Key Points
Disease summary:
Hematopoietic stem cell transplantation (HSCT) is a curative therapy for a number of malignant and nonmalignant conditions including leukemia, bone marrow failure, severe immunodeficiencies, and other disorders involving the hematopoietic system such as thalassemia and sickle cell anemia.
The HSCT procedure involves multiple phases including a preparative regimen consisting of cytotoxic and immune-depleting agents followed by infusion of a hematopoietic graft. Recovery of neutrophils and platelets typically occurs within 2 to 4 weeks but full immunologic recovery can take months to more than a year.
Graft sources currently being used include bone marrow, peripheral blood stem cells collected via an apheresis procedure, and cryopreserved umbilical cord blood.
Graft-versus-host disease (GVHD) represents one of the major causes of morbidity and mortality after allogeneic HSCT. However, donor-versus-recipient alloreactivity can also contribute to the curative potential of HSCT via the graft- versus-leukemia (GVL) reaction.
Degree of donor and recipient matching at the human leukocyte antigens (HLA) system encoded on chromosome 6 has a significant impact on transplant outcomes including the risk of GVHD and relapse of malignancy.
GHVD can occur even when donor and recipient are fully HLA-matched as in the matched sibling donor setting illustrating the importance of other non-HLA minor histocompatibility antigens (miHAs) such as those encoded on the Y chromosome.
Donor killer immunoglobulin-like receptor genotype encoded on chromosome 19 can also influence the potency of the GVL effect and the risk of leukemia relapse.
Polymorphisms in other genes such as those encoding cytokines, cytokine receptors, and innate immune response genes among others can also influence transplant outcomes.
Genome-wide associations:
The application of genome-wide association study (GWAS) to HSCT is relatively recent. Two studies have demonstrated that these approaches can be used to identify single-nucleotide polymorphisms (SNPs) that modulate outcomes following HSCT. Future studies will be needed to validate these findings in larger populations.
Pharmacogenomics:
Patients are frequently on multiple medications during HSCT including agents with substantial toxicity. Metabolism and clearance of many of these agents can be affected by pharmacogenomic variants (Table 174-1). Many of the medications used in HSCT are dose adjusted to achieve a target range.
Diagnostic Criteria and Clinical Characteristics
Primary factors affecting outcome after HSCT are GVHD, infection, organ toxicity, and relapse or recurrence of the underlying disease for which the HSCT is being performed. GVHD is a disorder arising from immunologic targeting of recipient tissues by donor cells. Due to better supportive care resulting in lower mortality from preparative regimen-related toxicity or infection, GVHD and relapse have become the primary causes of poor outcome. However, despite GVHD-associated morbidity and mortality, it is well established that donor immune reactivity against leukemia (the GVL effect) improves survival. Therefore, a major focus of HSCT research has sought to separate the harmful effects of GVHD from the beneficial effects of GVL at the molecular level. Genomics has played a role in this and other aspects of HSCT for over two decades, since the initial observation that matching at the HLA system resulted in reduced GVHD and improved outcomes.
Diagnostic evaluation should include at least one of the following:
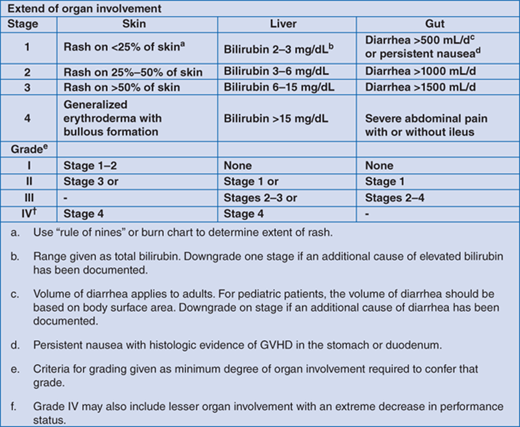
Scoring for GVHD is clinical, however, due to the large differential diagnosis and implications of delayed diagnosis for some of these conditions (eg, infection), prompt histologic confirmation of GVHD is often required to ensure appropriate therapy is initiated in a timely manner (Fig. 174-1).
The diagnostic evaluation depends on the organ system involved.
When there is cutaneous involvement, skin biopsies can be performed rapidly.
Biopsies of the gastrointestinal tract or liver may also be necessary in the event that there are no skin manifestations or the skin biopsies are inconclusive.
Chronic GVHD typically manifests with symptoms analogous to those seen in autoimmune diseases such as scleroderma and lupus. Biopsies of involved organs as well as evaluation of organ-targeted antibodies can aid in the diagnosis.
And the absence of
Infections are the major entity in the differential diagnosis of GVHD as post-HSCT patients are highly immunocompromised and the symptomatology for both is nonspecific and overlapping.
Sinusoidal obstructive syndrome (also known as veno-occlusive disease) must also be considered in the setting of hyperbilirubinemia.
GVHD can either be acute or chronic. Initially, acute and chronic GVHD were distinguished by the time in which they occurred following HSCT (acute <100 days). It is now recognized that these entities represent different clinical and pathologic conditions and should be distinguished on this basis rather than time post-HSCT. Acute GVHD is graded according to a number of scoring systems with the modified Glucksberg criteria most commonly used (Fig. 174-1). The target organs utilized are skin, liver (hyperbilirubinemia), and gastrointestinal tract. Chronic GNHD was originally characterized as limited (skin only) or extensive (other organs systems besides skin involved) but the recently developed National Institute of Health (NIH) Consensus Criteria is more predictive of outcome.
Management and Treatment
Prevention of GVHD is one of the most important considerations in HSCT. Donor lymphocytes (mainly T cells) contained in the graft and reactive against the recipient are the primary cells contributing to acute GVHD with inflammation of the target organs from the preparative regimen initiating the cascade of events leading to GVHD. Removal of T cells is effective at preventing GVHD but results in prolonged lymphocyte deficiency and increased risk of graft failure, infection, and relapse of malignancy. The most common type of transplant utilizes a T cell-containing unmanipulated graft with immunosuppression given to the recipient for GVHD prophylaxis. Typical prophylactic regimens include, but are not limited to, a calcineurin inhibitor (cyclosporine A or tacrolimus) plus another agent such as methotrexate, mycophenolate mofetil or sirolimus. T-cell–targeted antibodies such as antithymocyte globulin or alemtuzumab (anti-CD52) are also used, most commonly with unrelated donors where there is likely to be greater genetic disparity and potential alloreactivity (see section later for a discussion of donor-recipient matching). Although the molecular risk factors for chronic GVHD have not been well characterized, acute GVHD represents the greatest clinical risk factor. It should be noted that the development of acute or chronic GVHD is associated with a reduced risk of relapse in patients with malignancy. Thus, the intensity of GVHD prophylaxis is often tailored based on the underlying condition for which the HSCT is being performed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree