosomiasis. The metaplastic epithelium may be glycogenated (most commonly found in the trigone of the female bladder) or keratinizing. Keratinizing squamous metaplasia carries an increased risk of development of carcinoma, and its presence should be included in the final report (Figures 2.10 and 2.11).
BIOPSY AND TRANSURETHRAL RESECTION OF BLADDER TUMORS
Cystoscopically, the unremarkable bladder reveals a pink-tan smooth to mildly trabeculated surface without exophytic growths or marked erythema. Using ureteroscopy, the upper urothelial tract demonstrates similar white-tan flattened urothelium lining the ureters and renal pelvis.
Histologic evaluation of the bladder is achieved by the use of cold cup biopsy or TURBT. Cystoscopy is a procedure that fills the bladder with clear fluid and evaluates the mucosa using white- or blue-light flexible scopes, looking for mucosal irregularities or tumors.
Cystoscopy can be performed in the urologist’s office, without anesthesia, for surveillance of bladder cancer recurrence, initial workup of hematuria, or other lower urinary symptoms. During office-based flexible cystoscopy, cold cup biopsies may be obtained for evaluation of mucosal abnormalities. Specific cystoscopic findings include erythema, which may represent cystitis or urothelial carcinoma in situ (CIS), papillary neoplasms, or obvious invasive tumors. Some cystoscopic findings may suggest a high-grade tumor, such as necrosis or sessile growth pattern; however, histologic confirmation is necessary for final diagnosis. Blue-light cystoscopy is a recent technological advance where a light-sensitive substance (hexaminolevulinate) is instilled in the bladder prior to cystoscopy. Blue-light outperforms white-light cystoscopy in the detection of CIS and other high-risk lesions.3,4
Cold cup biopsies are subject to a number of artifactual distortions. When a large bite of tissue is present, you may have all layers of the bladder represented even in a small biopsy (Figure 2.12). However, these small biopsies often show significant crush artifact given the pincher-type forceps that are used. This can lead to difficulty assessing nuclear atypia for grading and displacing urothelial cells into the lamina propria, mimicking invasion. When possible, attempts should be made to identify intact, unperturbed urothelium for evaluation (Figure 2.13). Chronic inflammation, especially when crushed, can also mimic invasive disease (Figure 2.14). A simple pan-keratin stain can be used in this instance to rule out invasive carcinoma. With increased inflammation comes other changes in both the urothelium and the possibility of nonurothelial diagnoses; disrupted urothelial surfaces may harbor nephrogenic adenomas (Figure 2.15).
PEARLS AND PITFALLS: Cystoscopy and Ureteroscopy
Cystoscopy notes can provide guidance for the pathologist and should be reviewed with each bladder biopsy or TURBT specimen
If you are uncertain if a lesion represents a true papillary neoplasm, check the cystoscopic appearance in the procedure note. Papillary tumors are usually quite evident by cystoscopy; beware making a diagnosis of a papillary neoplasm if none was seen on cystoscopy
Ureteroscopy involves a very small scope and, therefore, results in very small biopsies. Beware over diagnosing a neoplasm or invasion on a small, crushed biopsy, as the clinical ramifications are very significant in the upper urinary tract
 Figure 2.15. Higher power image of the nephrogenic adenoma present in Figure 2.14. Multiple tubules are present in lamina propria and lining of both tubules and surface shows hobnail cytology. The tubules are lined with an amorphous pink basement membrane-like material. |
FAQ: What is the Clinical Approach to Workup of a Suspicious Bladder Mass?5
Suspicious bladder lesions are first evaluated with office cystoscopy, urine cytology, and biopsy or TURBT
Once tumor is identified, secondary evaluation includes complete TURBT
Noninvasive low-grade tumors may be observed or treated with intravesical therapy
Lamina propria involvement by low-grade tumors should be followed up with repeat TURBT; if residual disease, cystectomy may be considered
If high grade is suspected based on sessile appearance, complete TURBT with sampling of muscularis propria is necessary; possible mapping biopsies with sampling of prostatic urethra
Non-muscle-invasive high-grade tumors may be observed or treated with intravesical therapy
Muscle-invasive high-grade tumors require definitive treatment with cystectomy and possible neoadjuvant chemotherapy
Once a tumor is detected with cystoscopy and cold cup biopsy, a larger resection may be necessary for accurate staging of the tumor. A TURBT can be performed during cystoscopy under spinal or general anesthesia, as it requires longer procedure time and removal of deeper layers of the bladder wall. The general purpose of a TURBT is to remove all possible tumor burden, with sufficient sampling of muscle wall for accurate staging. The most critical element for staging is evaluation of muscularis propria invasion.
TURBT is performed using electrocautery, and therefore, a major artifact is thermal distortion of tissue (Figure 2.16). The urologist may start the resection with the current turned to a relatively high level, causing marked distortion and destruction of superficial tumor. Assigning histologic grade can be difficult in these cases, with low-grade or normal urothelium showing artifactual hyperchromasia, nuclear enlargement, and crowding (Figures 2.17 and 2.18). Ideally, the current is lower as the urologist performs the deeper part of the resection involving detrusor muscle; however, cautery at the deep aspect of tumors may lead to an inability to determine if a muscle bundle is detrusor or if tumor is present. In some cases, a cytokeratin stain may be useful to highlight cauterized tumor, and in some cases, adding desmin staining can highlight obscured large muscle bundles.
 Figure 2.18. Thermal artifact has completely obscured this small fragment of urothelium. No definitive diagnosis can be rendered in cases with this degree of cautery. |
A TURBT produces irregularly sized chips of tumor and bladder, which may be embedded tangentially and lead to artifacts. When a flat lesion is sectioned in a nonperpendicular plane, it may appear artifactually thickened (Figures 2.19,2.20,2.21,2.22). This could lead to an overdiagnosis of urothelial hyperplasia or potential confusion for CIS if through the basal layers. For papillary lesions, tangential sectioning can obscure the papillae entirely by obliquely sectioning one side of the fibrovascular core (Figures 2.23 and 2.24). In these cases, identifying other sectioned cores may help confirm the diagnosis of a papillary lesion. Tangential sectioning of an obvious papillary lesion may lead to confusion over assignment of histologic grade. Similar to the flat lesion that is sectioned through the basal layer, tangential sectioning of a papillary lesion may also result in the appearance of a higher-grade lesion. A pseudopapillary appearance can also occur in small biopsy specimens when the urothelial surface becomes detached and folds over on itself (Figures 2.25 and 2.26). A thorough search for definitive fibrovascular cores is critical before diagnosis of a papillary neoplasm.
 Figure 2.21. Deeper level of same specimen as Figure 2.19 showing the area of thickened urothelium now much thinner with better orientation of urothelial surface. |
 Figure 2.22. Higher power image of same area as Figure 2.20. Urothelium is thinner, and the few darker cells appear to be predominantly umbrella cells. Polarity is minimally altered, as compared with the artifactually jumbled in Figure 2.20. When possible, interpret the most well-oriented level of tissue available on the slide, or order recut levels if you have any suspicion that tangential sectioning is confounding interpretation. |
KEY FEATURES: Necessary elements for pathology report for a bladder biopsy or TURBT:
Histologic grade
Presence/absence of invasion
Depth of invasion
Presence/absence of muscularis propria in the specimen
Presence/absence of CIS
Presence/absence of lymphovascular space invasion
The American Joint Committee on Cancer (AJCC) staging for urothelial cancer differs slightly depending on whether or not the lesion is flat or papillary.6 For flat lesions, a noninvasive in situ component is represented by CIS and is staged as pTis. A noninvasive papillary lesion, regardless of its cytologic grade, is staged as pTa. For tumors that invade the lamina propria, the staging is the same regardless of the architectural pattern of the tumor.
CHECKLIST: Systematic Approach to Bladder Biopsy or TURBT Specimen
□ Assess urothelial surface
Thickness
Normal thickness, consider benign urothelial tissue, papilloma
Thickened, consider hyperplasia, CIS or papillary neoplasm
Architecture
Flat
Papillary
Atypia
None—benign, papilloma
Mild—reactive changes, PUNLMP
Moderate—dysplasia, low-grade papillary
Severe—CIS, high-grade papillary
Assess lamina propria
Invasion—scan for irregular nests, small nests or single cells, paradoxical maturation (round and pink)
Evidence of treatment effect—BCG (bacillus Calmette-Guerin) granulomas, radiation changes
□ Assess muscularis propria
Presence/absence of muscularis propria—adequacy of TURBT resection
Invasion—must involve thick, round muscle bundles for definitive muscularis propria invasion
SURFACE LESIONS OF THE BLADDER
Surface lesions of the urothelium generally correspond to mucosal surface irregularities seen on cystoscopy, including erythema or a velvety, irregular mucosa. Urologists sample these mucosal changes by cold cup biopsy. Cold cup biopsies are ideal for initial evaluation of mucosal findings, because they can be done in the office without general anesthesia, unlike a TURBT. A downside to cold cup biopsy is the relatively small sample size, which is prone to tangential sectioning and potentially tissue exhaustion if the block is not leveled carefully during sectioning. Clinical findings that may prompt an office biopsy include hematuria, lower urinary tract symptoms, or recurrent urinary tract infections. Each of these clinical scenarios may contribute valuable information to your interpretation of the bladder biopsy, so a review of the cystoscopy procedure notes and clinic notes is critical to accurate diagnosis.
FLAT LESIONS OF THE BLADDER
Using the “Checklist: Approach to Bladder Biopsy or TURBT Specimen,” the first step is to scan the surface epithelium to detect changes in the thickness of the urothelium, overall architectural pattern, and assess for presence of atypia. If the urothelium is of normal thickness (5-7 cells), is predominantly flat, and lacks atypia at medium magnification (10×), this most likely represents a benign process (Figure 2.27). The cells are aligned in an orderly manner, with the long axis of nuclei oriented perpendicular to the interface with the lamina propria. An intact umbrella cell layer is useful, but can be seen in atypical and malignant processes as well. Cytologic findings in benign urothelium include dispersed chromatin and longitudinal grooves within the nucleus (Figure 2.28). Benign urothelial mucosa may be sampled due to equivocal cystoscopic findings or in an effort to map out areas of CIS in the bladder. Areas of increased vascularity or dense chronic inflammation may also contribute to an unusual cystoscopic appearance, leading the urologist to biopsy the area (Figure 2.29).
Chronic Cystitis
Chronic cystitis is a general clinical term to describe inflammation within the bladder manifested by lower urinary tract symptoms, including urinary frequency, painful urination, or hematuria. There is not always a direct correlation between the clinical impression of cystitis and the histologic appearance. However, histologic descriptive diagnoses in this category include follicular cystitis, interstitial cystitis, and polypoid cystitis.
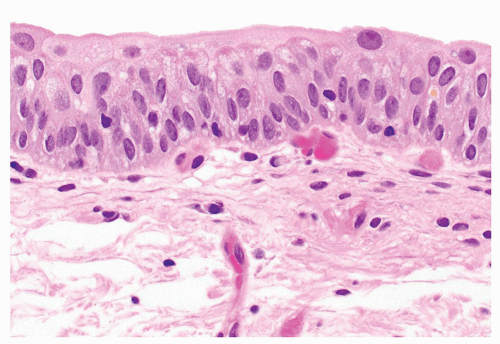 Figure 2.28. The cytologic features of benign urothelium include abundant eosinophilic cytoplasm and nuclei with open chromatin with occasional nuclear grooves. |
Follicular cystitis is a form of chronic inflammation in which germinal centers have formed within the lamina propria (Figure 2.30). The histologic appearance is nonspecific, but may be related to chronic urinary tract infection or other inflammatory reaction to BCG or intravesical chemotherapy. Cystoscopically, a small mucosal bump may be seen representing the bulging of the urothelium over the well-formed germinal centers. Interstitial cystitis is a clinical diagnosis often made in older women with painful bladder symptoms. These symptoms may prompt cystoscopic evaluation, which can show erythematous, friable bladder mucosa. Unfortunately, there are no specific pathologic diagnostic criteria for interstitial cystitis, though clinicians often ask for a specific diagnosis in this entity (Figures 2.31 and 2.32). The histopathologic findings in these biopsies are not predictive of severity of disease.7,8 The “Hunner ulcer” is often ascribed to this entity, though it is not specific for interstitial cystitis. Cystoscopically, Hunner ulcer shows a heaped up nodule with small vessels radiating outward. When biopsied, the Hunner ulcer is usually wedge shaped, with punctate hemorrhage and surrounding granulation tissue. No topline diagnosis of interstitial cystitis should be rendered; rather, excluding a more serious condition of dysplasia, CIS, or malignancy and a description of the findings is appropriate.
Occasionally, clinicians may ask for a mast cell count as part of their workup for interstitial cystitis; however, no diagnostic cutoff for mast cell count exists, and their significance is uncertain (Figures 2.33 and 2.34).
Occasionally, clinicians may ask for a mast cell count as part of their workup for interstitial cystitis; however, no diagnostic cutoff for mast cell count exists, and their significance is uncertain (Figures 2.33 and 2.34).
 Figure 2.32. Medium-power image of Figure 2.31 showing increased chronic inflammatory cell infiltrate and vessels with overlying urothelium with reactive atypia. |
 Figure 2.33. At higher power, the chronic inflammatory cells are a mix of lymphocytes, plasma cells, and occasional mast cells. |
KEY FEATURES: Spectrum of atypia and dysplasia
Atypia is defined as the presence of one or more cellular or architectural features that deviate from that of an otherwise normal appearing cell or group of cells9
Cytologic atypia is manifest by nuclear enlargement, change in nuclear shape, chromatin clumping, prominent nucleoli, or irregular nuclear outlines
Architectural atypia includes denudation, thickening, or changes in cell polarity
Reactive changes are included in atypia
Dysplasia is defined as a preneoplastic or neoplastic process
Cytologic and architectural dysplasia are evidenced by more severe alterations in the cells than expected in atypia
Changes worrisome for dysplasia or CIS should be included in the dysplastic category
Urothelium With Reactive Changes
Urothelium that either is thickened or demonstrates atypia at medium magnification should be closely examined for a more specific finding. First, reactive changes should be considered and the patient’s clinical history should be examined for potential causes, including urinary tract infection, stones, instrumentation or catheterization, and history of intravesical therapy or radiation. Clinical presentation includes lower urinary tract symptoms such as frequency, urgency, or dysuria. Cystoscopic findings demonstrate patches of erythema or roughened urothelium.
PEARLS AND PITFALLS: Reactive Urothelial Changes
No change or mild thickening of the urothelium
Obvious inflammation, often in the form of lymphocytes or neutrophils in the lamina propria
Inflammatory cells within the urothelium is highly suggestive of a reactive pattern when only mild-moderate atypia exists
One should exercise extreme caution in rendering a diagnosis of dysplasia or CIS in the setting of inflammation
Characteristic cytologic features of reactive changes may also be helpful in confirming reactive changes. Reactive urothelial cells are only slightly enlarged with vesicular or dispersed chromatin and a central small, but prominent nucleolus, in contrast to the hyperchromatic nuclei of CIS (Figures 2.35 and 2.36). Mitotic figures can be frequent in reactive urothelium, often confined to the basal half of the urothelium. The presence of mitoses does not necessarily connote dysplasia or worse. The routine use of Ki-67 for the assessment of CIS in a specimen with other reactive changes may confuse the issue, as an increased proliferation index is common in reactive specimens.
PEARLS AND PITFALLS: Malakoplakia
Reactive process in the bladder that can mimic malignancy
Caused by defective phagocytosis of urinary bacteria, usually Escherichia coli
Classic findings of histiocyte-rich mixed inflammation within the lamina propria (Figures 2.37 and 2.38)
Michaelis-Gutmann bodies are targetoid inclusions within histiocytes comprised of bacterial products, highlighted by von Kossa calcium or iron stain (Figure 2.39)
 Figure 2.39. von Kossa stain highlights the calcium-rich Michaelis-Gutmann bodies found in malakoplakia. |
Iatrogenic and Treatment-Related Cystitis
Iatrogenic causes of cystitis are frequently identified in bladder biopsies, due to the surveillance of many bladder tumors and repeat biopsies. In patients with high-grade urothelial cancer treated with intravesical therapy, BCG, mitomycin C, or gemcitabine may also induce specific changes in the bladder. Granulomatous inflammation is a common response to both prior biopsy and intravesical BCG. Subtle differences in the nature of the granulomas, in addition to good clinical history, can help distinguish between the two causes. Post-TURBT granulomas show central brightly eosinophilic amorphous material, consistent with fibrinoid necrosis, surrounded by palisading histiocytes (Figures 2.40 and 2.41).10 Foreign body-type giant cells are also a common finding after any surgical procedure (Figure 2.42). A prominent eosinophilic infiltrate is common after TURBT and does not connote allergic response (Figures 2.43 and 2.44). BCG granulomas are more consistent with the small epithelioid granulomas expected in tuberculosis infection, may be caseating or noncaseating, and may extend deep in the bladder wall.11 If these types of granulomas are observed in a patient without a history of BCG therapy, it is recommended that one perform stains
for acid-fast and fungal organisms. In a patient with prior BCG treatment, one should not perform acid-fast stains, as they may highlight nonpathogenic acid-fast bacilli and lead to confusion.12
for acid-fast and fungal organisms. In a patient with prior BCG treatment, one should not perform acid-fast stains, as they may highlight nonpathogenic acid-fast bacilli and lead to confusion.12
 Figure 2.41. At higher power, the palisading nature of the histiocytes in post-TURBT (transurethral resection of bladder tumor) granuloma is evident. |
 Figure 2.42. Any prior surgical procedure may result in foreign body giant cell reaction, including post-TURBT (transurethral resection of bladder tumor). |
Pseudocarcinomatous Urothelial Hyperplasia
A very specific reactive pattern may be observed in urothelium following radiation treatment or other ischemic insult. Pseudocarcinomatous urothelial hyperplasia (PCUH) classically occurs in the setting of pelvic radiation, but can be associated with any vascular insult, and may present clinically with hematuria.13,14 In addition to the clinical history of radiation, the key to the diagnosis is identifying hemorrhage, fibrin, and fibrin thrombi within the lamina propria and a regenerative appearance to the urothelium (Figures 2.45 and 2.46). PCUH shows atypical urothelial cells which may form small nests and appear to invade the lamina propria (Figures 2.47,2.48,2.49). The nests may show retraction artifact, suggestive of invasion.15 The urothelial nests occupy the superficial lamina propria and show minimal cytologic atypia with absent mitotic activity.
 Figure 2.46. At high power, the hemorrhage, extravasated blood cells and organizing fibrin, is evident in the lamina propria, along with scattered hemosiderin deposition. |
SAMPLE NOTE: Pseudocarcinomatous Urothelial Hyperplasia (See Note)
Note: The specimen demonstrates urothelium with atypia and downward growth into the lamina propria. Numerous vessels and hemorrhage are evident within the lamina propria, many of which show fibrin thrombi. The overall appearance is that of a reactive and regenerative process, consistent with PCUH. This entity is associated with prior radiation, chemotherapy, or other ischemic process.
Reference:
Kryvenko ON, Epstein JI. Pseudocarcinomatous urothelial hyperplasia of the bladder: clinical findings and follow-up of 70 patients. J Urol. 2013;189(6):2083-2086.
 Figure 2.51. Another example of flat urothelial hyperplasia showing well-oriented, but thickened, urothelium without atypia. |
Flat Urothelial Hyperplasia
Significantly thickened, but flat urothelium with minimal atypia falls into the category of flat urothelial hyperplasia, or urothelial proliferation of uncertain malignant potential (UPUMP) (Figures 2.50 and 2.51). There are no specific symptoms associated with hyperplasia, and it is likely sampled as a subtle irregularity seen on cystoscopy for workup of hematuria or bladder cancer surveillance. In many cases, it is even more likely that it represents an artifact of tangential sectioning of normal urothelium. Flat urothelial hyperplasia may be associated or adjacent to low-grade papillary urothelial carcinoma and is a possible precursor lesion.16 Cells can show some mild loss of polarity, but maturation is preserved, and there should not be any cytologic atypia or mitotic figures identified (Figure 2.52).17 This diagnosis should be used sparingly, with more definitive diagnostic categories considered where possible.
Flat Urothelial Lesions With Significant Atypia or Dysplasia
Flat urothelium with atypia raises several diagnostic possibilities, largely dependent on the severity of atypia. If the atypia is significant, i.e., beyond that expected with reactive
changes, the spectrum of dysplasia includes atypia of unknown significance, urothelial dysplasia, and urothelial carcinoma in situ.
changes, the spectrum of dysplasia includes atypia of unknown significance, urothelial dysplasia, and urothelial carcinoma in situ.
 Figure 2.54. In atypia of uncertain significance, scattered larger and darker nuclei are seen in the urothelium, without obvious inflammation or clinical history to explain the changes. |
Atypia of Unknown Significance
When urothelial atypia beyond that expected from reactive changes is present, or no explanation for reactive-type atypia can be readily identified, the lowest level of atypia one can diagnose is atypia of unknown significance. This category was introduced into the ISUP/WHO consensus guidelines in 1998.18,19 The atypical cells may be slightly enlarged with some hyperchromasia and nuclear pleomorphism (Figures 2.53,2.54,2.55,2.56,2.57,2.58). This finding falls short of the diagnosis of dysplasia, but the atypia is concerning or unexplained by other causes (inflammation, instrumentation, stones, radiation, intravesical therapy, etc.). A diagnosis of atypia of unknown significance carries no established risk of adverse outcomes and in general is thought to be equivalent to a diagnosis of reactive atypia.20 Because of the lack of clinical significance for this diagnosis, every attempt should be made
to find a more concrete category for these minimal changes when possible. However, in rare cases, such terminology may be necessary to convey a lack of certainty regarding the pathologic findings. Numerous images are included as this diagnosis exists on a spectrum between reactive atypia and dysplasia, and significant subjectivity exists in the diagnosis (Figures 2.57 and 2.58).
to find a more concrete category for these minimal changes when possible. However, in rare cases, such terminology may be necessary to convey a lack of certainty regarding the pathologic findings. Numerous images are included as this diagnosis exists on a spectrum between reactive atypia and dysplasia, and significant subjectivity exists in the diagnosis (Figures 2.57 and 2.58).
 Figure 2.58. Higher power image of Figure 2.57 showing scattered atypical cells in a background of cells with open chromatin and rare nuclear grooves. |
Urothelial Dysplasia
Urothelial dysplasia is a histologic term describing flat urothelium with atypia that is beyond reactive changes, but which does not have significant cytologic or architectural disarray to reach a diagnosis of CIS (Figures 2.59 and 2.60).17 Unfortunately, because it lacks specific diagnostic criteria, this entity suffers from poor interobserver reliability and lacks good data on outcomes. Regardless, it is felt to represent a preneoplastic lesion, with some risk of developing CIS and warrants clinical follow-up.21
Urothelial Carcinoma In Situ
At the far end of the spectrum of atypia in flat urothelium, urothelial carcinoma in situ (CIS) shows high-grade cytology with enlarged hyperchromatic cells with obvious disorganization and frequent mitoses (Figures 2.61,2.62,2.63). The marked atypia and hyperchromasia in these cases is apparent at scanning power (10×), though more subtle findings may exist in the form of small cell CIS or CIS that is partially or predominantly denuded (Figure 2.64). The loss of orientation of the urothelium to the surface is also evident at low power, with a jumbled appearance and lack of even distribution of cells throughout the urothelium (Figures 2.65 and 2.66). CIS cells are dyscohesive and prone to denudation; identification and close evaluation of denuded urothelium may actually alert the astute pathologist to the possibility of CIS. Various morphologic appearances exist in CIS, including large cell with nuclear pleomorphism, large cell without nuclear pleomorphism, small cell, and clinging and pagetoid spreading type (Figures 2.67,2.68,2.69).22 It is important to note that an intact umbrella cell layer does not exclude the possibility of CIS, especially when present in the pagetoid form.
PEARLS AND PITFALLS: Urothelial Carcinoma In Situ
CIS is usually evident at 10× scanning magnification. If you are going to high power to convince yourself of atypia, it is probably not CIS!
Nuclear enlargement of 4 to 5× the size of a resting lymphocyte is a helpful diagnostic criterion for CIS; however, small cell CIS does exist
Always raise your suspicion for CIS in denuded specimens; hunt around the edges for focally intact urothelium and level into the block if any concerning features are identified (Figure 2.70)
In denuded specimens, recommending urine cytology may capture dyscohesive CIS cells shed from the bladder wall
Immunohistochemistry for Urothelial Dysplasia/Carcinoma In Situ
In general, immunohistochemistry is not entirely robust for resolving the differential diagnosis between urothelial reactive changes and CIS; however, a number of potential markers have been studied for this purpose. The most commonly used antibodies include cytokeratin 20 (CK20), p53, and sometimes CD44.22,23,24 Other markers that have been studied or proposed include cytokeratin 5/6, p16, alpha-methylacyl-CoA racemase (AMACR), HER2, ProEx C, Ki67, among others.24,25,26,27,28,29
 Figure 2.66. Higher power of Figure 2.65 showing marked atypia, nuclear enlargement, and frequent mitotic figures in a disorganized, jumbled urothelium. The cellular dyscohesion is evident, as well as loss of the umbrella cell layer. |
With regard to the most common markers, CK20 generally shows negative staining or weak, subset staining of the superficial cells only in benign or reactive epithelium, whereas full-thickness staining favors a neoplastic lesion. For CD44, a reactive pattern is full-thickness staining of the reactive epithelium, whereas reduced staining in the neoplastic cells is found with CIS. For p53, a heterogeneous pattern of variable weak to moderate staining (a wild-type pattern) would be consistent with normal or reactive epithelium, whereas diffuse strong staining (and possibly completely negative/“null” staining) would favor CIS (Figure 2.71, Table 2.1).30
However, these stains can be fraught with technical and interpretative challenges, such that in a given patient, the pattern may not be definitive. For example, a moderate degree of p53 staining can be difficult to discern from strong diffuse staining, depending on laboratory technical staining conditions. In one study of diagnostically difficult cases where immunohistochemistry was used, very few patients without a history of prior urothelial
neoplasm went on to develop urothelial carcinoma, suggesting that careful attention to morphology and clinical history is more important than an immunohistochemical staining pattern.23
neoplasm went on to develop urothelial carcinoma, suggesting that careful attention to morphology and clinical history is more important than an immunohistochemical staining pattern.23
 Figure 2.68. Higher power image of clinging nature of carcinoma in situ (CIS), where cells are detaching from the surface and focally denuded. |
 Figure 2.69. Carcinoma in situ (CIS) may spread in a pagetoid fashion, where individual or nests of malignant cells percolate through the benign urothelium. |
TABLE 2.1: Most Common Immunohistochemical Patterns for Distinction of CIS From Reactive Epithelium | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||
In the absence of thermal injury from electrocautery, it is important to recognize that entirely denuded urothelium should not be diagnosed as benign, and a comment raising the possibility of a denuded CIS with the suggestion of obtaining urine cytology should be included (Figures 2.72,2.73,2.74).31,32
SAMPLE NOTE: Denuded Urothelial Mucosa (See Note)
Note: Denudation of the urothelium may occur when dyscohesive CIS cells detach from the lamina propria and exfoliate into the urine. Urine cytology or additional sampling may be useful in this setting.
 Figure 2.74. At high power, the intact urothelial cells from Figure 2.73 show high-grade atypia with increased nuclear size and hyperchromasia, sufficient for a diagnosis of carcinoma in situ (CIS). |
PEARLS AND PITFALLS: CIS Involving von Brunn Nests
CIS commonly involves VBN, as they represent a downward growth into the lamina propria which is in continuity with the surface (Figures 2.75 and 2.76)
VBN involved by CIS usually appears as moderate to large nests with rounded contours and lack of specific features of invasion (no retraction artifact, small or irregular nests, or infiltrating single cells)
PAPILLARY LESIONS OF THE BLADDER—UROTHELIAL
Papillary lesions of the bladder are evident by exophytic papillary fronds, projecting into the lumen of the bladder and readily identified on cystoscopic evaluation. The cystoscopy procedure note is of paramount importance when diagnosing papillary lesions, especially if the histologic appearance is equivocal for true papillary architecture. A helpful urologist
will comment on the exact nature of the lesion, often using descriptors such as papillary, polypoid, sessile, low-grade appearing, etc. Their description can help confirm a histologic impression or prompt deeper levels on a specimen to identify a possible small papillary lesion when none is identified on initial sections.
will comment on the exact nature of the lesion, often using descriptors such as papillary, polypoid, sessile, low-grade appearing, etc. Their description can help confirm a histologic impression or prompt deeper levels on a specimen to identify a possible small papillary lesion when none is identified on initial sections.
 Figure 2.77. Whole slide image of a TURBT (transurethral resection of bladder tumor) fragment demonstrating the broad mucosal folds of polypoid cystitis. |
Papillary/Polypoid Cystitis
Before discussion of true urothelial neoplasms, it is important to be aware of the benign mimickers of papillary neoplasms. A true papillary neoplasm is defined by the presence of true fibrovascular cores. Without well-formed papillae, all caution is warranted to avoid overdiagnosis of a neoplasm. Papillary/polypoid cystitis is one potential pitfall in this differential diagnosis.33 The classic appearance of polypoid cystitis is the presence of broad, edematous folds of urothelial mucosa and lamina propria (Figure 2.77). Unlike a true papillary neoplasm, there are no long, thin papillary projects with fibrovascular cores. The lamina propria is markedly edematous, often appearing as a cleared out space with the urothelium floating above (Figures 2.78 and 2.79). Inflammation is usually evident in both the lamina propria and within the urothelium (Figure 2.80). Given the inflammatory and reactive nature of these lesions, a clinical history of recent catheter use, indwelling catheter, or stones may be solicited from the medical record.
PEARLS AND PITFALLS: Polypoid Cystitis
Low-power clues include broad folds with pale, edematous lamina propria and inflammatory cells
Overlying urothelium may be thickened and show reactive epithelial changes; however, no more than minimal atypia should be present in these cases
Over time, polypoid cystitis may become more papillary in nature as inflammation resolves and fibrosis contracts the broad folds into smaller papillary fronds (Figures 2.81,2.82,2.83)
In papillary/polypoid cystitis, fronds lack the true fibrovascular cores found in papillary neoplasms
Identifying the overall inflammatory context is important for accurate diagnosis of these lesions
 Figure 2.78. At low power, the broad folds and underlying edematous lamina propria are a distinct appearance from a true papillary neoplasm. |
 Figure 2.79. At higher power, polypoid cystitis shows only minimal reactive changes in the urothelium. The lamina propria is edematous and pale, bulging into the lumen of the bladder. |
 Figure 2.80. Within the lamina propria, a chronic inflammatory infiltrate is readily identifiable in most cases of polypoid cystitis. |
Papillary Hyperplasia
Another quasi-papillary lesion to consider in this spectrum of lesions is papillary urothelial hyperplasia, also referred to as urothelial proliferation of uncertain malignant potential by the WHO. While this lesion lacks true fibrovascular cores, it may raise the possibility of a true neoplasm in small biopsy specimens. The diagnosis of papillary urothelial hyperplasia should be used only when limited folds or “tenting” of the urothelium are present. Papillary urothelial hyperplasia will show a wave-like periodicity to the mucosal folds, leading to a corrugated appearance of the urothelium (Figure 2.84). True fibrovascular cores are absent, though small capillary-sized vessels may be present at the tips of the folds. Papillary urothelial hyperplasia may represent a precursor lesion to low-grade papillary urothelial carcinoma and has been noted associated with established low-grade papillary carcinomas as a “shoulder” lesion (Figure 2.85).34,35 In biopsy settings, the diagnosis of papillary hyperplasia should be reserved for lesions not directly associated with low-grade papillary urothelial carcinoma.
 Figure 2.82. At higher power, this polypoid/papillary cystitis frond lacks fibrovascular cores; instead, it shows contracted and fibrotic fronds with lamina propria edema and inflammation. |
Benign Urothelial Papilloma
Now that we have discussed mimickers of papillary neoplasms, we can begin to address true papillary urothelial neoplasms. These lesions are exophytic lesions with true fibrovascular cores lined by urothelium (Figure 2.86). Referring again to the checklist, diagnosing papillary lesions of the bladder also begins with an assessment of the urothelial thickness. Once the papillary architecture has been identified, the thickness of the urothelium will help to focus the diagnostic possibilities. Papillary lesions with normal thickness urothelium that lacks atypia are considered benign urothelial papillomas. In addition to well-formed papillary cores, papillomas should have a urothelial lining that closely approximates that of normal flat benign urothelium, 5 to 7 cells thick and lacking atypia. At low power, urothelial papillomas often appear as pale eosinophilic to clear lesions, due to the small nuclei and ample cytoplasm that often has clearing (Figures 2.87 and 2.88). If stretched out flat, the urothelial lining of a papilloma looks the same as benign urothelium lining the bladder (Figure 2.89). The papillary fronds are usually very slender and long, a feature that may be remarked on in the cystoscopy note and lead the urologist to suggest a papilloma or “low-grade appearance.” Patients with benign papillomas are often younger than those diagnosed with papillary carcinoma, and rarely progress to higher grade disease.36,37
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree