Antimicrobial Surfaces
Nicola J. Irwin
Colin P. McCoy
Matthew P. Wylie
Sean P. Gorman
Healthcare-associated infections (HAIs) constitute a major challenge for hospitals worldwide and pose a significant risk to patient health. The most recent prevalence surveys report respective HAI incidence rates of 4% and 4.5% in the US and UK health care systems and direct treatments costs of $9.8 billion and $8.1 billion in the United States and Europe, respectively.1,2,3,4 In a recent Scotland-based study, 57.1% of all reported HAIs were attributed to lower respiratory tract infections, urinary tract infections (UTIs), and bloodstream infections, of which 28.0%, 48.7% and 25.0% of these respective infections were associated with the presence of an indwelling device.1 Indeed, microbial colonization of hospital surfaces, and subsequent cross-contamination of infecting pathogens, is now widely accepted as a predominant source of HAIs.5 On account of this surface “reservoir” of infection and the increasing prevalence of antibiotic-resistant bacterial strains, there has been widespread implementation of public health guidelines for hygiene control in health care settings over recent years.6 Poor adherence to these guidelines and the reported inefficiency of cleaning and disinfection procedures to eradicate contamination have fuelled research into the development of materials with inherent antimicrobial properties for use in medical devices and general health care environments to provide an extra layer of protection from fouling pathogens.7,8
The focus on the surface results from the preference of bacteria to live as sessile communities, known as biofilms, rather than as free-flowing cells in suspension (see chapter 67). Indeed, many pathogens have been found to persist on surfaces for periods exceeding 5 months at concentrations sufficient for transmission and subsequent establishment of infection.8 Biofilm development can be broadly categorized in six stages, as illustrated in Figure 73.1. Briefly, this process involves the initial reversible attachment of free-floating planktonic bacteria to a surface, after which the bacterial secretion of extracellular polymeric substances promotes a “gluing” effect at the cell-substrate interface, leading to irreversible cellular attachment.9,10,11 Accumulation of cells and continued secretion of an exopolysaccharide matrix result in formation of a diverse and highly coordinated bacterial community in which cells undergo phenotypic variation and exhibit significant resistance to external and internal stresses, such as shear flow, host defense systems, and, more importantly, antimicrobial agents. The subsequent shedding of surface layers enables dispersal and colonization of distant surfaces, facilitating the spread of infection.12,13,14 Eradication of biofilm-associated infections is therefore a major challenge, with some studies highlighting the need for antibiotic concentrations up to 1000-fold higher than those used for treatment of their planktonic counterparts.15
In light of the escalating incidence of antibioticresistant bacteria, prevention of HAIs is now of paramount importance. The role of antimicrobial surfaces in preventing HAIs stems from their promising ability to inhibit microbial colonization of, for example, medical devices and health care environments such as floors, textiles, bed rails, door handles, and sinks, thereby preventing subsequent biofilm formation and, ultimately, transmission of antimicrobial-resistant pathogens.7
An antimicrobial surface has traditionally been described as a surface containing an antimicrobial agent that inhibits survival of microorganisms.16 This was based on early development of contact-killing surfaces prepared from deposition of, or coating with, an antimicrobial agent such as silver or copper. McLean et al17 were one of the first groups to show the bactericidal effects of catheter surfaces coated with a silver-copper film against Staphylococcus aureus, Staphylococcus epidermidis, and Pseudomonas aeruginosa. The term has since evolved to include a growing combination of surfaces that prevent bacterial colonization through both lethal and nonlethal mechanisms.18 As modern health care continues to develop, people are living longer and health care systems are therefore faced with a growing influx of vulnerable
patients requiring treatment or repair of failing physiological functions.19 The record numbers of hospital admissions coupled with the increasing reliance of medical devices in routine patient care make prevention of surface fouling and transmission of bacteria and other microbes of paramount importance.20
patients requiring treatment or repair of failing physiological functions.19 The record numbers of hospital admissions coupled with the increasing reliance of medical devices in routine patient care make prevention of surface fouling and transmission of bacteria and other microbes of paramount importance.20
This chapter discusses the current development and implementation of antimicrobial surfaces in the manufacture of medical devices and high-touch surfaces as a primary mechanism of addressing the high-priority challenge of HAIs. Many of these concepts may also apply in reducing similar risks in other applications such as industrial, manufacturing, food handling, and public health situations.
Adhesion of bacteria to a material surface is governed by several factors, including characteristics of the adhering bacteria, such as surface charge, hydrophobicity, and the presence of surface appendages, in addition to properties of the surface itself, such as polarity, roughness, surface energy, and porosity.21,22 Antiadherent surfaces are based on physical or chemical modifications of a material surface to reduce or eliminate the attachment of bacterial cells or other microorganisms.23
Superhydrophilic Surfaces
In terms of surface wettability, water contact angles between 54 and 130 degrees have been reported to be favorable for adsorption of bacterial peptidoglycan.24 Superhydrophilic surfaces are surfaces with a water contact angle below 5 degrees. Their self-cleaning properties are attributed to the pooling and rapid spread of water droplets, which pick up associated debris as they roll across the surface, as illustrated in Figure 73.2.25
Methods to increase surface hydrophilicity include plasma treatment, exposure to ultraviolet radiation, and electrochemical treatments.26 Reductions in surface contact angles of polycarbonate surfaces from 85 to <5 degrees have been reported on coating with titanium dioxide (TiO2) and subsequent irradiation by ultraviolet A (UVA) light.27 In another report, hydrophilization of fibrous polystyrene substrates with air plasma treatment reduced surface contact angles to 0 degree leading to complete wetting of the surface. These superhydrophilic surfaces, with a zeta potential of -40 mV, exhibited significantly greater resistance to adherence of Escherichia coli than their comparatively more hydrophobic untreated and fluorinated counterparts due to a combination of electrostatic repulsion and weakened hydrophobic interactions with the gram-negative bacterial membrane lipopolysaccharides.28 More important, the presence of repulsive surface-bound films of water on superhydrophilic substrates provides an antifouling effect by preventing hydrophobic interactions between adhering macromolecules and the underlying surface.26
Superhydrophobic Surfaces
In contrast to superhydrophilic surfaces, a superhydrophobic surface is defined as a surface possessing a water contact angle greater than 150 degrees and typically consists of micro- or nanoscale features inspired by the low-adhesion functionality of leaves of the lotus flower.28 On contacting a superhydrophobic surface, a water droplet can wet the surface in one of two modes. First, the droplet can fully penetrate the groves, producing a continuous film with a contact angle dictated by physicochemical properties of the liquid-solid interface,
known as the Wenzel state. In the second mode, known as Cassie-Baxter wetting, the water droplet is effectively suspended on a pocket of air trapped between neighboring surface protrusions, with the corresponding contact angle dictated by properties of an additional gas-air interface in addition to the liquid-solid interface present during Wenzel wetting.29 Both types of surface wetting are illustrated in Figure 73.3.
known as the Wenzel state. In the second mode, known as Cassie-Baxter wetting, the water droplet is effectively suspended on a pocket of air trapped between neighboring surface protrusions, with the corresponding contact angle dictated by properties of an additional gas-air interface in addition to the liquid-solid interface present during Wenzel wetting.29 Both types of surface wetting are illustrated in Figure 73.3.
 FIGURE 73.2 Illustration of water droplet wetting on self-cleaning (A) and superhydrophobic surfaces (B). |
 FIGURE 73.3 Water droplets on a rough surface illustrating the wetted Wenzel state (A), the Cassie-Baxter state (B), low-contact angle hysteresis (C), and high-contact angle hysteresis (D). |
In addition to water contact angles above 150 degrees, many self-cleaning superhydrophobic surfaces exhibit low contact angle hysteresis, defined as the difference between the advancing (θA) and receding (θR) contact angles of a
moving or evaporating water droplet.30,31 Contact angle hysteresis is directly proportional to the surface retention force exerted on the droplet, and when low, as is typical of most droplets in the Cassie-Baxter state, droplets can roll and slide over the surface with little resistance at low tilt angles, collecting surface debris, and other particulate matter as they pass.32
moving or evaporating water droplet.30,31 Contact angle hysteresis is directly proportional to the surface retention force exerted on the droplet, and when low, as is typical of most droplets in the Cassie-Baxter state, droplets can roll and slide over the surface with little resistance at low tilt angles, collecting surface debris, and other particulate matter as they pass.32
The design and synthesis of self-cleaning superhydrophobic surface analogues mimicking the nonfouling nanoarchitectures of highly water-repellant plant leaves, such as the lotus leaf, has been aided by the parallel investigation of mechanisms responsible for their self-cleaning effects and the associated development of advanced nanofabrication methods, including reactive-ion beam etching, nanoimprint lithography, and laser ablation.33 By studying the dynamics of droplet impaction on biomimetic hierarchical surfaces fabricated using the Tobacco mosaic virus, McCarthy et al34 have experimentally demonstrated the role of the surface nanoscale features in providing high antiwetting capillary pressures and the distinct, yet complementary, role of surface microstructures in pressure dissipation, thereby impeding transition to a fully wetted state.35
Although superhydrophobic surfaces have gained much attention for their self-cleaning properties, particularly regarding their lotus-like ability to remove particulate matter, in light of the escalating incidence of antimicrobial resistance and the toxic properties of many biocides, there has been increasing interest in understanding the potential of these surfaces to resist contamination and therefore help combat HAIs when used in health care environments.36,37 In studies conducted to date, however, the inherent microbiological resistance of these surfaces have been found to vary widely. For instance, bioinspired highly rough films of silicone elastomer with water contact angles of 165 degrees, prepared via an aerosol-assisted chemical vapor deposition process, achieved modest reductions in adherence of E coli and S aureus of 38% and 63%, respectively, relative to dip-coated glass controls. Despite a lower overall degree of contact between the elastomer and aqueous bacterial suspensions as a result of the Cassie-Baxter wetting state of the superhydrophobic surface, colonization continued to proceed at the tips of the surface protrusions.37 In addition, although adhesion of P aeruginosa was completely inhibited on two-tier microscale- and nanoscale-patterned superhydrophobic titanium surfaces with water contact angles of 166 degrees, fabricated by laser ablation to mimic the lotus leaf topography, increased colonization of S aureus was reported relative to the unmodified smooth surfaces.38 Indeed, the selective antifouling behavior observed in this study highlights the need for further investigation of the mechanisms controlling bacterial attachment to surfaces to more fully understand factors governing the antiadhesive properties of biomimetic superhydrophobic surfaces. Moreover, the finite sustainability of the Cassie-Baxter state of wetting as a result of diffusion of the air pockets into water upon exposure to conditions of high hydrostatic pressure, temperature, humidity, or shear flow limits their potential utility in, for example, nonfouling marine applications or implanted medical devices. The corresponding transition from the metastable Cassie-Baxter state to the Wenzel state leads to complete wetting of the surface.35,39
In addition to physical texturing of the surface as a mechanism to engineer superhydrophobic surfaces, functionalization of polyvinyl chloride with low surface energy fluorinated compounds has generated interest for the development of superhydrophobic surfaces inhibiting the early stages of bacterial adhesion and ultimately preventing biofilm formation on medical devices, such as endotracheal tubes.40 Longevity of the nonfouling properties of superhydrophobic surfaces remains an issue due to masking of the low-energy functionalized surface groups and/or the change in surface structure resulting from attachment of conditioning layers of macromolecules and proteins.41 One example relates to the heavy surface fouling of roughened superhydrophobic polysiloxane surfaces by macroalgae, bryozoans, and barnacles observed after only 2 months immersion in seawater in contrast to their high resistance to fouling over a 6-month period of exposure to mixed cultures of naturally occurring microfoliant when submerged in tap water.42 Exposure to shear flow has been reported as an efficacious method of removing surface-adsorbed protein from nanoscale-roughened surfaces; however, this is not a feasible approach in many applications.43 Therefore, although superhydrophobic surfaces exhibit promising potential for short-term antifouling applications or in situations where periodic resurfacing can be facilitated, further investigation of their antifouling behavior over extended periods is needed to increase longevity of their self-cleaning properties.42
Slippery Liquid-Infused Porous Surfaces
A novel self-cleaning surface inspired by the Nepenthes pitcher plant has recently been developed, possessing similar textured features to those of superhydrophobic surfaces. Instead of trapping air within the grooves of the surface, however, the nano- and microstructured features entrap lubricating liquid to create self-healing, slippery liquid-infused porous surfaces (SLIPS).44,45 These SLIPS demonstrate extreme temperature and pressure stability and superior antiwetting behavior in comparison to their air-entrapped microtextured counterparts.44 The infused lubricant becomes locked in place through cohesive forces, as illustrated in Figure 73.4.
Stability of SLIPS has been found to be dependent on three key criteria. First, the affinity of the porous substrate to the lubricating liquid must be greater than the affinity of the surface to the liquid to be repelled. Second, the lubricant must be able to infiltrate, wet and robustly adhere
to the substrate and, third, the lubricating liquid must be immiscible with the repellant fluids.46 In contrast to superhydrophobic, lotus leaf-inspired, Cassie-Baxter-type surfaces, which conventionally favor multitiered, hierarchical structures for improved robustness and pressure tolerance, enhanced stability to high-simulated shear and robust nonfouling properties have been observed on uniformly nanostructured SLIPS surfaces. This phenomenon has been attributed to the formation of a stable smooth overcoating of lubricant as a result of the comparatively smaller length scales of the nanotextured structures than the capillary length of the lubricant layer. Even at high spin speeds of 10 000 rpm, where this layer was compressed under gravitational forces to approximately 400 nm, surfaces with nanoscale features approximately 170 nm tall exhibited no signs of deterioration in antiwetting performance.47
to the substrate and, third, the lubricating liquid must be immiscible with the repellant fluids.46 In contrast to superhydrophobic, lotus leaf-inspired, Cassie-Baxter-type surfaces, which conventionally favor multitiered, hierarchical structures for improved robustness and pressure tolerance, enhanced stability to high-simulated shear and robust nonfouling properties have been observed on uniformly nanostructured SLIPS surfaces. This phenomenon has been attributed to the formation of a stable smooth overcoating of lubricant as a result of the comparatively smaller length scales of the nanotextured structures than the capillary length of the lubricant layer. Even at high spin speeds of 10 000 rpm, where this layer was compressed under gravitational forces to approximately 400 nm, surfaces with nanoscale features approximately 170 nm tall exhibited no signs of deterioration in antiwetting performance.47
To date, SLIPS have primarily been investigated for potential application as anti-icing coatings, with only limited assessment of their antibiofouling properties.47,48 One such study reported the resistance of water-repellent slippery surfaces of microporous poly(butyl methacrylateco-ethylene glycol dimethacrylate) (BMA-EDMA) infused with a selection of fluorocarbon lubricants, including Krytox 103 and Krytox 100, to attachment of marine microorganisms, namely zoospores of the alga Ulva linza and cypris larvae of the barnacle Balanus amphitrite.49 Furthermore, attachment of P aeruginosa biofilm was reduced by up to 99.6% on SLIPS prepared from porous TeflonTM membranes with perfluorinated lubricating fluids relative to porous polytetrafluoroethylene (PTFE) membrane controls, based on crystal violet absorbance, following a 7-day challenge period in infected flow cells. Adherence of S aureus and E coli to the SLIPS was reduced by 97.2% and 96%, respectively, in comparison to PTFE controls over 48 hours under identical flow conditions.50 Similarly, Li et al51 demonstrated resistance of BMA-EDMA SLIPS to biofilm formation of various strains of opportunistic P aeruginosa for up to 7 days in low-nutrient medium. After a 7-day incubation period, 1.8% and approximately 55% of the surfaces of slippery BMA-EDMA and uncoated glass controls, respectively, were covered by the P aeruginosa PA49 strain.51 With regard to their potential application in antimicrobial medical devices, Leslie et al52 have reported antithrombogenic and potent antibiofouling properties of medical tubing modified with SLIPS technology, specifically a tethered liquid perfluorocarbon (TLP) surface. More important, an 8-fold reduction in P aeruginosa biofilm formation was reported on TLP-coated loops relative to control tubing after more than 6 weeks incubation in a continuous flow model.52
The SLIPS technology undoubtedly holds promising potential for preventing biofilm formation by common bacterial pathogens; however, careful consideration of factors such as the type of lubricant, substrate, durability, and the degree and pattern of surface roughness is required for development of an efficacious SLIPS to prevent contamination while remaining nontoxic to patients.51
Antiadherent Strategies Based on Bacterial Targets
Bacterial colonization and subsequent biofilm formation is controlled by a plethora of structural and signaling pathways that have, to date, provided key targets for the action of many conventional antibiotics. On a structural level, bacterial appendages, such as pili and flagella, are important in the anchoring of bacteria to a solid surface, whereas macromolecules, such as enzymes and proteins, are involved in the signaling and regulation of crucial steps during biofilm formation, cell aggregation, swarming, and exopolysaccharide production, in a process of quorum sensing (QS).53
A rationale strategy for development of antiadhesive surfaces would therefore involve interference with structural and operational targets of bacteria, for example, by immobilization of inhibitors to facilitate downregulation of key processes involved in bacterial attachment and propagation.54 Covalent immobilization of virstatin, a small molecule which interferes with the formation of type IV
bacterial pili, onto silanized surfaces of silicone was shown to reduce adhesion of Acinetobacter baumannii by up to 46% relative to silanized controls after 4 hours incubation.55 Similarly, covalent attachment of the QS inhibitors, dihydropyrrolones, onto glass substrates reduced adherence of P aeruginosa and S aureus by up to 97% in comparison to untreated controls. Immobilized dihydropyrrolones could disrupt QS of P aeruginosa by interference with the N-acyl homoserine lactone-mediated las QS system, as demonstrated by the observed repression of a lasB-gfp reporter protein of P aeruginosa in vitro.56 This approach has been extended further by combining quorum-quenching and matrix-degrading enzymes into urinary catheter coatings. The enzymes acylase and α-amylase, which degrade bacterial QS molecules and biofilm exopolysaccharides, respectively, were incorporated into multilayer coatings on silicone catheters and challenged with single-species infections of S aureus and P aeruginosa, and mixed-cultures of P aeruginosa and E coli in a catheterized bladder model. In dynamic bladder model conditions, coatings with acylase deposited as the outermost layer demonstrated comparatively higher efficacy against clinically relevant gram-negative pathogens, with reductions in biofilm mass of 70%, 50%, and 15%, relative to uncoated controls, after 7 days incubation with P aeruginosa, dual-species P aeruginosa and E coli, and S aureus challenges, respectively. In contrast, coatings with amylase as the outermost layer were most active against the gram-positive bacteria, with reductions in biofilm formation of 30% and 10% when infected with S aureus and P aeruginosa, respectively. Furthermore, after 7 days in an in vivo rabbit model, coatings with acylase as the outermost layer reduced biofilm formation by up to 70% on the balloon portion of the catheter and delayed the spread of biofilm along the inner side of the catheter shaft by 30% in comparison to uncoated silicone controls.57 These encouraging in vivo results, coupled with the absence of toxicity to human cells in vitro, demonstrate the promising potential of this enzyme-integrated strategy for antifouling medical device coatings.
bacterial pili, onto silanized surfaces of silicone was shown to reduce adhesion of Acinetobacter baumannii by up to 46% relative to silanized controls after 4 hours incubation.55 Similarly, covalent attachment of the QS inhibitors, dihydropyrrolones, onto glass substrates reduced adherence of P aeruginosa and S aureus by up to 97% in comparison to untreated controls. Immobilized dihydropyrrolones could disrupt QS of P aeruginosa by interference with the N-acyl homoserine lactone-mediated las QS system, as demonstrated by the observed repression of a lasB-gfp reporter protein of P aeruginosa in vitro.56 This approach has been extended further by combining quorum-quenching and matrix-degrading enzymes into urinary catheter coatings. The enzymes acylase and α-amylase, which degrade bacterial QS molecules and biofilm exopolysaccharides, respectively, were incorporated into multilayer coatings on silicone catheters and challenged with single-species infections of S aureus and P aeruginosa, and mixed-cultures of P aeruginosa and E coli in a catheterized bladder model. In dynamic bladder model conditions, coatings with acylase deposited as the outermost layer demonstrated comparatively higher efficacy against clinically relevant gram-negative pathogens, with reductions in biofilm mass of 70%, 50%, and 15%, relative to uncoated controls, after 7 days incubation with P aeruginosa, dual-species P aeruginosa and E coli, and S aureus challenges, respectively. In contrast, coatings with amylase as the outermost layer were most active against the gram-positive bacteria, with reductions in biofilm formation of 30% and 10% when infected with S aureus and P aeruginosa, respectively. Furthermore, after 7 days in an in vivo rabbit model, coatings with acylase as the outermost layer reduced biofilm formation by up to 70% on the balloon portion of the catheter and delayed the spread of biofilm along the inner side of the catheter shaft by 30% in comparison to uncoated silicone controls.57 These encouraging in vivo results, coupled with the absence of toxicity to human cells in vitro, demonstrate the promising potential of this enzyme-integrated strategy for antifouling medical device coatings.
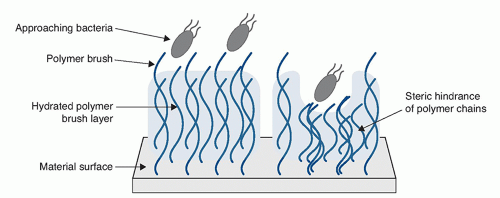 FIGURE 73.5 Hydrated polymer brush surface preventing bacterial adhesion by steric and osmotic repulsion. |
An increased knowledge and understanding of the bacterial components involved in surface adhesion and regulation of biofilm development, and the associated identification of new molecular targets, is expected to further fuel developments in this area of target-based antifouling surfaces.58
Polymer Brushes
Polymer brushes are assemblies of polymer chains tethered at one end to a surface or interface, through covalent attachment or physical adsorption, with sufficient density that the polymer chains stretch away from the surface into the surrounding aqueous medium.59 Compression of the highly hydrated polymer layer on bacterial approach generates a repulsive osmotic force which, in combination with the reduction in conformational entropy, can significantly reduce particle deposition, bacterial attachment, and resultant surface adhesion, as shown in Figure 73.5.60
Polymer chains can be tethered to the surface via the “grafting to” or “grafting from” technique. In the former method, polymerized chains with reactive end-groups, for example, carboxylic, amino, or thiol groups, are attached to a polymer substrate; however, grafting density is limited by the steric barrier generated by previously tethered chains toward the incoming macromolecules. Conversely, in the latter method, polymer chains are grown via surface-initiated polymerization from a substrate functionalized with polymerization initiators, which typically facilitates higher grafting densities and film thicknesses.59,61
A common method of producing a polymer brush surface is through a process known as (PEG)ylation, whereby chains of the polymer, PEG, are grafted onto a surface to prevent protein adsorption and cell adhesion.62 With respect to their antimicrobial abilities, PEGylated surfaces have demonstrated efficacy in delaying biofilm formation of staphylococci and enabling more facile removal of attached biofilm under shear flow.63
Reduced adhesion of S epidermidis, P aeruginosa, Candida albicans, and Candida tropicalis has been observed on surfaces grafted with PEG chains of molecular weights of 526, 2000, and 9800 Da and corresponding chain lengths of 2.8, 7.5, and 23.7 nm, respectively, relative to pristine glass slides. The effect of molecular weight on adherence was found to be most significant for the smaller bacterial cells than the larger yeast cells, with comparable adhesion of C albicans observed between all three brush lengths, and reduced attachment of S epidermidis and P aeruginosa reported on the longer brushes than on the 2.8 nm brush, as a result of the greater distance of separation from the surface and the corresponding attenuation of the long-range Lifshitz-van der Waals attractions.64,65 Al-Ani et al66 have explored the effect of PEG chain grafting density on adherence of proteins, bacteria, and cells by employing different concentrations of the surface initiator, (3-aminopropyl)triethoxysilane, and grafting PEG in solutions of varying salt conditions. The authors observed that adsorption of proteins and human MG63 osteoblast-like and mesenchymal cells was minimal on high polymer density surfaces but increased in a surfaceand cell-dependent manner at low and medium grafting densities. With regard to bacterial adherence, attachment of P aeruginosa showed a clear relationship with PEG chain density and, accordingly, the high-density PEG layers demonstrated the highest fouling resistance.66
Reduced adhesion of S epidermidis, P aeruginosa, Candida albicans, and Candida tropicalis has been observed on surfaces grafted with PEG chains of molecular weights of 526, 2000, and 9800 Da and corresponding chain lengths of 2.8, 7.5, and 23.7 nm, respectively, relative to pristine glass slides. The effect of molecular weight on adherence was found to be most significant for the smaller bacterial cells than the larger yeast cells, with comparable adhesion of C albicans observed between all three brush lengths, and reduced attachment of S epidermidis and P aeruginosa reported on the longer brushes than on the 2.8 nm brush, as a result of the greater distance of separation from the surface and the corresponding attenuation of the long-range Lifshitz-van der Waals attractions.64,65 Al-Ani et al66 have explored the effect of PEG chain grafting density on adherence of proteins, bacteria, and cells by employing different concentrations of the surface initiator, (3-aminopropyl)triethoxysilane, and grafting PEG in solutions of varying salt conditions. The authors observed that adsorption of proteins and human MG63 osteoblast-like and mesenchymal cells was minimal on high polymer density surfaces but increased in a surfaceand cell-dependent manner at low and medium grafting densities. With regard to bacterial adherence, attachment of P aeruginosa showed a clear relationship with PEG chain density and, accordingly, the high-density PEG layers demonstrated the highest fouling resistance.66
In addition to PEG, polymer brush designs based on neutral hydrophilic polymers, including polyacrylamide and poly(2-alkyl-2-oxazoline), have demonstrated promising abilities to resist protein and bacterial adhesion in vitro.60 Alternatively, zwitterionic polymers, such as poly(2-methacryloyloxyethyl phosphorylcholine), poly(sulfobetaine methacrylate) (PSBMA), poly(N4-(2-methacrylamidoethyl) asparagine) (pAspAA), and poly(N5-(2-methacrylamidoethyl) glutamine) (pGluAA), have been employed as candidates in the development of nonfouling polymer brush surfaces.60,67,68 A UV-induced graft polymerization of zwitterionic PSBMA onto hydrophobic microporous polypropylene membranes (MPPMs) at a surface density of 560 µg/cm2 enhanced surface hydrophilicity, characterized by a decrease in water contact angle from 145 to 15 degrees. The observed significant reduction in fouling by bovine serum albumin and lysine was attributed to steric repulsion from the hydrated polymer chains on approach of proteins to the surface. Additionally, PSBMA-modified MPPMs completely inhibited adhesion of E coli, Pseudomonas fluorescens, and S aureus over a 3 hour incubation period.69
Current research is aimed at designing bifunctional polymer brush coatings, via the localization of antimicrobial agents onto the polymer chain ends, in an effort to optimize their inherent antiadhesive properties and impart the polymers with lethal activity. For example, the conjugation of an optimized series of antimicrobial peptides (AMPs) to amine-functionalized polymer chains on titanium substrates demonstrated that, despite a possible alteration in the bactericidal mechanism of the AMPs following polymer chain immobilization, broad-spectrum antimicrobial activities were retained. The degree of resistance to bacterial adherence was dependent on the specific conjugated peptide but, in all cases, exceeded that of control copolymer brushes devoid of AMPs, both in vitro and in vivo. Furthermore, the coatings were nontoxic to mammalian cells, compatible with human serum, and displayed antimicrobial properties that could be readily optimized through modification of the copolymer composition and grafting density.70 Alternatively, a high-efficiency antibacterial surface, demonstrating both antiadherent and microbicidal activities toward Actinomyces naeslundii and E coli, has been developed through conjugation of the antimicrobial triclosan to zwitterionic PSBMA polymer brushes on a silicone substrate.71
Polymer brush surfaces could therefore act as useful “carrier platforms” for antimicrobial agents, with potential benefits of long-term stability, improved biocompatibility of tethered agents, and robustness, in addition to their inherent antiadhesive properties.
Active, or bactericidal, surfaces possess the ability to physically or chemically disrupt cells leading to their death, either before or shortly after attachment to the surface.72
Antibiotic-Incorporated Surfaces
Many early release-based systems were developed via impregnation of surfaces with antimicrobial agents. These first-generation coatings typically exhibit first- or second-order release kinetics, characterized by an initial burst release of drug followed by a much slower rate of prolonged release. A combination of rifampicin and minocycline in urinary catheter coatings has been found to successfully reduce gram-positive bacteriuria, and, with the addition of chlorhexidine, broad-spectrum coverage against gram-positive and gram-negative bacteria, and fungi, has been reported during short-term implantation of antimicrobial-coated central venous catheters.73,74 In addition, gentamicin-loaded polymer bone cements have been well described in the literature, and a review of these products has shown their characteristic matrix-style release properties, with a high initial burst release during the first few hours of surface wetting, after which the rate of release significantly declines.75 Although this release profile is beneficial during the critical first few hours postimplantation, infection-resistance may be required for several weeks or months to facilitate implant-tissue integration as well as providing antimicrobial cover during checkup or resection procedures. Initial attempts to improve the release kinetics of these first-generation coatings
typically involved promotion of polymer-antibiotic or antibiotic-antibiotic interactions within polymer matrices to retard release. For example, Piozzi et al76 loaded a series of acidic and basic polyurethanes with antibiotics containing at least one acidic functional group, including amoxicillin, cefamandole nafate, rifampicin, and vancomycin. Similarly, Ruggeri et al77 reported synergistic effects of polyurethane-loaded rifampicin and cefamandole nafate against rifampicin-resistant S aureus and, moreover, when formulated with the pore-former PEG 10 000, activity was maintained for up to 23 days.
typically involved promotion of polymer-antibiotic or antibiotic-antibiotic interactions within polymer matrices to retard release. For example, Piozzi et al76 loaded a series of acidic and basic polyurethanes with antibiotics containing at least one acidic functional group, including amoxicillin, cefamandole nafate, rifampicin, and vancomycin. Similarly, Ruggeri et al77 reported synergistic effects of polyurethane-loaded rifampicin and cefamandole nafate against rifampicin-resistant S aureus and, moreover, when formulated with the pore-former PEG 10 000, activity was maintained for up to 23 days.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




